Abstract
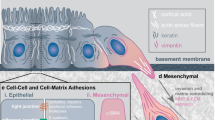
The epithelial to mesenchymal transition (EMT) is known to involve several physiological and pathological phenomena. In this study, we utilized a microplate measurement system (MMS) approach based on the deflection of a flexible micro-cantilever to measure cell stiffness (in Pa) and adhesion force (in nN) of a single cell during EMT with nN resolution. Our results demonstrated that after transforming growth factor-β1 (TGF-β1) induced EMT (tEMT), NMuMG cells became stiffer due to thicker and more abundant F-actin and displayed stronger vinculin accumulation after long-term cell-substrate adhesion. The MMS could distinguish differences in compressive stiffness (219 ± 10 and 287 ± 14 Pa), tensile stiffness (114 ± 14 and 132 ± 12 Pa), and adhesion force (150 ± 42 and 192 ± 31 nN) between cells before and after tEMT. However, without proper development of the F-actin structure and adequate adherent time, the mechanical differences were diminished. After tEMT, the cells with increased stiffness and a cell-substrate adhesion force benefited by migrating more rapidly and had more invasiveness. Thus, this technology has the potential to benefit research focused on cancer diagnosis, drug development, and cell-substrate interactions.





Similar content being viewed by others
References
A. Albini, Y. Iwamoto, H.K. Kleinman, G.R. Martin, S.A. Aaronson, J.M. Kozlowski, R.N. McEwan, Cancer Res. 47, 3239 (1987)
R.C. Bates et al., J. Clin. Investig. 115, 339 (2005)
J.R. Beach et al., Proc. Natl. Acad. Sci. U. S. A. 108, 17991 (2011)
W. Birchmeier, EMBO Rep. 6, 413 (2005)
M.A. Brown, C.S. Wallace, C.C. Anamelechi, E. Clermont, W.M. Reichert, G.A. Truskey, Biomaterials 28, 3928 (2007)
S.T. Buckley, C. Medina, A.M. Davies, C. Ehrhardt, Nanomedicine 8, 355 (2012)
D. Buergy, F. Wenz, C. Groden, M.A. Brockmann, Int. J. Cancer 130, 2747 (2012)
E. Canetta, A. Duperray, A. Leyrat, C. Verdier, Biorheology 42, 321 (2005)
O. Chaudhuri, S.H. Parekh, W.A. Lam, D.A. Fletcher, Nat. Methods 6, 383 (2009)
J. Chen, H. Li, N. SundarRaj, J.H. Wang, Cell Motil. Cytoskeleton 64, 248 (2007)
C.-C. Chen, P.C.-H. Hsieh, G.-M. Wang, W.-C. Chen, M.-L. Yeh, Mater. Let. 63, 1872 (2009)
Y.S. Chu, W.A. Thomas, O. Eder, F. Pincet, E. Perez, J.P. Thiery, S. Dufour, J. Cell Biol. 167, 1183 (2004)
M.J. Colbert, F. Brochard-Wyart, C. Fradin, K. Dalnoki-Veress, Biophys. J. 99, 3555 (2010)
S.E. Cross, Y.S. Jin, J. Tondre, R. Wong, J. Rao, J.K. Gimzewski, Nanotechnology 19, 384003 (2008)
S.E. Cross, Y.S. Jin, Q.Y. Lu, J. Rao, J.K. Gimzewski, Nanotechnology 22, 215101 (2011)
N. Desprat, A. Guiroy, A. Asnacios, Rev. Sci. Instrum. 77, 055111 (2006)
D. Docheva, D. Padula, M. Schieker, H. Clausen-Schaumann, Biochem. Biophys. Res. Commun. 402, 361 (2010)
A.J. Engler, S. Sen, H.L. Sweeney, D.E. Discher, Cell 126, 677 (2006)
J. Friedrichs et al., J. Biol. Chem. 282, 29375 (2007)
J. Friedrichs, J. Helenius, D.J. Muller, Nat. Protoc. 5, 1353 (2010a)
J. Friedrichs, J. Helenius, D.J. Muller, Proteomics 10, 1455 (2010b)
J. Friedrichs, A. Zieris, S. Prokoph, C. Werner, Macromol. Rapid Commun. 33, 1453 (2012)
J. Friedrichs, K.R. Legate, R. Schubert, M. Bharadwaj, C. Werner, D.J. Muller, M. Benoit, Methods 60, 169 (2013a)
J. Friedrichs, C. Werner, D.J. Muller, Methods Mol. Biol. 1046, 19 (2013b)
A. Fuhrmann, J.R. Staunton, V. Nandakumar, N. Banyai, P.C. Davies, R. Ros, Phys. Biol. 8, 015007 (2011)
N.D. Gallant, K.E. Michael, A.J. Garcia, Mol. Biol. Cell 16, 4329 (2005)
S. Giampieri, C. Manning, S. Hooper, L. Jones, C.S. Hill, E. Sahai, Nat. Cell Biol. 11, 1287 (2009)
F. Guilak, V.C. Mow, J. Biomech. 33, 1663 (2000)
T. Itabashi et al., Nat. Methods 6, 167 (2009)
A. Kabashima et al., Int. J. Cancer 124, 2771 (2009)
D.H. Kim, S.B. Khatau, Y. Feng, S. Walcott, S.X. Sun, G.D. Longmore, D. Wirtz, Sci. Rep. 2, 555 (2012)
M. Labelle, S. Begum, R.O. Hynes, Cancer Cell 20, 576 (2011)
W.A. Lam, M.J. Rosenbluth, D.A. Fletcher, Br. J. Haematol. 142, 497 (2008)
J. Lee, M. Leonard, T. Oliver, A. Ishihara, K. Jacobson, J. Cell Biol. 127, 1957 (1994)
M.J. Lee, J. Kim, K.I. Lee, J.M. Shin, J.I. Chae, H.M. Chung, Cytotherapy 13, 165 (2011)
M. Lekka, P. Laidler, Nat. Nanotechnol. 4, 72 (2009). author reply 72
F. Li, S.D. Redick, H.P. Erickson, V.T. Moy, Biophys. J. 84, 1252 (2003)
M.M. Lotz, C.A. Burdsal, H.P. Erickson, D.R. McClay, J. Cell Biol. 109, 1795 (1989)
S.A. Mani et al., Cell 133, 704 (2008)
J.C. Martens, M. Radmacher, Pflugers Arch. - Eur. J. Physiol. 456, 95 (2008)
T.S. Matsui, S. Deguchi, N. Sakamoto, T. Ohashi, M. Sato, Biorheology 46, 401 (2009)
P.J. Miettinen, R. Ebner, A.R. Lopez, R. Derynck, J. Cell Biol. 127(6 Pt 2), 2021 (1994)
D. Mitrossilis, J. Fouchard, A. Guiroy, N. Desprat, N. Rodriguez, B. Fabry, A. Asnacios, Proc. Natl. Acad. Sci. U. S. A. 106, 18243 (2009)
D. Mitrossilis, J. Fouchard, D. Pereira, F. Postic, A. Richert, M. Saint-Jean, A. Asnacios, Proc. Natl. Acad. Sci. U. S. A. 107, 16518 (2010)
M. Mori et al., Mol. Biol. Cell 20, 3115 (2009)
K. Nagayama, S. Yanagihara, T. Matsumoto, Med. Eng. Phys. 29, 620 (2007)
R.M. Nerem, Adv. Exp. Med. Biol. 534, 1 (2003)
G. Ofek, D.C. Wiltz, K.A. Athanasiou, Biophys. J. 97, 1873 (2009)
C. Rotsch, M. Radmacher, Biophys. J. 78, 520 (2000)
A. Saterbak, D.A. Lauffenburger, Biotechnol. Prog. 12, 682 (1996)
S. Sen, S. Kumar, Cell. Mol. Bioeng. 2, 218 (2009)
Y. Shen, M. Nakajima, S. Kojima, M. Homma, T. Fukuda, Biochem. Biophys. Res. Commun. 409, 160 (2011)
J. Solon, I. Levental, K. Sengupta, P.C. Georges, P.A. Janmey, Biophys. J. 93, 4453 (2007)
D.A. Starr, M. Han, J. Cell Sci. 116, 211 (2003)
M.P. Stewart, J. Helenius, Y. Toyoda, S.P. Ramanathan, D.J. Muller, A.A. Hyman, Nature 469, 226 (2011)
S. Suresh, Nat. Nanotechnol. 2, 748 (2007)
V. Swaminathan, K. Mythreye, E.T. O’Brien, A. Berchuck, G.C. Blobe, R. Superfine, Cancer Res. 71, 5075 (2011)
A. Taubenberger, D.A. Cisneros, J. Friedrichs, P.H. Puech, D.J. Muller, C.M. Franz, Mol. Biol. Cell 18, 1634 (2007)
A.V. Taubenberger, D.W. Hutmacher, D.J. Muller, Tissue Eng. B Rev. 20, 40 (2014)
G. Thoelking, B. Reiss, J. Wegener, H. Oberleithner, H. Pavenstaedt, C. Riethmuller, Nanotechnology 21, 265102 (2010)
O. Thoumine, A. Ott, D. Louvard, Cell Motil. Cytoskeleton 33, 276 (1996)
Q. Tseng et al., Lab Chip 11, 2231 (2011)
C.E. Turner, J.R. Glenney Jr., K. Burridge, J. Cell Biol. 111, 1059 (1990)
S. Vichare, M.M. Inamdar, S. Sen, Soft Matter, (2012a).
S. Vichare, M.M. Inamdar, S. Sen, Soft Matter 8, 10464 (2012b)
X. Wang, S. Cooper, Tissue Eng. A 19, 1113 (2012)
N. Wang, I.M. Tolic-Norrelykke, J. Chen, S.M. Mijailovich, J.P. Butler, J.J. Fredberg, D. Stamenovic, Am. J. Physiol. Cell Physiol. 282, C606 (2002)
N. Wang, J.D. Tytell, D.E. Ingber, Nat. Rev. Mol. Cell Biol. 10, 75 (2009)
C.C. Wu, H.W. Su, C.C. Lee, M.J. Tang, F.C. Su, Biochem. Biophys. Res. Commun. 329, 256 (2005)
T.-H. Wu, C.-H. Li, M.-J. Tang, J.-I. Liang, C.-H. Chen, M.-L. Yeh, Cell Commun. Adhes. 20, 115 (2013).
J. Zavadil, E.P. Bottinger, Oncogene 24, 5764 (2005)
J. Zavadil et al., Proc. Natl. Acad. Sci. U. S. A. 98, 6686 (2001)
Acknowledgments
The authors acknowledge financial support from the National Science Council of Taiwan (98-2627-B-006-009-, 99-2627-B-006-009-, 100-2627-B-006-009-).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
(DOCX 347 kb)
Figure S2
(DOCX 736 kb)
(AVI 24023 kb)
(AVI 26634 kb)
Supplementary Table 1
(DOCX 22 kb)
Rights and permissions
About this article
Cite this article
Wu, T.H., Chiou, Y.W., Chiu, W.T. et al. The F-actin and adherence-dependent mechanical differentiation of normal epithelial cells after TGF-β1-induced EMT (tEMT) using a microplate measurement system. Biomed Microdevices 16, 465–478 (2014). https://doi.org/10.1007/s10544-014-9849-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-014-9849-1