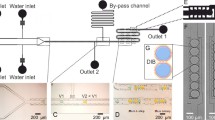
Abstract
Reconstitution of ion channels and transmembrane proteins in planar lipid bilayer membranes allow for their scientific study in highly controlled environments. Recent work with lipid bilayers formed from mechanically joined monolayers has shown their potential for wider technological application, including automation and parallelization. However, bilayer areas are highly sensitive to variations in mechanical position and the bilayers themselves cannot withstand significant perfusion of adjacent solutions. Toward this end, here we describe use of an aperture that masks the monolayer contact area, enabling formation of highly consistent bilayer areas and significantly reducing their variation with changes in relative position of the monolayers. Further, use of the aperture enables flow of solution adjacent to the bilayer without rupture or significant change in bilayer area. The device design is scalable and compatible with SBS standard instrumentation and automation technology, potentially enabling its use for rapid, parallel automated measurements of ion channels for large scale scientific studies and pharmaceutical screening.



Similar content being viewed by others
References
H. Bayley, B. Cronin, A. Heron, M.A. Holden, W. Hwang, R. Syeda, J. Thompson, M. Wallace, Mol. Biosyst. 4, 1191–1208 (2008)
K. Funakoshi, H. Suzuki, S. Takeuchi, Anal. Chem. 78, 8169–74 (2006)
A.J. Heron, J.R. Thompson, A.E. Mason, M.I. Wallace, J. Am, Chem. Soc. 129, 16042–16047 (2007)
M.A. Holden, D. Needham, H. Bayley, J. Am, Chem. Soc. 129, 8650–5 (2007)
S. Leptihn, J.R. Thompson, J.C. Ellory, S.J. Tucker, M.I. Wallace, J. Am, Chem. Soc. 133, 9370–9375 (2011)
N. Malmstadt, M.A. Nash, R.F. Purnell, J.J. Schmidt, Nano Letters 6, 1961–1965 (2006)
M. Mayer, J.K. Kriebel, M.T. Tosteson, G.M. Whitesides, Biophysical Journal 85, 2684–2695 (2003)
J.L. Poulos, T.J. Jeon, R. Damoiseaux, E.J. Gillespie, K.A. Bradley, J.J. Schmidt, Biosens. Bioelectron. 24, 1806–10 (2009)
J.L. Poulos, S.A. Portonovo, H. Bang, J.J. Schmidt, J. Phys. Condens. Matter 22, 454105 (2010)
T. Thapliyal, J.L. Poulos, J.J. Schmidt, Biosens. Bioelectron. 26, 2651–2654 (2010)
L.M. Tsofina, E.A. Liberman, A.V. Babakov, Nature 212, 681–3 (1966)
W.F. Wonderlin, A. Finkel, R.J. French, Biophys. J. 58, 289–297 (1990)
M. Zagnoni, M.E. Sandison, P. Marius, H. Morgan, Anal. Bioanal. Chem. 393, 1601–1605 (2009)
Acknowledgments
We thank Prof. Takashi Nisisako for assistance with fluid exchange experiments, Dr. Jason L. Poulos for chamber design contributions, and Ahmad El-Arabi for assistance in producing manuscript figures. Funding provided by National Science Foundation CAREER (0644442).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure
Masking apertures increase tolerance of bilayers to flow of adjacent solution. Bilayer capacitance is measured as a function of flow rate of the buffer in the lower aqueous chamber. Data shown from two separate trials forming bilayers on a 200 μm diameter aperture. Without the masking aperture, bilayer failure was immediate even for flow rates < 1 ml/hr. (DOC 49.5 kb)
Rights and permissions
About this article
Cite this article
Portonovo, S.A., Schmidt, J. Masking apertures enabling automation and solution exchange in sessile droplet lipid bilayers. Biomed Microdevices 14, 187–191 (2012). https://doi.org/10.1007/s10544-011-9596-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-011-9596-5