Abstract
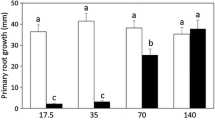
Morphological root plasticity optimizes nutrient and water uptake by plants and is a promising target to improve tolerance to metal toxicity. Exposure to sublethal chromate [Cr(VI)] concentrations inhibits root growth, decreases photosynthesis and compromises plant development and productivity. Despite the increasing environmental problem that Cr(VI) represents, to date, the Cr tolerance mechanisms of plants are not well understood, and it remains to be investigated whether root architecture remodelling is important for plant adaptation to Cr(VI) stress. In this report, we analysed the growth response of Arabidopsis thaliana seedlings to concentrations of Cr(VI) that strongly repress primary and lateral root growth. Interestingly, adventitious roots started developing, branched and allowed seedlings to grow under highly growth-repressing Cr(VI) concentrations. Cr(VI) negatively regulates auxin transport and response gene expression in the primary root tip, as evidenced by decreased expression of auxin-related reporters DR5::GFP, DR5::uidA and PIN1::PIN1::GFP, and then, another auxin maximum is established at the site of adventitious root initiation that drives adventitious root organogenesis. Both primary root growth inhibition and adventitious root formation induced by high Cr(VI) levels are blocked by a gain-of-function mutation in the SOLITARY-ROOT/IAA14 gene of Arabidopsis. These data provide evidence that suggests a critical role for auxin transport and signalling via IAA14/SLR1 in the developmental program linking Cr(VI) to root architecture remodelling.







Similar content being viewed by others
References
Barceló J, Poschenrieder C, Gunse B (1985) Effect of chromium VI on mineral element composition of bush beans. J Plant Nutr 8:211–217
Bellini C, Pacurar DI, Perrone I (2014) Adventitious roots and lateral roots: similarities and differences. Annu Rev Plant Biol 65:639–666
Benjamins R, Scheres B (2008) Auxin: the looping star in plant development. Annu Rev Plant Biol 59:443–465
Benková E, Michniewicz M, Sauer M, Teichmann T, Seifertová D, Jürgens G, Friml J (2003) Local, efflux-dependent auxin gradients as a common module for plant organ formation. Cell 115:591–602
Blilou I, Xu J, Wildwater M, Willemsen V, Paponov I, Friml J, Heidstra R, Aida M, Palme K, Scheres B (2005) The PIN auxin efflux facilitator network controls growth and patterning in Arabidopsis roots. Nature 433:39–44
Davies J (2010) Plant hormones: biosynthesis, signal transduction, action, 3rd edn. Kluwer Academic Publishers, Dordrecht
Den Herder G, Van Isterdael G, Beeckman T, De Smet I (2010) The roots of a new green revolution. Trends Plant Sci 15:600–607
Dharmasiri N, Dharmasiri S, Estelle M (2005) The F-box protein TIR1 is an auxin receptor. Nature 435:441–445
Dolan L, Janmaat K, Willemsen V, Linstead P, Poethig S, Roberts K, Scheres B (1993) Cellular organisation of the Arabidopsis thaliana root. Development 119:71–84
Dubey S, Misra P, Sanjay S, Chatterjee S, Bag SK, Mantri S, Assif MH, Rai A, Kumar S, Tripathi P, Tripathi R, Trivedi PK, Chakrabarty D, Tuli R (2010) Transcriptomic and metabolomics shifts in rice roots in response to Cr(VI) stress. BMC Genom 11:648–667
Dubrovsky JG, Sauer M, Napsucialy-Mendivil S, Ivanchenko MG, Friml J, Shishkova S, Celenza J, Benková E (2008) Auxin acts as a local morphogenetic trigger to specify lateral root founder cells. Proc Natl Acad Sci USA 105:8790–8794
Fukaki H, Tameda S, Masuda H, Tasaka M (2002) Lateral root formation in blocked by a gain-of-function mutation line in the SOLITARY-ROOT/IAA14 gene of Arabidopsis. Plant J 29:153–168
Giehl RFH, von Wirén N (2014) Root nutrient foraging. Plant Physiol 166:509–517
Gifford ML, Dean A, Gutiérrez RA, Coruzzi GM, Birnbaum KD (2008) Cell-specific nitrogen responses mediate developmental plasticity. Proc Natl Acad Sci USA 105:803–808
Gutiérrez L, Bussell JD, Pacurar DI, Schwambach J, Pacurar M, Bellini C (2009) Phenotypic plasticity of adventitious rooting in Arabidopsis is controlled by a complex regulation of AUXIN RESPONSE FACTOR transcripts and MicroRNA abundance. Plant Cell 21:3119–3132
Gutiérrez L, Mongelard G, Floková K, Pacurar DI, Novák O, Staswick P, Kowalczyk M, Pacurar M, Demailly H, Geiss G, Bellini C (2012) Auxin controls Arabidopsis adventitious root initiation by regulating jasmonic acid homeostasis. Plant Cell 24:2515–2527
Hagen G, Guilfoyle T (2002) Auxin-responsive gene expression: genes, promoters and regulatory factors. Plant Mol Biol 49:373–385
Hayat S, Khalique G, Irfan M, Wani AS, Tripathi BN, Ahmad A (2012) Physiological changes induced by chromium stress in plants: an overview. Protoplasma 249:599–611
Holland S, Avery SV (2011) Chromate toxicity and the role of sulfur. Metallomics 3:1119–1123
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusion: β-glucuronidase as a sensitive and versatile fusion marker in higher plants. EMBO J 6:3901–3907
Krecek P, Skupa P, Libus J, Naramoto S, Tejos R, Friml J, Zazimalová E (2009) The PIN-FORMED (PIN) protein family of auxin transporters. Genome Biol 10(249–249):11
Lavenus J, Goh T, Roberts I, Guyomarch S, Lucas M, De Smet I, Fukaki H, Beeckman T, Bennet M, Laplace L (2013) Lateral root development in Arabidopsis: fifty shades of auxin. Trends Plant Sci 18:450–458
Lima JE, Kojima S, Takahashi H, von Wirén N (2010) Ammonium triggers lateral root branching in Arabidopsis in an AMMONIUM TRANSPORTER1;3-dependent manner. Plant Cell 22:3621–3633
Long TA, Tsukagohi H, Busch W, Lahner B, Salt DE, Benfey PN (2010) The bHLH transcription factor POPEYE regulates response to iron deficiency in Arabidopsis roots. Plant Cell 22:2219–2236
López-Bucio J, Hernández-Abreu E, Sánchez-Calderón L, Nieto-Jacobo MF, Simpson J, Herrera-Estrella L (2002) Phosphate availability alters architecture and causes changes in hormone sensitivity in the Arabidopsis root system. Plant Physiol 129:244–256
López-Bucio J, Cruz-Ramírez A, Herrera-Estrella L (2003) The role of nutrient availability in regulating root architecture. Curr Opin Plant Biol 6:280–287
López-Arredondo DL, Leyva-González MA, González-Morales SI, López-Bucio J, Herrera-Estrella L (2014) Phosphate nutrition: improving low-phosphate tolerance in crops. Annu Rev Plant Biol 65:95–123
López-Bucio J, Hernández-Madrigal F, Cervantes C, Ortiz-Castro R, Carreón-Abud Y, Martínez-Trujillo M (2014) Phosphate relieves chromium toxicity in Arabidopsis thaliana plants by interfering with chromate uptake. Biometals 27:363–370
Lynch J (1995) Root architecture and plant productivity. Plant Physiol 109:7–13
Malamy JE (2005) Intrinsic and environmental response pathways that regulate root system architecture. Plant Cell Environ 28:67–77
Malamy JE, Benfey PN (1997) Organization and cell differentiation in lateral roots of Arabidopsis thaliana. Development 124:33–44
Martínez-Trujillo M, Méndez-Bravo A, Ortiz-Castro R, Hernández-Madrigal F, Ibarra-Laclette E, Ruiz-Herrera LF, Long TA, Cervantes C, Herrera-Estrella L, López-Bucio J (2014) Chromate alters root system architecture and activates expression of genes involved in iron homeostasis and signaling in Arabidopsis thaliana. Plant Mol Biol 86:35–50
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Okushima Y, Fukaki H, Onoda M, Theologis A, Tasaka M (2007) ARF7 and ARF19 regulate lateral root formation via direct activation of LBD/ASL genes in Arabidopsis. Plant Cell 19:118–130
Ortiz-Castro R, Martínez-Trujillo M, López Bucio J, Cervantes C (2007) Effects of dichromate on growth and root system architecture of Arabidopsis thaliana seedlings. Plant Sci 172:684–691
Ottenschläger I, Wolff P, Wolverton C, Bhalerao RP, Sandberg G, Ishikawa H, Evans M, Palme K (2003) Gravity-regulated differential auxin transport from columella to lateral root cap cells. Proc Natl Acad Sci USA 100:2987–2991
Palmer C, Guerinot ML (2009) Facing the challenges of Cu, Fe and Zn homeostasis in plants. Nature Chem Biol 5:333–340
Pérez-Torres CA, López-Bucio J, Cruz-Ramírez A, Ibarra-Laclette E, Dharmasiri S, Estelle M, Herrera-Estrella L (2008) Phosphate availability alters lateral root development in Arabidopsis by modulating auxin sensitivity via a mechanism involving the TIR1 auxin receptor. Plant Cell 20:3258–3272
Potters G, Pasternak TP, Guisez Y, Jansen M (2009) Different stresses, similar morphogenic responses: integrating a plethora of pathways. Plant Cell Environ 32:158–169
Quint M, Gray WM (2006) Auxin signaling. Curr Opin Plant Biol 9:448–453
Rogg LE, Lasswell J, Bartel B (2001) A gain-of-function mutation in IAA28 suppresses lateral root development. Plant Cell 13:465–480
Ruíz-Herrera LF, López-Bucio J (2013) Aluminum induces low phosphate adaptive responses and modulates primary and lateral root growth by differentially affecting auxin signaling in Arabidopsis seedlings. Plant Soil 371:593–609
Ruíz-Herrera LF, Sánchez-Calderón L, Herrera-Estrella L, López-Bucio J (2012) Rare earth elements lanthanum and gadolinium induce phosphate-deficiency responses in Arabidopsis thaliana seedlings. Plant Soil 353:231–247
Sabatini S, Beis D, Wolkenfelt H, Murfett J, Guilfoyle T, Malamy J, Benfey P, Leyser O, Bechtold N, Weisbeek P, Scheres B (1999) An auxin-dependent distal organizer of pattern and polarity in the Arabidopsis root. Cell 99:463–472
Sánchez-Calderón L, López-Bucio B, Chacón-López A, Cruz-Ramírez A, Nieto-Jacobo MF, Dubrovsky J, Herrera-Estrella L (2005) Phosphate starvation induces a determinate developmental program in the roots of Arabidopsis thaliana. Plant Cell Physiol 46:174–184
Sorin C, Bussell JD, Camus I, Ljung K, Kowalczyk M, Geiss G, McKhann H, Garcion C, Vaucheret H, Sandberg G, Bellini C (2005) Auxin and light control of adventitious rooting in Arabidopsis require ARGONAUTE1. Plant Cell 17:1343–1359
Ulmasov T, Murfett J, Hagen G, Guilfoyle TJ (1997) Aux/IAA proteins repress expression of reporter genes containing natural and highly active synthetic auxin response elements. Plant Cell 9:1963–1971
Verbruggen N, Hermans C, Schat C (2009) Molecular mechanisms of metal hyperaccumulation in plants. New Phytol 181:759–776
Verstraeten I, Schotte S, Geelen D (2014) Hypocotyl adventitious root organogenesis differs from lateral root development. Front Plan Sci. doi:10.3389/fpls.2014.00495
Vieten A, Vanneste S, Wisniewska J, Benková E, Benjamins R, Beeckman T, Luschnig C, Friml J (2005) Functional redundancy of PIN proteins is accompanied by auxin-dependent cross-regulation of PIN expression. Development 132:4521–4531
Yang Y, Jung JY, Song W, Suh HS, Lee Y (2000) Identification of rice varieties with high tolerance or sensitivity to lead and characterization of the mechanism of tolerance. Plant Physiol 124:1019–1026
Acknowledgments
We thank Peter Doerner, Athanasios Theologis, Thomas J. Guilfoyle, Bonnie Bartel, Masao Tasaka and Mark Estelle for kindly providing the seeds of the transgenic and mutant lines. Juan José Valdez Alarcón is appreciated for his permission to use the confocal microscope. This work was supported by grants from the Consejo Nacional de Ciencia y Tecnología (CONACYT, México, Grants No. 60999, 177775 and 169769), the Consejo de la Investigación Científica (UMSNH, México, Grant No. CIC 2.26) and the Marcos Moshinsky Foundation.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.

Fig. S1
Effect of Cr(VI) on root system architecture. A. thaliana wild-type (Col-0) seedlings were grown on MS 0.2× agar-medium supplemented with the indicated concentration of Cr(VI). Note the inhibition of primary root growth and emergence of adventitious roots at high concentrations of Cr(VI). Photographs are representative individuals from at least 30 seedlings. Scale bar 500 µm. Supplementary material 1 (JPEG 281 kb)

Fig. S2
Effects of Cr(VI) on quiescent centre viability. Twelve-day-old A. thaliana seedlings expressing the QC46::uidA quiescent centre marker were grown on different concentrations of Cr(VI), and GUS expression was analysed in the primary root tip. Photographs are representative of individuals from at least 15 stained seedlings. The experiment was repeated twice with similar results. Scale bar 100 µm. Supplementary material 2 (JPEG 405 kb)
Rights and permissions
About this article
Cite this article
López-Bucio, J., Ortiz-Castro, R., Ruíz-Herrera, L.F. et al. Chromate induces adventitious root formation via auxin signalling and SOLITARY-ROOT/IAA14 gene function in Arabidopsis thaliana . Biometals 28, 353–365 (2015). https://doi.org/10.1007/s10534-015-9838-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-015-9838-8