Abstract
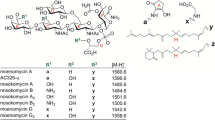
Ramoplanins produced by Actinoplanes are new structural class of lipopeptide and are currently in phase III clinical trials for the prevention of vancomycin-resistant enterococcal infections. The depsipeptide structures of ramoplanins are synthesized by non-ribosomal peptide synthetases (NRPS). Romo-orf17, a stand-alone NRPS, is responsible for the recruitment of Thr into the linear NRPS pathways for which the corresponding adenylation domain is absent. Here, systematical gene inactivation and complementation have been carried out in a Actinoplanes sp. using homologous recombination and site-specific integration methods. A hybrid gene coding for the N-terminal region of the stand-alone NRPS and the A-PCP domains of a heterologous NRPS restored production of ramoplanins. The results elucidate the unusual N-terminal region which is essential for the biosynthesis of ramoplanins.




Similar content being viewed by others
References
Butler MS (2008) Natural products to drugs: natural product-derived compounds in clinical trials. Nat Prod Rep 25:475–516
Cavalleri B, Pagani H, Volpe G, Selva E, Parenti F (1984) A-16686, a new antibiotic from Actinoplanes. I. Fermentation, isolation and preliminary physico-chemical characteristics. Antibiotics 37:309–317
Cragg GM, Grothaus PG, Newman DJ (2009) Impact of natural products on developing new anti-cancer agents. Chem Rev 109:3012–3043
Farnet CM, Zazopoulos E, Staffa A (2002) In: PCT Int Appl (Ecopia Biosciences Inc., Can.): Wo 0231155.p 212
Felnagle EA, Jackson EE, Chan YA, Podevels AM, Berti AD, McMahon MD, Thomas MG (2008) Non-ribosomal peptide synthetases involved in the production of medically relevant natural products. Mol Pharm 5:191–211
Fischbach MA, Walsh CT (2006) Assembly-line enzymology for polyketide and non-ribosomal Peptide antibiotics: logic, machinery, and mechanisms. Chem Rev 106:3468–3496
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical streptomyces genetics. John Innes Foundation, Norwich
McCafferty DG, Cudic P, Frankel BA, Barkallah S, Kruger RG, Li W (2002) Chemistry and biology of the ramoplanin family of peptide antibiotics. Biopolymers 66:261–284
Parenti F, Ciabatti R, Cavalleri B, Kettenring J (1990) Ramoplanin: a review of its discovery and its chemistry. Drugs Exp Clin Res 16:451–455
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Strieker M, Tanović A, Marahiel MA (2010) Non-ribosomal peptide synthetases: structures and dynamics. Curr Opin Struct Biol 20:234–240
Sundlov JA, Shi C, Wilson DJ, Aldrich CC, Gulick AM (2012) Structural and functional investigation of the intermolecular interaction between NRPS adenylation and carrier protein domains. Chem Biol 19:188–198
Walker S, Chen L, Hu Y, Rew Y, Shin D, Boger DL (2005) Chemistry and biology of ramoplanin: a lipoglycodepsipeptide with potent antibiotic activity. Chem Rev 105:449–475
Yin X, Zabriskie TM (2006) The enduracidin biosynthetic gene cluster from Streptomyces fungicidicus. Microbiology 152:2969–2983
Acknowledgments
This work was supported in part by grants from the National Natural Science Foundation of China (81072557, 81172962 and 30801449), the Science and Technology Commission of Shanghai Municipality (11QB1406300) and the Ministry of Science and Technology of China (Grant No. 2009ZX09301-007).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pan, HX., Li, JA., Shao, L. et al. Genetic manipulation revealing an unusual N-terminal region in a stand-alone non-ribosomal peptide synthetase involved in the biosynthesis of ramoplanins. Biotechnol Lett 35, 107–114 (2013). https://doi.org/10.1007/s10529-012-1056-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-012-1056-7