Abstract
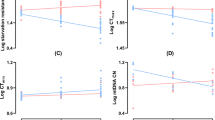
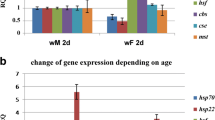
Heat-induced hormesis, the beneficial effect of mild heat-induced stress, increases the average lifespan of many organisms. Yet little is known about the mechanisms underlying this effect. We used nuclear magnetic resonance spectroscopy to investigate the long-term effects of repeated mild heat treatments on the metabolome of male Drosophila melanogaster. 10 days after the heat treatment, metabolic aging appears to be slowed down, and a treatment response with 40 % higher levels of alanine and lactate and lower levels of aspartate and glutamate were measured. All treatment effects had disappeared 16 days later. Metabolic reprogramming has been associated with the life extending effects of dietary restriction. The metabolite changes induced by the hormetic treatment suggest that the positive effects might not be limited to the repair pathways induced, but that there also is a change in energy metabolism. A possible direct link between changes in energy metabolism and heat induced increase in Hsp70 expression is discussed.



Similar content being viewed by others
References
Arbona V, Manzi M, de Ollas C, Gómez-Cadenas A (2013) Metabolomics as a tool to investigate abiotic stress tolerance in plants. IJMS 14:4885–4911. doi:10.3390/ijms14034885
Beedholm R, Clark B, Rattan S (2004) Mild heat stress stimulates 20S proteasome and its 11S activator in human fibroblasts undergoing aging in vitro. Cell Stress Chaperon. doi:10.1379/475.1
Bubliy OA, Loeschcke V (2005) Correlated responses to selection for stress resistance and longevity in a laboratory population of Drosophila melanogaster. J Evol Biol 18:789–803. doi:10.1111/j.1420-9101.2005.00928.x
Butler JA, Mishur RJ, Bhaskaran S, Rea SL (2013) A metabolic signature for long life in the Caenorhabditis elegans Mit mutants. Aging Cell 12:130–138. doi:10.1111/acel.12029
Chung J, Nguyen A-K, Henstridge DC et al (2008) HSP72 protects against obesity-induced insulin resistance. Proc Natl Acad Sci USA 105:1739–1744. doi:10.1073/pnas.0705799105
Colinet H, Larvor V, Laparie M, Renault D (2012) Exploring the plastic response to cold acclimation through metabolomics. Funct Ecol 26:711–722. doi:10.1111/j.1365-2435.2012.01985.x
Craig A, Cloarec O, Holmes E et al (2006) Scaling and normalization effects in NMR spectroscopic metabonomic data sets. Anal Chem 78:2262–2267. doi:10.1021/ac0519312
Dekker P, Meissner A, Dirks RW et al (2012) Human in vivo longevity is reflected in vitro by differential metabolism as measured by 1H-NMR profiling of cell culture supernatants. Mol BioSyst 8:783–789. doi:10.1039/C2MB05237G
Du H, Wang Z, Yu W et al (2011) Differential metabolic responses of perennial grass Cynodon transvaalensis × Cynodon dactylon (C4) and Poa Pratensis (C3) to heat stress. Physiol Plant 141:251–264. doi:10.1111/j.1399-3054.2010.01432.x
Easlon E, Tsang F, Skinner C et al (2008) The malate-aspartate NADH shuttle components are novel metabolic longevity regulators required for calorie restriction-mediated life span extension in yeast. Genes Dev 22:931–944. doi:10.1101/gad.1648308
Edwards C, Canfield J, Copes N et al (2015) Mechanisms of amino acid-mediated lifespan extension in Caenorhabditis elegans. BMC Genet 16:8–25. doi:10.1186/s12863-015-0167-2
Gems D, Partridge L (2008) Stress-response hormesis and aging: “that which does not kill us makes us stronger”. Cell Metab 7:200–203. doi:10.1016/j.cmet.2008.01.001
Gómez FH, Bertoli CI, Sambucetti P (2009) Heat-induced hormesis in longevity as correlated response to thermal-stress selection in Drosophila buzzatii. J Therm Biol. doi:10.1016/j.jtherbio.2008.09.003
Hawes TC, Hines AC, Viant MR et al (2008) Metabolomic fingerprint of cryo-stress in a freeze tolerant insect. Cryo Lett 29:505–515
Henstridge DC, Forbes JM, Penfold SA et al (2010) The relationship between heat shock protein 72 expression in skeletal muscle and insulin sensitivity is dependent on adiposity. Metab Clin Exp 59:1556–1561. doi:10.1016/j.metabol.2010.01.027
Henstridge DC, Bruce CR, Drew BG et al (2014a) Activating HSP72 in rodent skeletal muscle increases mitochondrial number and oxidative capacity and decreases insulin resistance. Diabetes 63:1881–1894. doi:10.2337/db13-0967
Henstridge DC, Whitham M, Febbraio MA (2014b) Chaperoning to the metabolic party: the emerging therapeutic role of heat-shock proteins in obesity and type 2 diabetes. Mol Metab 3:781–793. doi:10.1016/j.molmet.2014.08.003
Hercus MJ, Loeschcke V, Rattan SIS (2003) Lifespan extension of Drosophila melanogaster through hormesis by repeated mild heat stress. Biogerontology 4:149–156
Kane DA (2014) Lactate oxidation at the mitochondria: a lactate-malate-aspartate shuttle at work. Front Neurosci 8:366. doi:10.3389/fnins.2014.00366
Kaplan F, Guy CL (2004) beta-Amylase induction and the protective role of maltose during temperature shock. Plant Physiol 135:1674–1684. doi:10.1104/pp.104.040808
Keating ST, El-Osta A (2015) Epigenetics and metabolism. Circ Res 116:715–736. doi:10.1161/CIRCRESAHA.116.303936
Khazaeli A, Tatar M, Pletcher S, Curtsinger J (1997) Heat-induced longevity extension in Drosophila 1. Heat treatment, mortality, and thermotolerance. J Gerontol Ser A 52:B48–B52. doi:10.1093/gerona/52a.1.b48
Kuether K, Arking R (1999) Drosophila selected for extended longevity are more sensitive to heat shock. Age 22:175–180. doi:10.1007/s11357-999-0020-5
Le Bourg É (2010) A cold stress applied at various ages can increase resistance to heat and fungal infection in aged Drosophila melanogaster flies. Biogerontology 12:185–193. doi:10.1007/s10522-010-9309-0
Le Bourg É (2015) Life-time protection against severe heat stress by exposing young Drosophila melanogaster flies to a mild cold stress. Biogerontology 17:409–415. doi:10.1007/s10522-015-9629-1
Le Bourg É, Toffin E, Massé A (2004) Male Drosophila melanogaster flies exposed to hypergravity at young age are protected against a non-lethal heat shock at middle age but not against behavioral impairments due to this shock. Biogerontology 5:431–443. doi:10.1007/s10522-004-3200-9
Malmendal A, Overgaard J, Bundy JG et al (2006) Metabolomic profiling of heat stress: hardening and recovery of homeostasis in Drosophila. Am J Physiol Regul Integr Comp Physiol 291:R205–R212. doi:10.1152/ajpregu.00867.2005
Martin-Montalvo A, de Cabo R (2013) Mitochondrial metabolic reprogramming induced by calorie restriction. Antioxid Redox Signal 19:310–320. doi:10.1089/ars.2012.4866
Masoro EJ (2005) Overview of caloric restriction and ageing. Mech Ageing Dev 126:913–922. doi:10.1016/j.mad.2005.03.012
Massie HR, Williams TR (1979) Increased longevity of Drosophila melanogaster with lactic and gluconic acids. Exp Gerontol 14:109–115
Michaud RM, Benoit JB, Lopez-Martinez G et al (2008) Metabolomics reveals unique and shared metabolic changes in response to heat shock, freezing and desiccation in the Antarctic midge, Belgica antarctica. J Insect Physiol 54:645–655. doi:10.1016/j.jinsphys.2008.01.003
Overgaard J, Malmendal A, Sørensen JG et al (2007) Metabolomic profiling of rapid cold hardening and cold shock in Drosophila melanogaster. J Insect Physiol 53:1218–1232. doi:10.1016/j.jinsphys.2007.06.012
Rattan S (1998) Repeated mild heat shock delays ageing in cultured human skin fibroblasts. IUBMB Life. doi:10.1080/15216549800203162
Sarup P, Loeschcke V (2011) Life extension and the position of the hormetic zone depends on sex and genetic background in Drosophila melanogaster. Biogerontology 12:109–117. doi:10.1007/s10522-010-9298-z
Sarup P, Sørensen P, Loeschcke V (2011) Flies selected for longevity retain a young gene expression profile. Age 33:69–80. doi:10.1007/s11357-010-9162-8
Sarup P, Pedersen SMM, Nielsen NC et al (2012) The metabolic profile of long-lived Drosophila melanogaster. PLoS One 7:e47461. doi:10.1371/journal.pone.0047461
Sarup P, Sørensen P, Loeschcke V (2014) The long-term effects of a life-prolonging heat treatment on the Drosophila melanogaster transcriptome suggest that heat shock proteins extend lifespan. Exp Gerontol 50:34–39. doi:10.1016/j.exger.2013.11.017
Savorani F, Tomasi G, Engelsen SB (2010) icoshift: a versatile tool for the rapid alignment of 1D NMR spectra. J Magn Reson 202:190–202. doi:10.1016/j.jmr.2009.11.012
Sørensen JG, Nielsen MM, Kruhøffer M (2005) Full genome gene expression analysis of the heat stress response in Drosophila melanogaster. Cell Stress Chaperones. doi:10.1379/CSC-128R1.1
Sørensen JG, Kristensen TN, Kristensen KV, Loeschcke V (2007) Sex specific effects of heat induced hormesis in Hsf-deficient Drosophila melanogaster. Exp Gerontol 42:1123–1129. doi:10.1016/j.exger.2007.09.001
Sørensen JG, Holmstrup M, Sarup P, Loeschcke V (2009) Evolutionary theory and studies of model organisms predict a cautiously positive perspective on the therapeutic use of hormesis for healthy aging in humans. Dose Response 8:53–57. doi:10.2203/dose-response.09-040.Sorensen
Tower J (2011) Heat shock proteins and Drosophila aging. Exp Gerontol 46:355–362. doi:10.1016/j.exger.2010.09.002
Trygg J, Wold S (2002) Orthogonal projections to latent structures (O-PLS). J Chemom 16:119–128. doi:10.1002/cem.695
Vaiserman AM (2011) Hormesis and epigenetics: is there a link? Ageing Res Rev 10:413–421. doi:10.1016/j.arr.2011.01.004
Verbeke P, Clark BF, Rattan SI (2000) Modulating cellular aging in vitro: hormetic effects of repeated mild heat stress on protein oxidation and glycation. Exp Gerontol 35:787–794
Verdin E (2015) NAD+ in aging, metabolism, and neurodegeneration. Science 350:1208–1213. doi:10.1126/science.aac4854
Vermeulen CJ, Loeschcke V (2007) Longevity and the stress response in Drosophila. Exp Gerontol. doi:10.1016/j.exger.2006.09.014
Wit J, Sarup P, Lupsa N et al (2013) Longevity for free? Increased reproduction with limited trade-offs in Drosophila melanogaster selected for increased life span. Exp Gerontol 48:349–357. doi:10.1016/j.exger.2013.01.008
Wold S (1978) Cross-validatory estimation of number of components in factor and principal components models. Technometrics 20:397–405. doi:10.2307/1267639
Wu D, Cypser JR, Yashin AI, Johnson TE (2009) Multiple mild heat-shocks decrease the gompertz component of mortality in Caenorhabditis elegans. Exp Gerontol 44:607–612. doi:10.1016/j.exger.2009.06.007
Wu Z, Song L, Liu SQ, Huang D (2013) Independent and additive effects of glutamic acid and methionine on yeast longevity. PLoS One 8:e79319. doi:10.1371/journal.pone.0079319
Ye Y, Zhang L, Hao F et al (2012) Global metabolomic responses of Escherichia coli to heat stress. J Proteome Res 11:2559–2566. doi:10.1021/pr3000128
Yuneva AO, Kramarenko GG, Vetreshchak TV et al (2002) Effect of carnosine on Drosophila Melanogaster lifespan. Bull Exp Biol Med 133:559–561. doi:10.1023/A:1020273506970
Acknowledgments
The authors are grateful to Doth Andersen and Marie Rosenstand Hansen for technical assistance, to Ole Hartvig Mortensen, Niels Grunnet and Robert Brinzer for helpful discussions, to three anonymous reviewers for constructive comments and to the Danish Natural Sciences Research Council (frame and center grants to VL), Villum Kann Rasmussen Foundation (VL), Danish Natural Sciences Research Council, Lundbeck Foundation and Carlsbergfondet (stipend to PS).
Authors’ contribution
PS: planned the experiment, cultured and sampled the flies, contributed to statistical analysis, discussed the results and wrote the paper. SMMP: performed NMR experiments, initial statistical analysis and discussion of results, contributed to the paper. NCN: funding of instrument, planed the experiment, contributed to the paper. VL: funding of fly work, initiation and maintenance of selection lines, planned the experiment, discussed the results and contributed to the manuscript. AM: planed the experiment performed NMR experiments, statistical analysis, discussed the results and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarup, P., Petersen, S.M.M., Nielsen, N.C. et al. Mild heat treatments induce long-term changes in metabolites associated with energy metabolism in Drosophila melanogaster . Biogerontology 17, 873–882 (2016). https://doi.org/10.1007/s10522-016-9657-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-016-9657-5