Abstract
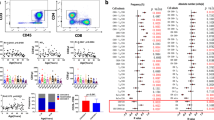
Advancing age is characterized by functional and phenotypic alterations in the distribution of circulating T-cell subsets, some of which are exacerbated by a latent infection with the persistent herpesvirus, cytomegalovirus (CMV). The influence of age, sex and CMV-infection on T-cell subpopulations in the peripheral blood remains incompletely understood. Here, T cells from 157 participants of the Berlin Aging Study II (BASE-II) were characterized at 21–34 (n = 59) and 62–85 (n = 98) years of age. We found that the frequency of naïve CD8+ T cells was significantly lower in the older group than in the young, and was different in men and women. Elderly men had a significantly lower proportion of naïve CD8+ T cells than younger men, regardless of their CMV-status, but in older women, this was seen only in the CMV-seropositive group. Reciprocally, older men had a higher proportion of late-differentiated, potentially “senescent” CD57+ T cells. Thus, T-cell senescence may be more pronounced in older men than women. Within the CD4+ population, in the elderly of both sexes there was a significantly higher proportion of late-differentiated TEMRA cells (T effector memory cells re-expressing CD45RA), but these were present exclusively in CMV-positive subjects. Finally, for the first time, we examined the so-called TSCM cell (T-stem cell-like memory) subpopulations in both CD4+ and CD8+ subsets and found that neither CMV-seropositivity nor age or sex affected their frequencies. This study confirms significant cross-sectional age-associated differences of T-cell subset distribution in a representative German urban population and emphasizes the impact of both sex and CMV-infection on T-cell naïve and memory phenotypes, but unaffected frequencies of T-stem cell-like memory cells.



Similar content being viewed by others
References
Akbar AN, Fletcher JM (2005) Memory T cell homeostasis and senescence during aging. Curr Opin Immunol 17:480–485. doi:10.1016/j.coi.2005.07.019
Almanzar G et al (2005) Long-term cytomegalovirus infection leads to significant changes in the composition of the CD8+ T-cell repertoire, which may be the basis for an imbalance in the cytokine production profile in elderly persons. J Virol 79:3675–3683. doi:10.1128/JVI.79.6.3675-3683.2005
Ansar Ahmed S, Penhale WJ, Talal N (1985) Sex hormones, immune responses, and autoimmune diseases. Mechanisms of sex hormone action. Am J Pathol 121:531–551
Barber DL et al (2006) Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439:682–687. doi:10.1038/nature04444
Bertram L et al (2014) Cohort profile: the Berlin Aging Study II (BASE-II). Int J Epidemiol 43:703–712. doi:10.1093/ije/dyt018
Bouman A, Schipper M, Heineman MJ, Faas MM (2004) Gender difference in the non-specific and specific immune response in humans. Am J Reprod Immunol 52:19–26. doi:10.1111/j.1600-0897.2004.00177.x
Caruso C, Accardi G, Virruso C, Candore G (2013) Sex, gender and immunosenescence: a key to understand the different lifespan between men and women? Immun Ageing 10:20. doi:10.1186/1742-4933-10-20
Chidrawar S, Khan N, Wei W, McLarnon A, Smith N, Nayak L, Moss P (2009) Cytomegalovirus-seropositivity has a profound influence on the magnitude of major lymphoid subsets within healthy individuals. Clin Exp Immunol 155:423–432. doi:10.1111/j.1365-2249.2008.03785.x
Derhovanessian E, Pawelec G (2012) Vaccination in the elderly. Microb Biotechnol 5:226–232. doi:10.1111/j.1751-7915.2011.00283.x
Derhovanessian E et al (2010) Hallmark features of immunosenescence are absent in familial longevity. J Immunol 185:4618–4624. doi:10.4049/jimmunol.1001629
Derhovanessian E et al (2012) Lower proportion of naive peripheral CD8+ T cells and an unopposed pro-inflammatory response to human Cytomegalovirus proteins in vitro are associated with longer survival in very elderly people. Age (Dordr) 35:1387–1399. doi:10.1007/s11357-012-9425-7
Fülöp T, Larbi A, Pawelec G (2013) Human T cell aging and the impact of persistent viral infections. Front immunol 4:271. doi:10.3389/fimmu.2013.00271
Gameiro C, Romao F (2010) Changes in the immune system during menopause and aging. Front Biosci (Elite Ed) 2:1299–1303
Garcia Verdecia B et al (2013) Immunosenescence and gender: a study in healthy Cubans. Immun Ageing 10:16. doi:10.1186/1742-4933-10-16
Gattinoni L et al (2011) A human memory T cell subset with stem cell-like properties. Nat Med 17:1290–1297. doi:10.1038/nm.2446
Hirokawa K, Utsuyama M, Hayashi Y, Kitagawa M, Makinodan T, Fulop T (2013) Slower immune system aging in women versus men in the Japanese population. Immun Ageing 10:19. doi:10.1186/1742-4933-10-19
Koch S, Larbi A, Derhovanessian E, Ozcelik D, Naumova E, Pawelec G (2008) Multiparameter flow cytometric analysis of CD4 and CD8 T cell subsets in young and old people. Immun Ageing 5:6. doi:10.1186/1742-4933-5-6
Lachmann R, Bajwa M, Vita S, Smith H, Cheek E, Akbar A, Kern F (2012) Polyfunctional T cells accumulate in large human cytomegalovirus-specific T cell responses. J Virol 86(2)1001–1009. doi:10.1128/JVI.00873-11
Looney RJ et al (1999) Role of cytomegalovirus in the T cell changes seen in elderly individuals. Clin Immunol 90:213–219. doi:10.1006/clim.1998.4638
Lugli E et al (2013) Superior T memory stem cell persistence supports long-lived T cell memory. J Clin Invest 123:594–599. doi:10.1172/JCI66327
Lynch HE, Goldberg GL, Chidgey A, Van den Brink MR, Boyd R, Sempowski GD (2009) Thymic involution and immune reconstitution. Trends Immunol 30:366–373. doi:10.1016/j.it.2009.04.003
McCombe PA, Greer JM, Mackay IR (2009) Sexual dimorphism in autoimmune disease. Curr Mol Med 9:1058–1079
Müller L, Fülöp T, Pawelec G (2013) Immunosenescence in vertebrates and invertebrates. Immun Ageing 10:12. doi:10.1186/1742-4933-10-12
Nunn CL, Lindenfors P, Pursall ER, Rolff J (2009) On sexual dimorphism in immune function. Philos Trans R Soc London B 364:61–69. doi:10.1098/rstb.2008.0148
Nussinovitch U, Shoenfeld Y (2012) The role of gender and organ specific autoimmunity. Autoimmun Rev 11:A377–A385. doi:10.1016/j.autrev.2011.11.001
Ongradi J, Kovesdi V (2010) Factors that may impact on immunosenescence: an appraisal. Immun Ageing 7:7. doi:10.1186/1742-4933-7-7
Pawelec G (2012) Hallmarks of human “immunosenescence”: adaptation or dysregulation? Immun Ageing 9:15. doi:10.1186/1742-4933-9-15
Pawelec G (2014a) Immunosenenescence: role of cytomegalovirus. Exp Gerontol 54:1–5. doi:10.1016/j.exger.2013.11.010
Pawelec G (2014b) T-cell immunity in the aging human. Haematologica 99:795–797. doi:10.3324/haematol.2013.094383
Pawelec G, Derhovanessian E, Larbi A, Strindhall J, Wikby A (2009) Cytomegalovirus and human immunosenescence. Rev Med Virol 19:47–56. doi:10.1002/rmv.598
Pera A, Campos C, Corona A, Sanchez-Correa B, Tarazona R, Larbi A, Solana R (2014) CMV latent infection improves CD8+ T response to SEB due to expansion of polyfunctional CD57+ cells in young individuals. PLoS One 9:e88538. doi:10.1371/journal.pone.0088538
Qi Q et al (2014a) Diversity and clonal selection in the human T-cell repertoire. Proc Natl Acad Sci USA 111:13139–13144. doi:10.1073/pnas.1409155111
Qi Q, Zhang DW, Weyand CM, Goronzy JJ (2014b) Mechanisms shaping the naive T cell repertoire in the elderly—thymic involution or peripheral homeostatic proliferation? Exp Gerontol 54:71–74. doi:10.1016/j.exger.2014.01.005
Romero P et al (2007) Four functionally distinct populations of human effector-memory CD8+ T lymphocytes. J Immunol 178:4112–4119
Sakiani S, Olsen NJ, Kovacs WJ (2013) Gonadal steroids and humoral immunity. Nat Rev Endocrinol 9:56–62. doi:10.1038/nrendo.2012.206
Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A (1999) Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401:708–712. doi:10.1038/44385
Smithey MJ, Li G, Venturi V, Davenport MP, Nikolich-Zugich J (2012) Lifelong persistent viral infection alters the naive T cell pool, impairing CD8 T cell immunity in late life. J Immunol 189:5356–5366. doi:10.4049/jimmunol.1201867
Stowe RP, Kozlova EV, Yetman DL, Walling DM, Goodwin JS, Glaser R (2007) Chronic herpesvirus reactivation occurs in aging. Exp Gerontol 42:563–570. doi:10.1016/j.exger.2007.01.005
Strioga M, Pasukoniene V, Characiejus D (2011) CD8+ CD28− and CD8+ CD57+ T cells and their role in health and disease. Immunology 134:17–32. doi:10.1111/j.1365-2567.2011.03470.x
Tarazona R, DelaRosa O, Alonso C, Ostos B, Espejo J, Pena J, Solana R (2000) Increased expression of NK cell markers on T lymphocytes in aging and chronic activation of the immune system reflects the accumulation of effector/senescent T cells. Mech Ageing Dev 121:77–88
Tower J, Arbeitman M (2009) The genetics of gender and life span. J Biol 8:38. doi:10.1186/jbiol141
Warren LA, Rossi DJ (2009) Stem cells and aging in the hematopoietic system. Mech Ageing Dev 130:46–53. doi:10.1016/j.mad.2008.03.010
Wertheimer AM et al (2014) Aging and cytomegalovirus infection differentially and jointly affect distinct circulating T cell subsets in humans. J Immunol 192:2143–2155. doi:10.4049/jimmunol.1301721
Wikby A et al (2006) The immune risk phenotype is associated with IL-6 in the terminal decline stage: findings from the Swedish NONA immune longitudinal study of very late life functioning. Mech Ageing Dev 127:695–704. doi:10.1016/j.mad.2006.04.003
Yan J, Greer JM, Hull R, O’Sullivan JD, Henderson RD, Read SJ, McCombe PA (2010) The effect of ageing on human lymphocyte subsets: comparison of males and females. Immun Ageing 7:4. doi:10.1186/1742-4933-7-4
Acknowledgments
The authors thank participants and colleagues of the interdisciplinary group of the BASE-II study. We would like to thank Karin Hähnel and Lilly Öttinger for their excellent organizational and technical assistance and Nicole Janssen for performing CMV-ELISAs. We thank all colleagues of the TATI-Group and especially Kilian Wistuba-Hamprecht, whose skills and commitment made this study possible. We gratefully acknowledge the support from the German Ministry for Education and Research Grant Nos. 16SV5536K and FKZ 01EI1401 and from the European Commission Grant FP7 259679, as well as from the Max Planck Institute for Human Development, Berlin.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10522_2015_9563_MOESM1_ESM.ppt
Supplementary Figure S1: Gating strategy to identify distinct subsets of CD4+ (a) and CD8+ (b) T cells. Viable lymphocytes were gated within the CD3+ gate and selected for CD8+ or CD4+ T cell subsets, which were subdivided into four (N, CM, EM and TEMRA-E) using CD45RA and CCR7. CD27 and CD28 staining was used to characterize their differentiation status: N (CD45RA+CCR7+CD27+CD28+); CM (CD45RA-CCR7+CD27+CD28+); EM3 (CD45RA−CCR7−CD27−CD28−); E (CD45RA+CCR7−CD27−CD28−). N cells have been gated for CD95 to identify TSCM (CD45RA+CCR7+CD27+ CD28+CD95+) cells. Supplementary material 1 (PPT 345 kb)
Rights and permissions
About this article
Cite this article
Di Benedetto, S., Derhovanessian, E., Steinhagen-Thiessen, E. et al. Impact of age, sex and CMV-infection on peripheral T cell phenotypes: results from the Berlin BASE-II Study. Biogerontology 16, 631–643 (2015). https://doi.org/10.1007/s10522-015-9563-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-015-9563-2