Abstract
Improving the accuracy of phenotyping through the use of advanced psychometric tools will increase the power to find significant associations with genetic variants and expand the range of possible hypotheses that can be tested on a genome-wide scale. Multivariate methods, such as structural equation modeling (SEM), are valuable in the phenotypic analysis of psychiatric and substance use phenotypes, but these methods have not been integrated into standard genome-wide association analyses because fitting a SEM at each single nucleotide polymorphism (SNP) along the genome was hitherto considered to be too computationally demanding. By developing a method that can efficiently fit SEMs, it is possible to expand the set of models that can be tested. This is particularly necessary in psychiatric and behavioral genetics, where the statistical methods are often handicapped by phenotypes with large components of stochastic variance. Due to the enormous amount of data that genome-wide scans produce, the statistical methods used to analyze the data are relatively elementary and do not directly correspond with the rich theoretical development, and lack the potential to test more complex hypotheses about the measurement of, and interaction between, comorbid traits. In this paper, we present a method to test the association of a SNP with multiple phenotypes or a latent construct on a genome-wide basis using a diagonally weighted least squares (DWLS) estimator for four common SEMs: a one-factor model, a one-factor residuals model, a two-factor model, and a latent growth model. We demonstrate that the DWLS parameters and p-values strongly correspond with the more traditional full information maximum likelihood parameters and p-values. We also present the timing of simulations and power analyses and a comparison with and existing multivariate GWAS software package.



Similar content being viewed by others
References
Abecasis GR, Cherny SS, Cookson WO, Cardon LR (2002) Merlinrapid analysis of dense genetic maps using sparse gene flow trees. Nat Genet 30(1):97–101
Agresti, A. (2002). Categorical data analysis [second edition]. Wiley-Interscience
Bock RD and Aitkin M (1981) Marginal maximum likelihood estimation of item parameters: application of an EM algorithm. Psychometrika 46(4):443–459
Boker S, Neale M, Maes H, Wilde M, Spiegel M, Brick T, Fox J (2011) Openmx: an open source extended structural equation modeling framework. Psychometrika 76(2):306–311
Boker SM, Neale MC, Maes HH, Wilde MJ, Spiegel M, Brick TR et al. (2015) Openmx 2.3.1 user guide. [Computer software manual]
Blangero J, Lange K, Almasy L, Williams J, Dyer T, Peterson C (2000) Sequential oligogenic linkage analysis routines (SOLAR). [Computer software manual]
Browne MW (1984) Asymptotically distribution-free methods for the analysis of covariance structures. Br J Math Stat Psychol 37:62–83
Carragher N, Teesson M, Sunderland M, Newton NC, Krueger RF, Conrod PJ, Slade T (2016) The structure of adolescent psychopathology: a symptom-level analysis. Psychol Med 46(5):981–994. doi:10.1017/S0033291715002470
Chin WW (1998) Issues and opinion on structural equation modeling. MIS Q 22(1):vii–xvi
Choh AC, Lee M, Kent JW, Diego VP, Johnson W, Curran JE, Dyer TD, Bellis C, Blangero J, Siervogel RM, Towne B, Demerath EW, Czerwinski SA (2014) Gene-by-age effects on BMI from birth to adulthood: the Fels Longitudinal Study. Obesity 22(3):875–881
CONVERGE consortium (2015) Sparse whole-genome sequencing identifies two loci for major depressive disorder. Nature 523:588–591. doi:10.1038/nature14659
Cross-Disorder Group of the Psychiatric Genomics Consortium (2013) Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet 381(9875):1371–1379. doi:10.1016/S0140-6736(12)62129-1
Dahl A, Iotchkova V, Baud A, Johansson A, Gyllensten U, Soranzo N, Marchini J (2016) A multiple-phenotype imputation method for genetic studies. Nat Genet 48:466–472. doi:10.1038/ng.3513
DiStefano C, Morgan GB (2014) A comparison of diagonal weighted least squares robust estimation techniques for ordinal data. Struct Equ Model 21(3):425–438
Doyle MM, Murphy J, Shevlin M (2016) Competing factor models of child and adolescent psychopathology. J Abnorm Child Psychol 44:1559–1571. doi:10.1007/s10802-016-0129-9
Duell EJ, Sala N, Travier N, Munoz X, Boutron-Ruault MC, Clavel-Chapelon F, Gonzalez CA (2012) Genetic variation in alcohol dehydrogenase (adh1a, adh1b, adh1c, adh7) and aldehyde dehydrogenase (aldh2), alcohol consumption and gastric cancer risk in the european prospective investigation into cancer and nutrition (epic) cohort. Carcinogenesis 33(2):361–367. doi:10.1093/carcin/bgr285
Duncan SC, Duncan TE, Strycker LA (2006) Alcohol use from ages 9 to 16: a cohort-sequential latent growth model. Drug Alcohol Depend 81(1):71–81. doi:10.1016/j.drugalcdep.2005.06.001
Duncan TE, Duncan SC, Alpert A, Hops H, Stoolmiller M, Muthen B (1997) Latent variable modeling of longitudinal and multilevel substance use data. Multivar Behav Res 32(3):275–318. doi:10.1207/s15327906mbr3203
Fardo DW, Zhang X, Ding L, He H, Kurowski B, Alexander ES, Mersha TB, Pilipenko V, Kottyan L, Nandakumar K, Martin L (2014) On family-based genome-wide association studies with large pedigrees: observations and recommendations. BMC Proc 8(Suppl 1):S26
Ferreira MAR, Purcell SM (2009) A multivariate test of association. Bioinformatics 25(1):132–133. doi:10.1093/bioinformatics/btn563
Furlotte NA, Eskin E (2015) Efficient multiple-trait association and estimation of genetic correlation using the matrix-variate linear mixed model. Genetics 200(1):59–68. doi:10.1534/genetics.114.171447
Grice JW (2001) Computing and evaluating factor scores. Psychol Methods 6(4):430–450
Hyde CL, Nagle MW, Tian C, Chen X, Paciga SA, Wendland JR, Winslow AR (2016) Identification of 15 genetic loci associated with risk of major depression in individuals of european descent. Nat Genet 48(9):1031–1036. doi:10.1038/ng.3623
Johnson DR, Creech JC (1983) Ordinal measures in multiple indicator models: a simulation study of categorization error. Am Soc Rev 48:398407
Joreskog KG, Sorbom D (1989) LISREL 7: a guide to the program and applications, 2nd edn. SPSS Inc, Chicago
Joreskog KG, Sorbom D (1993) New features in prelis 2. Scientific Software International, Chicago
Joreskog KG, Sorbom D (1996) Lisrel 8 users reference guide. Scientific Software International, Chicago
Joreskog KG, Sorbom D (1996) LISREL 8 users reference guide. Scientific Software Inc, Mooresville
Joreskog KG, Sorbom D (2001) LISREL 8: new statistical features. Scientific Software Inc, Mooresville
Kent JW, Peterson CP, Dyer TD, Almasy L, Blangero J (2009) Genome-wide discovery of maternal effect variants. BMC Proc 9(Suppl 7):S19
Kessler RC, Chiu WT, Demler O, Walters EE (2005) Prevalence, severity, and comorbidity of twelve-month DSM-IV disorders in the national comorbidity survey replication (NCS-R). Arch Gen Psychiatry 62(6):617627
Klei L, Luca D, Devlin B, Roeder K (2008) Pleiotropy and principal components of heritability combine to increase power for association analysis. Genet Epidemiol 32(1):9–19. doi:10.1002/gepi.20257
Krueger RF (1999) The structure of common mental disorders. Arch Gen Psychiatry 56(10):921–926
Lai K (2011) Abstract: sample size planning for latent curve models. Multivar Behav Res 46(6):1013. doi:10.1080/00273171.2011.636705
Laird NM (2011) Family-based association test (FBAT). Wiely, St. Hoboken
Li CH (2015) Confirmatory factor analysis with ordinal data: comparing robust maximum likelihood and diagonally weighted least squares. Behav Res Methods. doi:10.3758/s13428-015-0619-7
Lips EH, Gaborieau V, McKay JD, Chabrier A, Hung RJ, Boffetta P, Brennan P (2010) Association between a 15q25 gene variant, smoking quantity and tobacco-related cancers among 17 000 individuals. Int J Epidemiol 39(2):563–577. doi:10.1093/ije/dyp288
Little RJ, Rubin DB (1989) The analysis of social science data with missing values. Sociol Methods Res 18:292–326
Liu JZ, Tozzi F, Waterworth DM, Pillai SG, Muglia P, Middleton L, Marchini J (2010) Meta-analysis and imputation refines the association of 15q25 with smoking quantity. Nat Genet 42(5):436–440. doi:10.1038/ng.572
MacCallum RC, Hong S (1997) Power analysis in covariance structure modeling using GFI and AGFI. Multivar Behav Res 32(2):193–210. doi:10.1207/s15327906mbr3202
Marchini J, Howie B, Myers S, McVean G, Donnelly P (2007) A new multipoint method for genome-wide association studies by imputation of genotypes. Nat Genet 39(7):906–913. doi:10.1038/ng2088
McArdle JJ, Boker SM (1990) Rampath path diagram software. Data Transforms Inc, Denver
McArdle JJ, McDonald RP (1984) Some algebraic properties of the reticular action model for moment structures. Br J Math Stat Psychol 37:234–251
Medland SE, Neale MC (2010) An integrated phenomic approach to multivariate allelic association. Eur J Hum Genet 18(2):233–239. doi:10.1038/ejhg.2009.133
Medland SE, Nyholt DR, Painter JN, McEvoy BP, McRae AF, Zhu G, Martin NG (2009) Common variants in the trichohyalin gene are associated with straight hair in Europeans. Am J Hum Genet 85(5):750–755. doi:10.1016/j.ajhg.2009.10.009
Mehta PD, Neale MC, Flay BR (2004) Squeezing interval change from ordinal panel data: latent growth curves with ordinal outcomes. Psychol Methods 9(3):301–333
Meyer K, Tier B (2012) SNP snappy: a strategy for fast genome-wide association studies fitting a full mixed model. Genetics 190(1):275–277. doi:10.1534/genetics.111.134841
Miles J (2003) A framework for power analysis using a structural equation modelling procedure. BMC Med Res Methodol 3:27. doi:10.1186/1471-2288-3-27
Mindrila D (2010) Maximum likelihood (ml) and diagonally weighted least squares (DWLS) estimation procedures: a comparison of estimation bias with ordinal and multivariate non-normal data. Int J Digital Soc 1(1):60–66
Muhleisen TW, Leber M, Schulze TG, Strohmaier J, Degenhardt F, Treutlein J et al (2014) Genome-wide association study reveals two new risk loci for bipolar disorder. Nat Commun 5:3339. doi:10.1038/ncomms4339
Muthen B (1984) A general structural equation model with dichotomous, ordered categorical, and continuous latent variable indicators. Psychometrika 49:115–132
Nakamura K, Suwaki H, Matsuo Y, Ichikawa Y, Miyatake R, Iwahashi K (1995) Association between alcoholics and the genotypes of ALDH2, ADH2, ADH3 as well as P-4502E1. Arukoru Kenkyuto Yakubutsu Ison 30:33–42
Neale MC (1994) Mx: statistical modeling, 2nd edn. Medical College of Virginia, Richmond
Neale MC, Hunter MD, Pritikin JN, Zahery M, Brick TR, Kickpatrick RM et al. (in press) OpenMx 2.0: extended structural equation and statistical modeling. Psychometrika.
Neale MC, McArdle JJ (2000) Structured latent growth curves for twin data. Twin Res 3(3):165–177
Okbay A, Baselmans BML, De Neve J-E, Turley P, Nivard MG, Fontana MA, Cesarini D (2016) Genetic variants associated with subjective well-being, depressive symptoms, and neuroticism identified through genome-wide analyses. Nat Genet 48(6):624–633. doi:10.1038/ng.3552
OReilly PF, Hoggart CJ, Pomyen Y, Calboli FCF, Elliott P, Jarvelin M-R, Coin LJM (2012) Multiphen: joint model of multiple phenotypes can increase discovery in GWAS. PLoS ONE 7(5):e34861. doi:10.1371/journal.pone.0034861
Paltoo DN, Rodriguez LL, Feolo M, Gillanders E, Ramos EM, Rutter JL et al (2014) National Institutes of Health Genomic Data Sharing Governance Committees (2014, Sep). Data use under the nih gwas data sharing policy and future directions. Nat Genet 46(9):934–938. doi:10.1038/ng.3062
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MAR, Bender D, Sham PC (2007) Plink: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81(3):559–575. doi:10.1086/519795
R Development Core Team (2008) R: a language and environment for statistical computing[Computer software manual]. Vienna, Austria. http://www.R-project.org (ISBN 3-900051-07-0)
Saccone NL, Saccone SF, Hinrichs AL, Stitzel JA, Duan W, Pergadia ML, Bierut LJ (2009) Multiple distinct risk loci for nicotine dependence identified by dense coverage of the complete family of nicotinic receptor subunit (CHRN) genes. Am J Med Genet B Neuropsychiatr Genet 150B(4):453–466. doi:10.1002/ajmg.b.30828
Schizophrenia Psychiatric Genome-Wide Association Study (GWAS) Consortium (2011) Genome-wide association study identifies five new schizophrenia loci. Nat Genet 43(10):969–976. doi:10.1038/ng.940
Servin B, Stephens M (2007) Imputation-based analysis of association studies: candidate regions and quantitative traits. PLoS Genet 3(7):e114. doi:10.1371/journal.pgen.0030114
Smith DJ, Escott-Price V, Davies G, Bailey MES, Colodro-Conde L, Ward J et al (2016) Genome-wide analysis of over 106 000 individuals identifies 9 neuroticism-associated loci. Mol Psychiatry 21(11):1644. doi:10.1038/mp.2016.177
Stephens M (2013) A unified framework for association analysis with multiple related phenotypes. PLoS ONE 8(7):e65245. doi:10.1371/journal.pone.0065245
van der Sluis S, Posthuma D, Dolan CV (2013) Tates: efficient multivariate genotype-phenotype analysis for genome-wide association studies. PLoS Genet 9(1):e1003235. doi:10.1371/journal.pgen.1003235
Venables WN, Ripley BD (2002) Modern applied statistics with s, 4th edn. Springer, New York (ISBN 0-387-95457-0)
Visscher PM, Brown MA, McCarthy MI, Yang J (2012) Five years of gwas discovery. Am J Hum Genet 90(1):7–24. doi:10.1016/j.ajhg.2011.11.029
Whitfield JB, Nightingale BN, Bucholz KK, Madden PAF, Heath AC, Martin NG (1998) ADH genotypes and alcohol use and dependence in europeans. Alcoholism 22:1463–1469
Wolf EJ, Harrington KM, Clark SL, Miller MW (2013) Sample size requirements for structural equation models: an evaluation of power, bias, and solution propriety. Educ Psychol Meas 76(6):913–934. doi:10.1177/0013164413495237
Zhou X, Stephens M (2012) Genome-wide efficient mixed-model analysis for association studies. Nat Genet 44(7):821–824. doi:10.1038/ng.2310
Zhou X, Stephens M (2014) Efficient multivariate linear mixed model algorithms for genome-wide association studies. Nat Methods 11(4):407–409. doi:10.1038/nmeth.2848
Acknowledgements
An earlier draft of this paper was presented at the 44th meeting of the Behavioral Genetics Association in Charlottesville, Virginia, June 18 to June 21, 2014. We would like to thank the conference attendees for their suggestions to improve the paper.
Funding
This study was supported by NIDA Grants R01DA-025109, R01DA-024413, R01DA-018673 and R25DA-26119.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Brad Verhulst, Hermine H. Maes, and Michael C. Neale declare that they have no conflict of interest.
Statement of Human and Animal Rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
For this type of study formal consent is not required.
Additional information
Edited by Sarah Medland.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix 1: syntax and application
Appendix 1: syntax and application
A supplementary goal of GW-SEM is to create a user-friendly set of commands that researchers who may not be dedicated data analysts can use effectively. Therefore to demystify the process, in this section we explain the use of each of the principal functions.
The first step in the analysis is to calculate the SNP-invariant covariances. These calculations are conducted using the facCov() function:
facCov( dataset, VarNames, covariates)
where dataset is a dataframe in R, VarNames is a list of the variable names of the items, and covariates is a list of covariates. The function returns covariances, weights, and standard errors of all of the variances, covariances, means and thresholds for all of the items and covariates. Because this function runs quickly (even for a relatively large number of items), and is necessary for all subsequent functions, it is called directly by the other functions. Users can use this function to ensure that their data is properly organized, and to ensure that there are no peculiarities with any of the variables they plan on including in their analyses.
The second step in the analysis is to estimate the SNP-item and SNP-covariate covariances. These calculations are conducted using the snpCovs() function:
snpCovs(FacModelData, vars, covariates, SNPdata, output, zeroOne, runs, inc, start)
where FacModelData is the path to the text file with the item and covariate data, vars is a list of items, covariates is a list of covariates, SNPdata is the path to the text file with the SNP values, output is the prefix for the output files, zeroOne is a logical value indicating whether the first and second thresholds should be fixed at 0 and 1, freeing up parameters to estimate the mean and the variance following the liability-threshold Model (Mehta et al. 2004), runs is the number of batches of SNPs to be analyzed, inc is the number of SNPs included in each batch, and start, is the column in the SNP file of the first SNP to be sampled. The output from this function is saved in three separate files as specified by the output argument: the covariances, the weights and the standard errors.
The final step of the model fits the SEM using the gwasDWLS function:
gwasDWLS(itemData, snpCov, snpWei, VarNames, covariates, runs, output, inc)
where itemData is the path to the text file with the item and covariates, snpCov is the path to the text file with covariances between the SNPs and the item and covariates (calculated in the previous step), snpWei is the path to the text file with the weights, VarNames is a list of items, covariates is a list of covariates, runs is the number of batches of SNPs to be analyzed, output is the file name for the output file, and inc is the number of SNPs included in each batch. Due to identification restrictions, users must supply at least three items (indicators) for the latent factor. There is no minimum or maximum for the number of covariates that can be included in the analysis. Note that with these two lines of R code, it is possible to conduct the one-factor GWAS.
The next SEM is the residuals model. The syntax to fit the residuals model is:
snpCovs(FacModelData, vars, covariates, SNPdata, output, zeroOne, runs, inc, start)
resDWLS(itemData, snpCov, snpWei, VarNames, covariates, resids, factor, runs, output, inc)
As can be seen above, for the residuals model, the only two arguments that differ from the one-factor model are resids which is a list of the items to be regressed on the SNPs, and factor which is a logical value asking whether the latent factor is to be regressed on the SNPs. The other arguments operate in exactly the same way as with gwasDWLS. Further, the snpCovs function is equivalent for both the gwasDWLS and the resDWLS, making it possible to easily conduct additional analyses with minimal additional steps. Again, at least three items are required in order to provide an identified factor model.
The third model in the package is the two-factor SEM. The syntax to run the two-factor GWAS is:
snpCovs(FacModelData, vars, covariates, SNPdata, output, runs, inc, start)
twofacDWLS(itemData, snpCov, snpWei, f1Names, f2Names, covariates, runs, output, inc)
Again the snpCovs argument is identical to the previous GWAS models, and the only change in arguments from the gwasDWLS to the twofacDWLS is the addition of f1Names and f2Names, which are lists of the variable names that load on Factor 1 and Factor 2, respectively. These lists are not exclusive for generality but at least three items must be specified for each factor, with at least one item for each factor excluded from the alternative factor.
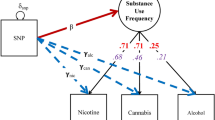
The last model included in the software is the LGM, depicted in Fig 1d. The syntax for the LGM GWAS is:
snpCovs(FacModelData, vars, covariates, SNPdata, output, zeroOne, runs, inc, start)
growDWLS(itemData, snpCov, snpWei, VarNames, covariates, quadratic, orthogonal, runs, output, inc)
The snpCovs function is again equivalent to the function described above, except that for categorical data, the zeroOne argument should be specified as TRUE, to facilitate the estimation of the LGM. As the LGM is particularly focused on mean and variance changes, this is an important feature of the covariance model. The only change in the growDWLS() function from the gwasDWLS() function is the inclusion of the quadratic and orthogonal arguments. The quadratic argument is a logical value asking whether to include a latent quadratic growth parameter. The orthogonal argument is a logical value asking whether to use the standard growth loadings or orthogonal contrasts. The other arguments are exactly the same as the gwasDWLS function.
Rights and permissions
About this article
Cite this article
Verhulst, B., Maes, H.H. & Neale, M.C. GW-SEM: A Statistical Package to Conduct Genome-Wide Structural Equation Modeling. Behav Genet 47, 345–359 (2017). https://doi.org/10.1007/s10519-017-9842-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10519-017-9842-6