Abstract
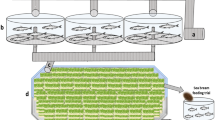
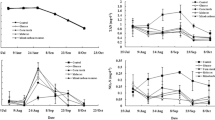
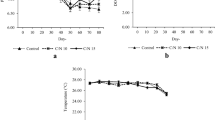
The objective of this study was to determine the effects of prawn juveniles and organic carbon source on the water quality and production performance of catfish. The experiment consisted of three treatments in triplicates, i.e. catfish monoculture as the control (treatment A); catfish–prawn co-culture at a prawn density of 20 prawn/m2 (treatment B) and catfish–prawn co-culture at a prawn density of 40 prawn/m2 (treatment C). Catfish was reared in a bamboo cage inside experimental concrete ponds at an initial density of 100/m2. Commercial feed pellets were only offered to the fish. In treatments B and C, organic carbon was added at an estimated C/N ratio of 10 to stimulate biofloc growth as the food source for the prawn juveniles. There was no significant difference in catfish growth among treatments. Dissolved inorganic nitrogen concentrations in B and C were generally lower than in the control. Although significantly affected by the initial stocking density, prawn growth was comparable to that fed with formulated feed. Catfish co-culture with prawn with C organic addition resulted in significantly higher feed nitrogen utilization. Overall results suggest that catfish–prawn co-culture with C organic addition may improve total food conversion ratio, reduce nitrogen waste and generate additional income for catfish farmers.




Similar content being viewed by others
References
APHA (2005) Standard methods for the examination of the water and wastewater, American Public Health Association, Washington, DC
Avnimelech Y (1999) Carbon/nitrogen ratio as a control element in aquaculture systems. Aquaculture 176:227–235
Azim ME, Little DC (2008) The biofloc technology (BFT) in indoor tanks: water quality, biofloc composition, and growth and welfare of Nile tilapia (Oreochromis niloticus). Aquaculture 283:29–35
Bauer W, Prentice-Hernandez C, Tesser MB et al (2012) Substitution of fishmeal with microbial floc meal and soy protein concentrate in diets for the pacific white shrimp Litopenaeus vannamei. Aquaculture 342–343:112–116
Crab R, Avnimelech Y, Defoirdt T et al (2007) Nitrogen removal techniques in aquaculture for a sustainable production. Aquaculture 270:1–14
Crab R, Chielens B, Wille M et al (2010) The effect of different carbon sources on the nutritional value of bioflocs a feed for Macrobrachium rosenbergii postlarvae. Aquac Res 41:559–567
De Schryver P, Verstraete W (2009) Nitrogen removal from aquaculture pond water by heterotrophic nitrogen assimilation in lab-scale sequencing batch reactors. Bioresour Technol 100:1162–1167
De Schryver P, Crab R, Defoirdt T et al (2008) The basics of bio-flocs technology: the added value for aquaculture. Aquaculture 277:125–137
Diana JS, Egna HS, Chopin T et al (2013) Responsible aquaculture in 2050: valuing local conditions and human innovations will be key to success. Bioscience 63:255–262
Ekasari J, Crab R, Verstraete W (2010) Primary nutritional content of bio-flocs cultured with different organic carbon sources and salinity. Hayati J Biosci 17:125–130
Hari B, Kurup BM, Varghese JT et al (2004) Effect of carbohydrate addition on production in extensive shrimp culture systems. Aquaculture 241:179–194
Huisman EA (1987) The principles of fish culture production. Department of Aquaculture, Wageningen University, Netherland, p 100
Ju ZY, Forster I, Conquest L, Dominy W (2008) Enhanced growth effects on shrimp (Litopenaeus vannamei) from inclusion of whole shrimp floc or floc fractions to a formulated diet. Aquac Nutr 14:533–543
Kuhn DD, Lawrence AL, Boardman GD et al (2010) Evaluation of two types of bioflocs derived from biological treatment of fish effluent as feed ingredients for Pacific white shrimp, Litopenaeus vannamei. Aquaculture 303:28–33
New MB (2002) Farming freshwater prawns: a manual for culture of the gaint river prawn (Macrobrachium rosenbergii). Roma: Food and Agriculture Organization of the United Nations
Peteri A, Nandi S, Chowdhury SN (1992) Manual on seed production of African catfish (Clarias gariepinus). Rome: Food and Agriculture Organization of the United Nations
Takeuchi T (1988) Laboratory work-chemical evaluation of dietary nutrients. In: Watanabe T (ed) Fish nutrition and mariculture. Kanagawa International Fisheries Training Center, Japan International Cooperation Agency, Kanagawa, pp 179–233
Verdegem MCJ (2013) Nutrient discharge from aquaculture operations in function of system design and production environment. Rev Aquac 4:1–14
Wedemeyer GA (1996) Physiology of fish in intensive culture systems. Chapman and Hall, New York
Xu WJ, Pan LQ (2012) Effects of bioflocs on growth performance, digestive enzyme activity and body composition of juvenile Litopenaeus vannamei in zero-water exchange tanks manipulating C/N ratio in feed. Aquaculture 356–357:147–152
Xu WJ, Pan LQ, Zhao DH et al (2012) Preliminary investigation into the contribution of bioflocs on protein nutrition of Litopenaeus vannamei fed with different dietary protein levels in zero-water exchange culture tanks. Aquaculture 350–353:147–153
Yi Y, Lin CK, Diana S (2003) Hybrid catfish (Clarias macrocephalus x C. gariepinus) and Nile tilapia (Oreochromis niloticus) in an integrated pen-cum pond system: growth performance and nutrient budgets. Aquaculture 217:395–408
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rohmana, D., Surawidjaja, E.H., Sukenda, S. et al. Water quality and production performance of catfish–prawn co-culture with organic carbon source addition. Aquacult Int 23, 267–276 (2015). https://doi.org/10.1007/s10499-014-9814-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-014-9814-2