Abstract
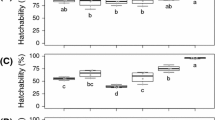
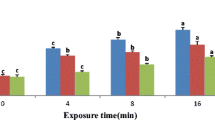
Egg hatchability of four predatory mites—Phytoseiulus persimilis Athias-Henriot, Iphiseius [Amblyseius] degenerans Berlese, Amblyseius swirskii Athias-Henriot, and Euseius finlandicus Oudemans (Acari: Phytoseiidae)—and the spider mite Tetranychus urticae Koch (Acari: Tetranychidae) was determined under various UV-B doses either in constant darkness (DD) or with simultaneous irradiation using white light. Under UV-B irradiation and DD or simultaneous irradiation with white light, the predator’s eggs hatched in significantly lower percentages than in the control non-exposed eggs, which indicates deleterious effects of UV-B on embryonic development. In addition, higher hatchability percentages were observed under UV-B irradiation and DD in eggs of the predatory mites than in eggs of T. urticae. This might be caused by a higher involvement of an antioxidant system, shield effects by pigments or a mere shorter duration of embryonic development in predatory mites than in T. urticae, thus avoiding accumulative effects of UV-B. Although no eggs of T. urticae hatched under UV-B irradiation and DD, variable hatchability percentages were observed under simultaneous irradiation with white light, which suggests the involvement of a photoreactivation system that reduces UV-B damages. Under the same doses with simultaneous irradiation with white light, eggs of T. urticae displayed higher photoreactivation and were more tolerant to UV-B than eggs of the predatory mites. Among predators variation regarding the tolerance to UV-B effects was observed, with eggs of P. persimilis and I. degenerans being more tolerant to UV-B radiation than eggs of A. swirskii and E. finlandicus.
Similar content being viewed by others
References
Abram P, Guerra-Grenier E, Després-Einspenner M, Ito S, Wakamatsu K, Boivin G, Brodeur J (2015) An insect with selective control of egg coloration. Curr Biol 25:2007–2011
Altincicek B, Kovacs JL, Gerardo NM (2012) Horizontally transferred fungal carotenoid genes in the two-spotted spider mite Tetranychus urticae. Biol Lett 8:253–257
Bryon A, Wybouw N, Dermauw W, Tirry L, Van Leeuwen T (2013) Genome wide gene-expression analysis of facultative reproductive diapause in the two-spotted spider mite Tetranychus urticae. BMC Genom 14:815
Finney DJ (1971) Probit analysis, 3rd edn. Cambridge University Press, London
Fukaya M, Uesugi R, Ohashi H, Sakai Y, Sudo M, Kasai A, Kishimoto H, Osakabe M (2013) Tolerance to solar ultraviolet-B radiation in the citrus red mite, an upper surface user of host plant leaves. PhotochemPhotobiol 89:424–431
Gerson U, Smiley RL, Ochoa R (2003) Mites (Acari) for pest control. Blackwell Science, Oxford, p 539
Ghazy NA, Osakabe M, Negm MW, Schausberger P, Gotoh T, Amano H (2016) Phytoseiid mites under environmental stress. Biol Control 96:120–134
Grbić M, Van Leeuwen T, Clark RM, Rombauts S, Rouzι P, Grbic V, Osborne EJ, Dermauw W, Ngoc PCT, Ortego F, Hernandez-Crespo P, Diaz I, Martinez M, Navaja M, Sucena Ι, Magalhães S, Nagy L, Pace RM, Djuranović S, Smagghe G, Iga M, Christiaens O, Veenstra JA, Ewer J, Villalobos RM, Hutter JL, Hudson SD, Velez M, Yi SV, Zeng J, PiresdaSilva A, Roch F, Cazaux M, Navarro M, Zhurov V, Acevedo G, Bjelica A, Fawcett JA, Bonnet E, Martens C, Baele G, Wissler L, Sanchez-Rodriguez A, Tirry L, Blais C, Demeestere K, Henz SR, Gregory TR, Mathieu J, Verdon L, Farinelli L, Schmutz J, Lindquist E, Feyereisen R, Van de Peer Y (2011) The genome of Tetranychus urticae reveals herbivorous pest adaptations. Nature 479:487492
He YY, Hader DP (2002) UV-B-induced formation of reactive oxygen species and oxidative damage of the cyanobacterium Anabaena sp.: protective effects of ascorbic acid and N-acetyl-l-cysteine. J Photochem Photobiol, B 66:115–124
Heck DE, Vetrano AM, Mariano TM, Laskin JD (2003) UVB light stimulates production of reactive oxygen species unexpected role of catalase. J Biol Chem 278:22432–22436
Jurkiewicz BA, Buettner GR (1994) Ultraviolet light-induced free radical formation in skin: an electron paramagnetic resonance study. Photochem Photobiol 59:1–4
Malloy KD, Holman MA, Mitchell D, Detrich HW (1997) Solar UVB-induced DNA damage and photoenzymatic DNA repair in Antarctic zooplankton. Proc Natl Acad Sci 94:1258–1263
McMurtry JA, Croft BA (1997) Life styles of phytoseiid mites and their role in biological control. Ann Rev Entomol 42:291–321
Miki W (1991) Biological functions and activities of animal carotenoids. Pure Appl Chem 63:141–146
Murata Y, Osakabe M (2013) The Bunsen-Roscoe reciprocity law in ultraviolet-B induced mortality of the two-spotted spider mite Tetranychus urticae. J Insect Physiol 59:241–247
Murata Y, Osakabe M (2014) Factors affecting photoreactivation in UVB-irradiated herbivorous spider mite (Tetranychus urticae). Exp Appl Acarol 62:253–265
Ohno S, Kijima K (2012) A handbook for identification and control spider mites on vegetables and ornamental Plants in the Ryukyus. Okinawa Plant Protection Association, Naha, p 52 (in Japanese)
Ohtsuka K, Osakabe M (2009) Deleterious effects of UV-B radiation on herbivorous spider mites: they can avoid it by remaining on lower leaf surfaces. Environ Entomol 38:920–929
Onzo A, Sabelis MW, Hanna R (2010) Effect of ultraviolet radiation and the role of refuges in plant structures. Environ Entomol 39:695–701
Sakai Y, Osakabe M (2010) Spectrum-specific damage and solar ultraviolet radiation avoidance in the two-spotted spider mite. Photochem Photobiol 86:925–932
Sakai Y, Sudo M, Osakabe M (2012) Seasonal changes in the deleterious effects of solar ultraviolet-B radiation on eggs of the twospotted spider mite, Tetranychus urticae (Acari: Tetranychidae). Appl Entomol Zool 47:67–73
Shindo Y, Witt E, Han D, Packer L (1994) Dose-response effects of acute ultraviolet irradiation on antioxidants and molecular markers of oxidation in murine epidermis and dermis. J Invest Dermatol 102:470–475
SPSS (2013) IBM SPSS Statistics for Windows, Version 22.0. IBM Corp, Armonk
Suzuki T, Takashima T, Izawa N, Watanabe M, Takeda M (2008) UV radiation elevates arylalkylamine N-acetyltransferase activity and melatonin content in the two-spotted spider mite, Tetranychus urticae. J Insect Physiol 54:1168–1174
Suzuki T, Watanabe M, Takeda M (2009) UV tolerance in the two-spotted spider mite, Tetranychus urticae. J Insect Physiol 55:649–654
Suzuki T, Yoshioka Y, Tsarsitalidou O, Ntalia V, Ohno S, Ohyama K, Kitashima Y, Gotoh T, Takeda M, Koveos D (2014) A LED-based UV-B irradiation system for tiny organisms: system description and demonstration experiment to determine the hatchability of eggs from four Tetranychus spider mite species from Okinawa. J Insect Physiol 62:1–10
Tachi F, Osakabe M (2012) Vulnerability and behavioral response to ultraviolet radiation in the components of a foliar mite prey-predator system. Naturwissenschaften 99:1031–1038
Van Lenteren JC (2012) The state of commercial augmentative biological control: plenty of natural enemies, but a frustrating lack of uptake. Biocontrol 57:1–20
Veerman A (1974) Carotenoid metabolism in Tetranychus urticae Koch (Acari: Tetranychidae). Comp Biochem Physiol B 47:101–116
Veerman A (1985) Diapause. In: Helle W, Sabelis MW (eds) Spider mites: their biology, natural enemies and control, vol 1A. Elsevier, Amsterdam, pp 279–316
Zhang GH, Liu H, Wang JJ, Wang ZY (2014) Effect of thermal stress on lipid peroxidation and antioxidant enzyme activities of the predatory mite, Neoseiulus cucumeris (Acari: Phytoseiidae). Exp Appl Acarol 64:73–85
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koveos, D.S., Suzuki, T., Terzidou, A. et al. Egg hatching response to a range of ultraviolet-B (UV-B) radiation doses for four predatory mites and the herbivorous spider mite Tetranychus urticae . Exp Appl Acarol 71, 35–46 (2017). https://doi.org/10.1007/s10493-016-0102-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-016-0102-x