Abstract
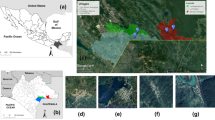
The ranges of many tick species are changing due to climate change and human alteration of the landscape. Understanding tick responses to environmental conditions and how sampling method influences measurement of tick communities will improve our assessment of human disease risk. We compared tick sampling by three collection methods (dragging, CO2 trapping and rodent surveys) in adjacent forested and grassland habitats in the lower Midwest, USA, and analyzed the relationship between tick abundance and microclimate conditions. The study areas were within the overlapping ranges of three tick species, which may provide conditions for pathogen exchange and spread into new vectors. Dermacentor variabilis (American dog tick) was found using all methods, Amblyomma americanum (lonestar tick) was found by dragging and CO2 trapping and Ixodes scapularis (blacklegged deer tick) was found only on rodents. Proportion of each species differed significantly among sampling methods. More ticks were found in forests compared to open habitats. Further, more ticks were collected by dragging and from rodents in hotter, drier conditions. Our results demonstrate that multiple sampling methodologies better measure the tick community and that microclimate conditions strongly influence the abundance and activity of individual tick species.




Similar content being viewed by others
References
Allan BF, Dutra HP, Goessling LS, Barnett K, Chase JM, Marquis RJ, Pang G, Storch GA, Thach RE, Orrock JL (2010) Invasive honeysuckle eradication reduces tick-borne disease risk by altering host dynamics. Proc Natl Acad Sci USA 107:18523–18527
Altizer S, Ostfeld RS, Johnson PTJ, Kutz S, Harvell CD (2013) Climate change and infectious diseases: from evidence to a predictive framework. Science 341:514–519
Anderson JF, Johnson RC, Magnarelli LA, Hyde FW, Myers JE (1986) Peromyscus leucopus and Microtus pennsylvanicus simultaneously infected with Borrelia burgdorferi and Babesia microti. J Clin Microbiol 23:135–137
Apperson CS, Engber B, Nicholson WL, Mead DG, Engel J, Yabsley MJ, Dail K, Johnson J, Watson DW (2008) Tick-borne diseases in North Carolina: is “Rickettsia amblyommii” a possible cause of rickettsiosis reported as rocky mountain spotted fever? Vector Borne Zoonotic Dis 8:597–606
Barre N, Garris GI, Lorvelec O (1997) Field sampling of the tick Amblyomma variegatum (Acari: Ixodidae) on pastures in guadeloupe; attraction of CO2 and/or tick pheromones and conditions of use. Exp Appl Acarol 21:95–108
Bertrand MR, Wilson ML (1996) Microclimate-dependent survival of unfed adult Ixodes scapularis (Acari: Ixodidae) in nature: life cycle and study design implications. J Med Entomol 33:619–627
Dantas-Torres F, Chomel BB, Otranto D (2012) Ticks and tick-borne diseases: a one health perspective. Trends Parasitol 28:437–446
Eisen L (2007a) A call for renewed research on tick-borne Francisella tularensis in the Arkansas-Missouri primary national focus of Tularemia in humans. J Med Entomol 44:389–397
Eisen L (2007b) Seasonal pattern of host-seeking activity by the human-biting adult life stage of Dermacentor andersoni (Acari:Ixodidae). J Med Entomol 44:359–366
Falco RC, Fish D (1991) Horizontal movement of adult Ixodes dammini (Acari, Ixodidae) attracted To Co2-baited traps. J Med Entomol 28:726–729
Falco RC, Fish D (1992) A comparison of methods for sampling the deer tick, Ixodes dammini, in a Lyme-disease endemic area. Exp Appl Acarol 14:165–173
Gage KL, Ostfeld RS, Olson JG (1995) Nonviral vector-borne zoonoses associated with mammals in The United-States. J Mammal 76:695–715
Harlan HJ, Foster WA (1990) Micrometeorological factors affecting field host-seeking activity of adult Dermacentor variabilis (Acari, Ixodidae). J Med Entomol 27:471–479
Hawlena H, Rynkiewicz E, Toh E, Alfred A, Durden LA, Hastriter MW, Nelson DE, Rong R, Munro D, Dong Q, Fuqua C, Clay K (2013) The arthropod, but not the vertebrate host or its environment, dictates bacterial community composition of fleas and ticks. ISME J 7:221–223
Hofmeister EK, Kolbert CP, Abdulkarim AS, Magera JMH, Hopkins MK, Uhl JR, Ambyaye A, Telford SR III, Cockerill FR III, Persing DH (1998) Cosegregation of a novel Bartonella species with Borrelia burgdorferi and Babesia microti in Peromyscus leucopus. J Infect Dis 177:409–416
Kensinger BJ, Allan BF (2011) Efficacy of dry ice-baited traps for sampling Amblyomma americanum (Acari: Ixodidae) varies with life stage but not habitat. J Med Entomol 48:708–711
Klompen JSH, Black WCV, Keirans JE, Oliver JH Jr (1996) Evolution of ticks. Annu Rev Entomol 41:141–161
Kollars TM, Oliver JH, Kollars PG, Durden LA (1999) Seasonal activity and host associations of Ixodes scapularis (Acari : Ixodidae) in southeastern Missouri. J Med Entomol 36:720–726
Kollars TM, Oliver JH, Durden LA, Kollars PG (2000) Host associations and seasonal activity of Amblyomma americanum (Acari : ixodidae) in Missouri. J Parasitol 86:1156–1159
Landsberg J, Sands P (2011) Physiological ecology of forest production: principles, processes and models, vol 4. Academic Press, London
Leger E, Vourc’h G, Vial L, Chevillon C, McCoy KD (2013) Changing distributions of ticks: causes and consequences. Exp Appl Acarol 59:219–244
LoGiudice K, Ostfeld RS, Schmidt KA, Keesing F (2003) The ecology of infectious disease: effects of host diversity and community composition on Lyme disease risk. Proc Natl Acad Sci USA 100:567–571
Needham GR, Teel PD (1991) Off-host physiological ecology of ixodid ticks. Annu Rev Entomol 36:659–681
Ostfeld R, Keesing F (2000) The function of biodiversity in the ecology of vector-borne zoonotic diseases. Can J Zool Revue Canadienne De Zoologie 78:2061–2078
Ostfeld RS, Cepeda OM, Hazler KR, Miller MC (1995) Ecology of lyme-disease—habitat associations of ticks (Ixodes scapularis) in a rural landscape. Ecol Appl 5:353–361
Perret JL, Rais O, Gern L (2004) Influence of climate on the proportion of Ixodes ricinus nymphs and adults questing in a tick population. J Med Entomol 41:361–365
R Core Team (2013) R: a language and environment for statistical computing. Austria, Vienna
Randolph SE, Storey K (1999) Impact of microclimate on immature tick-rodent host interactions (Acari:Ixodidae): implications for parasite transmission. J Med Entomol 36:741–748
Rulison EL, Kuczaj I, Pang G, Hickling GJ, Tsao JI, Ginsberg HS (2013) Flagging versus dragging as sampling methods for nymphal Ixodes scapularis (Acari:Ixodidae). J Vector Ecol 38:163–167
Rynkiewicz E, Hawlena H, Durden L, Hastriter M, Demas G, Clay K (2013) Associations between innate immune function and ectoparasites in wild rodent hosts. Parasitol Res 112:1763–1770
Schmidt KA, Ostfeld RS (2003) Mice in space: space use predicts the interaction between mice and songbirds. Ecology 84:3276–3283
Schulze TL, Jordan RA, Hung RW (1997) Biases associated with several sampling methods used to estimate abundance of Ixodes scapularis and Amblyomma americanum (Acari : Ixodidae). J Med Entomol 34:615–623
Solberg VB, Olson JG, Boobar LR, Burge JR, Lawyer PG (1996) Prevalence of Ehrlichia chaffeensis, spotted fever group Rickettsia, and Borrelia spp infections in ticks and rodents at Fort Bragg, North Carolina. J Vector Ecol 21:81–84
Teel PD, Fleetwood SC (1982) An integrated sensing and data acquisition system designed for unattended continuous monitoring of microclimate relative humidity and its use to determine the influence of vapor pressure deficits on tick acari ixodoidea activity. Agric Meteorol 27:145–154
Whitaker JO (1982) Ectoparasites of mammals of Indiana. Indiana Academy of Science, Indianapolis
Acknowledgments
The authors would like to acknowledge Hadas Hawlena for help with this project during 2009. We thank A. Avalos, A. Bechtel, and J. Kagemann for assistance with field collections of ticks, J.O. Whitaker and R. Pinger for help with tick identifications, the property owners and managers for access to study sites, S. Dickenson for statistical consulting, and B.F. Allan for advice in constructing CO2 traps and analysis of microclimate data. Funding was provided through an Indiana Academy of Science Senor Researcher Grant and NSF Doctoral Dissertation Improvement Grant (Award #1110514) to ECR, NSF-NIH grant DEB-03268742 to KC, the Indiana Metabolomics and Cytomics Initiative of Indiana University (IU) awarded to KC, and by the IU Center for Research in Environmental Sciences awarded to KC. All research was carried out with approval from the Bloomington Institutional Animal Care and Use Committee (permit # 11-001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rynkiewicz, E.C., Clay, K. Tick community composition in Midwestern US habitats in relation to sampling method and environmental conditions. Exp Appl Acarol 64, 109–119 (2014). https://doi.org/10.1007/s10493-014-9798-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-014-9798-7