Abstract
The capsular polysaccharides are an important virulence factor of Streptococcus iniae, protecting the bacterium from destruction and clearance by the immune system. The cpsJ gene encodes a putative UDP-glucose epimerase involved in the capsule synthesis system. To determine the role of the CpsJ protein in the production of the capsule, a ΔcpsJ mutant was generated and analyzed by comparing its growth performances and virulence with those of the wild type (WT) strain. The ΔcpsJ mutant had longer chains, smaller colonies, and a slower growth rate and decreased optical density than the WT, suggesting that the ΔcpsJ mutant produces less capsular polysaccharide. The ΔcpsJ mutant was more able to adhere to and invaded epithelioma papulosum cyprinid cells (EPCs) when its virulence in vitro was compared with that of the WT, but survived less well in the whole blood of channel catfish. When a channel catfish infection model was used to determine the virulence of the ΔcpsJ mutant in vivo, the mutant caused an increase in survival with the mutant (53.33 %) versus the WT (26.67 %). In summary, mutation of the cpsJ gene influenced both the capsule synthesis and virulence of S. iniae.
Similar content being viewed by others
Introduction
Streptococcus iniae is a zoonotic pathogen of both fish and humans. It is a serious pathogen of fish, causing invasive disease and significant losses to aquaculture farms. Infection can be transferred to humans through skin abrasions (Weinstein et al. 1997). S. iniae was first reported in 1976, when it was isolated from skin lesions on Inia geoffrensis (Pier and Madin 1976), and it is known to infect at least 30 species of wild and farmed fishes today, including tilapia (Perera et al. 1994), trout (Eldar and Ghittino 1999), and channel catfish (Ictalurus punctatus). The channel catfish was once recognized to be resistant to streptococcal infection, but its infection with S. iniae has reversed that opinion in recent years. Infected channel catfish become lethargic, swim erratically, display petechial hemorrhage on the skin, and die within several days, with a mortality rate of 50–60 % (Chen et al. 2011). With the wide use of capsular vaccines, two serotypes of this species have been identified. Sera against serotype I showed no cross-reactivity to serotype II capsular polysaccharide (CPS), whereas antisera to serotype II partially agglutinated serotype I CPS (Bachrach et al. 2001).
The CPS occurs around bacterial cells, including S. iniae (Lowe et al. 2007). During cell invasion, CPS plays an important role in evading the host’s immune responses, adapting the bacterium to the host environment (Marques et al. 1992; Millard et al. 2012). The S. iniae CPS is mainly composed of l-fucose, d-mannose, d-galactose, d-glucose, d-glucuronic acid, N-acetyl-d-galactosamine (Locke et al. 2007). UDP-galactose plays an important role as a substrate in the biosynthesis of CPS and the virulence of both Gram-negative and Gram-positive bacteria. UDP-galactose is synthesized when it is converted from UDP-glucose by UDP-glucose epimerase. A mutant strain of S. iniae deficient in UDP-glucose epimerase showed reduced resistance to host serum and reduced biofilm formation (Fry et al. 2000; Zou et al. 2013).
In S. iniae, the cpsG gene, encoding a putative UDP-glucose 4 epimerase, affects the final molecular constituent of the CPS polysaccharides (Millard et al. 2012). However, cpsJ has not yet been studied, although it is predicted to encode an UDP-glucose epimerase. To explore the functions of cpsJ in CPS synthesis and the virulence of S. iniae, we constructed a cpsJ knockout mutant (ΔcpsJ) and compared it with the WT parental strain using a series of tests in vitro and in vivo.
Materials and methods
Animal ethics
Channel catfish were used for the experiments. Fish weighing an average of 80–100 g were purchased from a commercial fish farm and acclimated for a minimum of 2 weeks in a flowing water system. Twenty fish were maintained in a 500 L tank containing 400 L of water/tank, and fed a commercial feed equivalent to 3 % of the fish bodyweight daily. All experiments were performed in accordance with the current legislation on handling experimental animals.
Bacterial strains and growth conditions
S. iniae (DGX07, type I) was isolated from diseased channel catfish (Chen et al. 2011) and incubated on Todd–Hewitt yeast agar (THYA) plates at 30 °C. The growth rate was measured by monitoring the optical density at a wavelength of 600 nm (OD600) in Todd–Hewitt yeast broth (THYB). Chloramphenicol (Cm, 10 μg/ml) and erythromycin (Erm, 5 μg/ml) was used to select the S. iniae mutant. Escherichia coli was incubated in LB broth and selected with 10 μg/ml Cm, 500 μg/ml Erm, or 100 μg/ml ampicillin (Amp).
Plasmids and primers
The Cm-resistance-encoding vector pACYC184 is maintained in our laboratory. The plasmid pMD19-T used for cloning was purchased from Takara (China). The thermosensitive suicide vector pJRS233 was a kind gift from Dr. Neely, Wayne State University. All the primers used in this study are shown in Table 1 and synthesized by Sangon, PR China.
Cells and culture
Epithelioma papulosum cyprinid cells (EPCs), donated by the Pearl River Fisheries Institute, were maintained in Dulbecco’s modified Eagle’s medium (DMEM; Gibco BRL) with 10 % (v/v) heat-inactivated fetal bovine serum (FBS; Gibco BRL). The cells were passaged at least 10 times.
Mutagenesis of the cpsJ gene
The ΔcpsJ mutant was created in S. iniae (DGX07) with in-frame allelic replacement and was selected for its resistance to Cm. A thermosensitive suicide vector, pJRS233, carrying left and right arms was constructed (Fig. 1). Briefly, the primers cpsJ- left F1/R1 and cpsJ- right F2/R2 were used to amplify the left and right fragments, respectively. A BamHI site and an XhoI site were added to the 5′ ends of the left F1 primer and the right R2 primer, respectively. The Cm-resistance gene was amplified from the pACYC184 vector with primers of F3/R3. The three amplified genes were then combined in a fusion PCR and the product was cloned into the pMD19-T vector to generate the plasmid pMD19-T–ΔcpsJ. pMD19-T–ΔcpsJ was then digested with BamHI/XhoI and cloned into the PJRS233 vector that had been digested with the same restriction enzymes. The plasmid was used to transform E. coli DH5α cells. S. iniae (DGX07) was transformed with the recombinant plasmid by electroporation, designated PJRS233–ΔcpsJ, and the mutant strain was selected as described previously (Ruiz et al. 1998).
PCR screening of ΔcpsJ
The chromosomal structure of the resulting mutant was confirmed by PCR. In the successful cpsJ knockout mutant (designated ‘ΔcpsJ mutant’), the CM-resistance gene should replace the cpsJ gene. Therefore, the PCR product amplified with primers F4/R4 was sequenced to confirm the mutant (Fig. 1). The PCR amplicons generated with primers cpsJ F/R and Cm F/R were also used to detect the mutants. To determine the effects of knockdown on the expression of genes downstream from cpsJ in the mutant strain, primers cpsK F/R were used for reverse transcription (RT)–PCR with cDNA templates.
Growth analysis
The S. iniae ΔcpsJ mutant was cultured to determine the bacterial growth characteristics. (1) Colonial and bacterial morphology. The WT and ΔcpsJ mutant were cultured in THYA at 30 °C, and the colonial and bacterial morphology were recorded after 48 h. (2) Growth curve. Bacteria cultured in THYB to OD600 0.08 were added to a 96-well plate, and the growth rate was monitored every 30 min during 8 h with readings of OD600 (Fuller et al. 2002).
Bacterial invasion and attachment assays
Invasion assay
The invasion of EPCs was quantified as described previously, with minor modifications (Burnham et al. 2007). Briefly, EPCs were grown to confluence in 96-well plates, and then the medium was changed to 100 μl of DMEM containing 2 % FBS. Bacteria (WT or ΔcpsJ mutant) from a mid-log-phase culture were diluted in DMEM, and 100 μl of the dilution was added to the wells at a (bacteria to cells) multiplicity of infection of 50. After centrifugation at 350×g for 5 min, the plate was incubated at 28 °C for 1 h with 5 % CO2 to allow the internalization of the bacteria. The unbound bacteria were removed by washing the cells three times with DMEM. The bacteria bound to the surfaces of the cells were killed by incubation for another 2 h with fresh DMEM containing 20 μg/ml penicillin (Invitrogen) and 200 μg/ml gentamicin (Invitrogen). The cells were then washed with phosphate-buffered saline (PBS) and lysed with 0.1 % Triton X-100 (Sigma). Tenfold serial dilutions of the lysates were then spread onto THYA, and incubated for 48 h to determine the number of colony-forming units (CFU) that had invaded the monolayer.
Adherence assay
Adherence to EPCs was tested in a similar manner, except that no antibiotics were used. Briefly, EPC cells were infected with the WT or ΔcpsJ mutant for 30 min to permit bacterial attachment and the monolayers were washed five times with PBS to remove nonadherent bacteria and the numbers of CFU were counted as described above.
Whole-blood killing assay
Anticoagulated fresh blood was extracted from the caudal vein of three healthy channel catfish. Bacteria (WT or ΔcpsJ), adjusted to the mid-log-phase concentration (approximately 300 CFU in total) with PBS, was mixed with 300 μl of whole blood. The mixtures were incubated at 30 °C for 1 h and plated onto THYA after serial dilution, to determine the CFU. The survival rate (%) was calculated as (the number of remaining bacteria/the number of starting bacteria) × 100.
Challenge of channel catfish
Bacteria from a mid-log-phase culture were washed and adjusted to 1 × 108 CFU with PBS. Three groups of 20 fish each were injected intraperitoneally with WT S. iniae, ΔcpsJ, or PBS. All the groups were established in triplicate. Postchallenge morbidity and mortality were monitored over a period of 2 weeks.
Statistical analysis
Experiments including the growth analysis, invasion and attachment assays, whole-blood killing assay and challenge assays were performed in triplicate The data were analyzed with SPSS 16.0 (SPSS Institute, Inc.,).
Results
Mutagenesis of the cpsJ gene
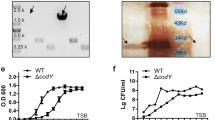
The ΔcpsJ mutant was constructed in S. iniae (DGX07) by homologous replacement. After amplification by fusion PCR, a product of 2000 bp was obtained and was ligated into plasmid pJRS233. The WT strain was then transformed with the combined plasmid. With a series of antibiotic screens, the knockout mutant was selected from the WT strain and identified by PCR using various specific primers. Using the ΔcpsJ mutant as the DNA template and primers F1/R3, the generated product indicated that a cpsI-Cm-cpsK fusion product of 2000 bp was obtained and ligated in the ΔcpsJ mutant DNA (Fig. 2a), whereas a band of ~825 bp was amplified from WT DNA with primers cpsJ F/R (Fig. 2b). DNA sequencing confirmed that the amplicon generated from the ΔcpsJ mutant DNA with primers F4/R4 encoded a Cm-resistance gene which had replaced the cpsJ gene (Fig. 2d). RT-PCR using amplicon primers cpsK F/R showed no polar effects to the cpsK gene in the ΔcpsJ mutant (Fig. 2c), and the sequence data is identical to the Genebank’s cpsK of S. iniae (accession no.AY904444.1).
ΔcpsJ mutant identified with PCR and sequencing. a PCR analysis with primers F1/R3; b PCR analysis with primers cpsJ F/R; c RT–PCR analysis with primers cpsK F/R; d sequence analysis of production from PCR with primers F4/R4. Lane M1, DL10000 marker; lane M2, DL2000 marker; ΔcpsJ, PCR product from ΔcpsJ mutant DNA/cDNA template; WT, PCR product from WT DNA/cDNA template; N, Negative control
CpsJ modulates S. iniae growth characteristics
Morphology
After the ΔcpsJ and WT strains were cultured on THYA for 48 h, they both formed wet, circular, ridged, ivory-white colonies, but the ΔcpsJ colonies were smaller than the WT colonies (Fig. 3a, b). Both strains were Gram positive, but the chain length of the ΔcpsJ mutant was longer than that of the WT strain (Fig. 3c, d).
Growth rate
Cultures of both strains were adjusted to OD600 0.08 and were cultured for 8 h in 96-well plate, and the growth rate was monitored at 30 min interval respectively. The two strains reached stationary phase at the same time. However, the optical density of the WT bacteria was notably greater than that of the ΔcpsJ mutant (Fig. 3e).
Effects of cpsJ gene on S. iniae growth characteristics. The colonial morphology of WT strain (a) and ΔcpsJ mutant (b) by macroscopic observation; c WT strain showed Gram positive and short chain length (Gram staining, ×1000); d ΔcpsJ mutant showed Gram positive and long chains length(Gram staining, ×1000). e Growth curves for WT strain and ΔcpsJ mutant. ΔcpsJ mutant showed significantly reduced growth compared with the WT strain. In stationary phase, the bacterial concentration of WT was twice higher than that of ΔcpsJ mutant
CpsJ modulates S. iniae’s capacity to adhere to and invade EPCs
Previous studies aimed at investigating the effects of CPS deficiency showed that the deficient mutant displayed enhanced capacities for both cellular adherence and invasion compared with those of the WT strain (Hammer schmidt et al. 2005; Jeng et al. 2003). To determine whether the loss of the cpsJ gene plays any role in bacterial virulence, the adherence and invasiveness of the ΔcpsJ mutant were tested using EPCs. Compared with the WT strain, the ΔcpsJ mutant displayed a ~20-fold increase in adherence and a fivefold increase in invasion (Fig. 4a, b).
CpsJ modulates bacterial resistance to whole-blood killing
The innate immune system is a fundamental defense mechanism of fish and plays an important role in the acquired immune response. Blood contains monocytes, inflammatory cells, cytotoxic cells, complement, and other humoral factors that are capable of killing bacteria (Magnadóttir 2006). Therefore, the resistance of the ΔcpsJ mutant to fresh whole blood of the channel catfish was investigated in this study. After coincubation with whole blood, the WT strain showed greater resistance to clearance by the innate immune defenses of fresh whole channel catfish blood than the ΔcpsJ mutant. The survival rate of the WT strain (137.4 %) was significantly higher (p < 0.01) than that of the mutant strain (45.9 %) (Fig. 5).
CpsJ contributes to the virulence of S. iniae in channel catfish
To compare the virulence of the WT and ΔcpsJ mutant S. iniae strains, channel catfish were challenged with the same concentrations of the WT strain or ΔcpsJ mutant, and PBS was used as the negative control. After 2 weeks, the survival rates of the groups were 26.7, 53.3 and 96.7 %, respectively (Fig. 6). The fish that succumbed to S. iniae showed no classic signs in the first 2 days, but on day 3 after challenge, the fish died with typical signs of Streptococcus infection, including lethargy and ataxia.
Discussion
CPS are a major component of the bacterial capsule, and are encoded by the capsular polysaccharide synthesis operon, in a chromosomal region of approximately 21 kb. Several capsule synthesis genes have been analyzed, and their importance in capsule synthesis and virulence has been determined with site-directed mutants. CpsA is responsible for the determination of chain length, and its knockout reduced the production of CPS with longer chains and greater buoyancy. cpsD encodes an autophosphorylating tyrosine kinase that controls capsule polymerization and export, and the knockout of this gene caused a marked reduction in capsular monosaccharides and a high degree of attenuation (Locke et al. 2007). CpsY is a regulator of CPS, so the cpsY mutant showed slightly less encapsulation (Lowe et al. 2007). The loss of acidic polysaccharides was also identified in insertion mutants of cpsH, cpsM, cpsI, and open reading frame (ORF) 276 of S. iniae (Shutou et al. 2007). Genes associated with the polysaccharide configuration, such as cpsG and pgm, have also been studied and identified as targets for vaccine development (Buchanan et al. 2005; Millard et al. 2012). The streptococcal cpsJ gene was predicted to encode a UDP-glucose epimerase that participates in the synthesis of UDP-Gal. Studies have shown that strains with abundant capsules produce shorter chain lengths (Ekstedt and Stollerman 1960). Therefore, we suspected that CpsJ influences the synthesis of CPS, leading to longer chains, smaller colonies, and slower growth.
Previous studies have shown that S. iniae adheres to, invades, and survives in host cells and also induces the apoptosis of phagocytes. In this way, S. iniae penetrates deeper into the organism, its blood and nervus centralis, producing a systemic infection (Lowe et al. 2007). In vitro assays with EPCs were performed to determine whether the loss of the cpsJ gene disturbs capsular biosynthesis and diminishes the virulence of the bacterium for its host, using S. iniae WT as a control. Increased adherence and invasiveness were observed in the ΔcpsJ mutant. Hammerschmidt et al. (2005) investigated the adherence of S. pneumoniae to bronchial epithelial cells and found that an uncapsulated mutant bound much more extensively than the encapsulated parental strain, and these high levels of adherence may have correlated with its enhanced invasive capacity. Allelic exchange mutagenesis of cpsD also caused an increase in adherence to and invasion of epithelial cells (Locke et al. 2007). The increased affinity of the ΔcpsJ mutant of S. iniae for EPCs may be attributable to its capsule deficiency, which exposes surface-associated proteins or factors involved in host cell binding. Therefore, we suggest that the ΔcpsJ mutant has fewer capsular polysaccharides than the WT, allowing its easier adherence to and invasion of host cells (Hulse et al. 1993). A change in the surface charge of the bacterium may be another explanation of our findings, and further research into the capsular components is required.
Although the capsule reduces the bacterial attachment to and invasion of epithelial cells, it simultaneously reduces the phagocytosis of the bacterium by the host, so when the expression of the capsule is reduced, the bacterium becomes more susceptible to phagocytic clearance and attenuated virulence (Buchanan et al. 2005). In this study, the survival rate of the ΔcpsJ mutant was significantly reduced when it was cultured with whole catfish blood, indicating that the ΔcpsJ mutant is sensitive to the innate immune system of its host. We also used a channel catfish infection model to compare the virulence of the WT and the ΔcpsJ mutant, and after 2 weeks, the ΔcpsJ mutant showed a statistically significant reduction in virulence. Therefore, the replacement of the cpsJ gene reduced the virulence of S. iniae both in vivo and in vitro, so we conclude that cpsJ may be an important gene in the capsule operon.
Conclusions
The mutant ΔcpsJ strain showed an increased chain length, smaller colonies, a slower growth rate, and reduced virulence. These results imply that the CpsJ protein influences the synthesis of the CPS and bacterial virulence. Our findings extend the basic information available on the CPS and should allow the development of a genetically engineered vaccine. Further work is required to study the changes in the ΔcpsJ mutant at the molecular level of the capsule, including the components altered and changes in the levels of CPS.
References
Bachrach G, Zlotkin A, Hurvitz A, Evans DL, Eldar A (2001) Recovery of Streptococcus iniae from diseased fish Previously Vaccinated with a Streptococcus Vaccine. Appl Environ Microbiol 67:3756–3758
Buchanan JT, Stannard JA, Lauth X, Ostland VE, Powell HC, Westerman ME, Nizet V (2005) Streptococcus iniae phosphoglucomutase is a virulence factor and a target for vaccine development. Infect Immun 73:6935–6944
Burnham C-AD, Shokoples SE, Tyrrell GJ (2007) Invasion of HeLa cells by group B streptococcus requires the phosphoinositide-3-kinase signalling pathway and modulates phosphorylation of host-cell Akt and glycogen synthase kinase-3. Microbiology 153:4240–4252
Chen D-f, Wang K-y, Geng Y, Wang J, Huang L-y (2011) Streptococcus iniae isolated from channel catfish (Ictalurus punctatus) in China. Isr J Aquaculture-Bamidgeh 593:1–7
Ekstedt RD, Stollerman GH (1960) Factors affecting the chain length of group A Streptococci I demonstration of a metabolically active chain-splitting system. J Exp Med 112:671–686
Eldar A, Ghittino C (1999) Lactococcus garvieae and Streptococcus iniae infections in rainbow trout Oncorhynchus mykiss: similar, but different diseases. Dis aquat org 36:227–231
Fry BN, Feng S, Chen Y-Y, Newell DG, Coloe PJ, Korolik V (2000) The galE gene of Campylobacter jejuni is involved in lipopolysaccharide synthesis and virulence. Infect immun 68:2594–2601
Fuller JD, Camus AC, Duncan CL, Nizet V, Bast DJ, Thune RL, Low DE, de Azavedo JCS (2002) Identification of a streptolysin S-associated gene cluster and its role in the pathogenesis of Streptococcus iniae disease. Infect Immun 70(10):5730–5739
Hammerschmidt S, Wolff S, Hocke A, Rosseau S, Müller E, Rohde M (2005) Illustration of pneumococcal polysaccharide capsule during adherence and invasion of epithelial cells. Infect Immun 73:4653–4667
Hulse ML, Smith S, Chi E, Pham A, Rubens C (1993) Effect of type III group B streptococcal capsular polysaccharide on invasion of respiratory epithelial cells. Infect Immun 61:4835–4841
Jeng A, Sakota V, Li Z, Datta V, Beall B, Nizet V (2003) Molecular genetic analysis of a group A Streptococcus operon encoding serum opacity factor and a novel fibronectin-binding protein, SfbX. J Bacteriol 185:1208–1217
Locke JB, Colvin KM, Datta AK, Patel SK, Naidu NN, Neely MN, Nizet V, Buchanan JT (2007) Streptococcus iniae capsule impairs phagocytic clearance and contributes to virulence in fish. J Bacteriol 189(4):1279–1287
Lowe BA, Miller JD, Neely MN (2007) Analysis of the polysaccharide capsule of the systemic pathogen Streptococcus iniae and its implications in virulence. Infect Immun 75:1255–1264
Magnadóttir B (2006) Innate immunity of fish (overview). Fish Shellfish Immunol 20:137–151
Marques MB, Kasper D, Pangburn M, Wessels M (1992) Prevention of C3 deposition by capsular polysaccharide is a virulence mechanism of type III group B Streptococci. Infect Immun 60:3986–3993
Millard CM, Baiano JC, Chan C, Yuen B, Aviles F, Landos M, Chong RS, Benedict S, Barnes AC (2012) Evolution of the capsular operon of Streptococcus iniae in response to vaccination. Appl Environ Microbiol 78(23):8219–8226
Perera RP, Johnson SK, Collins MD, Lewis DH (1994) Streptococcus iniae associated with mortality of Tilapia nilotica × T. Aurea hybrids. J Aquat Anim Health 6:335–340
Pier GB, Madin SH (1976) Streptococcus iniae sp. nov., a beta-hemolytic streptococcus isolated from an Amazon freshwater dolphin, Inia geoffrensis. Int J Syst Bacteriol 26:545–553
Ruiz N, Wang B, Pentland A, Caparon M (1998) Streptolysin O and adherence synergistically modulate proinflammatory responses of keratinocytes to group A Streptococci. Mol Microbiol 27:337–346
Shutou K, Kanai K, Yoshikoshi K (2007) Virulence attenuation of capsular polysaccharide-deleted mutants of Streptococcus iniae in Japanese flounder Paralichthys olivaceus. Fish Reaserch 42:41–48
Weinstein MR, Litt M, Kertesz DA, Wyper P, Rose D, Coulter M, McGeer A, Facklam R, Ostach C, Willey BM (1997) Invasive infections due to a fish pathogen, Streptococcus iniae. New England J Med 337(9):589–594
Zou Y, Feng S, Xu C, Zhang B, Zhou S, Zhang L, He X, Li J, Yang Z, Liao M (2013) The role of galU and galE of Haemophilus parasuis SC096 in serum resistance and biofilm formation. Vet Microbiol 162(1):278–284
Acknowledgments
This work supported by Sichuan Technology Support Plannings No’s. 2014NZ0003 and 2014JY0143, Program for Changjiang Scholars and Innovative Research Team in University (No. IRT0848).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Yun Zeng and Yang He has contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zeng, Y., He, Y., Wang, Ky. et al. cpsJ gene of Streptococcus iniae is involved in capsular polysaccharide synthesis and virulence. Antonie van Leeuwenhoek 109, 1483–1492 (2016). https://doi.org/10.1007/s10482-016-0750-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-016-0750-1