Abstract
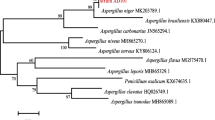
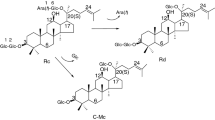
Ginsenosides are the major pharmacological components in ginseng. Microorganisms from a ginseng field were isolated to identify transformation of ginsenosides. Based on HPLC and LC–MS analysis, strain LFJ1403 showed strong activities to transform ginsenoside Rb1 to Rd as the sole product. Phylogenetic analysis of 18S rDNA indicated that LFJ1403 belonged to Aspergillus versicolor. Through comparing four systems of transforming Rb1 to Rd, strain LFJ1403 was found to secrete ginsenoside-converting enzymes in the spore production phase of plate culture. This result suggested that the enzyme could be directly obtained from the plate. The spore suspension, which contained the exocrine enzyme, was easy to prepare and efficient for biotransformation of ginsenoside Rb1 to Rd. Further study showed that the maximum bioconversion rate was 96 % (w/w) in shake flasks when a spore suspension system was used with optimized biotransformation conditions. Scale-up of this system to 2L resulted in an 85 % conversion rate. The ginsenoside Rb1 converting enzyme was separated by gradient HPLC with Q-Sepharose column, and its β-glucosidase activity and Rb1-converting ability was assayed by the 4-Nitrophenyl-β-d-glucopyranoside (PNPG) method and HPLC with C18 column, respectively. We obtained 130 U ml−1 enzymatic activity with the purified β-glucosidase. This is the first report on efficiently converting ginsenoside using extracellular enzyme directly from the fungus spore production phase of solid culture.








Similar content being viewed by others
References
Chang KH, Jo MN, Kim KT, Paik HD (2014) Evaluation of glucosidases of Aspergillus niger strain comparing with other glucosidases in transformation of ginsenoside Rb1 to ginsenosides Rg3. J Ginseng Res 38:47–51
Chen GT et al (2008) Microbial transformation of ginsenoside Rb1 by Acremonium strictum. Appl Microbiol Biotechnol 77:1345–1350
Eklund R, Galbe M, Zacchi G (1990) Optimization of temperature and enzyme concentration in the enzymatic saccharification of steam-pretreated willow. Enzyme Microb Technol 12:225–228
Frias-De Leon MG et al (2011) Phenotypic characteristics of isolates of Aspergillus section Fumigati from different geographic origins and their relationships with genotypic characteristics. BMC Infect Dis 11:116
Gao J et al (2012) Efficient biotransformation for preparation of pharmaceutically active ginsenoside Compound K by Penicillium oxalicum sp. 68. Ann Microbiol 63:139–149
Gao J, Wang J, Cui J, Wang N, Bai Y, Yuan Y, Zhou Y (2014) Purification and characterization of two novel β-glucosidases from Penicillium oxalicum and their application in bioactive ginsenoside production. Biocatal Biotransform 32:199–207
Hegazy ME et al (2015) Microbial biotransformation as a tool for drug development based on natural products from mevalonic acid pathway: a review. J Adv Res 6:17–33
Hsu BY, Lu TJ, Chen CH, Wang SJ, Hwang LS (2013) Biotransformation of ginsenoside Rd in the ginseng extraction residue by fermentation with lingzhi (Ganoderma lucidum). Food Chem 141:4186–4193
Huang C, Wang G, Li H, Xie H, Sun J, Lv H, Lv T (2006) Sensitive and selective liquid chromatography-electrospray ionisation-mass spectrometry analysis of astragaloside-IV in rat plasma. J Pharm Biomed Anal 40:788–793
Kim SN, Ha YW, Shin H, Son SH, Wu SJ, Kim YS (2007) Simultaneous quantification of 14 ginsenosides in Panax ginseng C.A. Meyer (Korean red ginseng) by HPLC-ELSD and its application to quality control. J Pharm Biomed Anal 45:164–170
Lee GW, Kim KR, Oh DK (2012) Production of rare ginsenosides (compound Mc, compound Y and aglycon protopanaxadiol) by β-glucosidase from Dictyoglomus turgidum that hydrolyzes β-linked, but not α-linked, sugars in ginsenosides. Biotechnol Lett 34:1679–1686
Li L et al (2011) Protective effects of ginsenoside Rd against okadaic acid-induced neurotoxicity in vivo and in vitro. J Ethnopharmacol 138:135–141
Li ZY et al (2015) A universal quantitative 1H nuclear magnetic resonance (qNMR) method for assessing the purity of dammarane-type ginsenosides. Phytochem Anal (PCA) 26:8–14
Lin T et al (2012) Promotive effect of ginsenoside Rd on proliferation of neural stem cells in vivo and in vitro. J Ethnopharmacol 142:754–761
Marjamaa K, Toth K, Bromann PA, Szakacs G, Kruus K (2013) Novel Penicillium cellulases for total hydrolysis of lignocellulosics. Enzyme Microb Technol 52(6–7):358–369
Oh JY et al (2014) Investigation of ginsenosides in different tissues after elicitor treatment in Panax ginseng. J Ginseng Res 38:270–277
Quan LH, Min JW, Yang DU, Kim YJ, Yang DC (2012) Enzymatic biotransformation of ginsenoside Rb1 to 20(S)-Rg3 by recombinant β-glucosidase from Microbacterium esteraromaticum. Appl Microbiol Biotechnol 94:377–384
Shi W, Wang Y, Li J, Zhang H, Ding L (2007) Investigation of ginsenosides in different parts and ages of Panax ginseng. Food Chem 102:664–668
Son JW, Kim HJ, Oh DK (2008) Ginsenoside Rd production from the major ginsenoside Rb1 by β-glucosidase from Thermus caldophilus. Biotechnol Lett 30:713–716
Sonia KG, Chadha BS, Badhan AK, Saini HS, Bhat MK (2008) Identification of glucose tolerant acid active β-glucosidases from thermophilic and thermotolerant fungi. World J Microbiol Biotechnol 24:599–604
Wu L, Jin Y, Yin C, Bai L (2012a) Co-transformation of Panax major ginsenosides Rb1 and Rg1 to minor ginsenosides C-K and F1 by Cladosporium cladosporioides. J Ind Microbiol Biotechnol 39:521–527
Wu W, Qin Q, Guo Y, Sun J, Liu S (2012b) Studies on the chemical transformation of 20(S)-protopanaxatriol (PPT)-type ginsenosides Re, Rg2, and Rf using rapid resolution liquid chromatography coupled with quadruple-time-of-flight mass spectrometry (RRLC-Q-TOF-MS). J Agric Food Chem 60:10007–10014
Yokozawa T, Dong E (2001) Role of ginsenoside-Rd in cisplatin-induced renal injury: special reference to DNA fragmentation. Nephron 89(4):433–438
Zeng S et al (2003) Synthesis of 12-epi-ginsenoside Rd and its effects on contractions of rat aortic rings. Chinese Pharmacol Bull 19:282–286
Zhang C, Yu H, Bao Y, An L, Jin F (2001) Purification and characterization of ginsenoside β-glucosidase from ginseng. Chem Pharm Bull 49(7):795–798
Zhao X et al (2009a) A novel ginsenoside Rb1-hydrolyzing β-d-glucosidase from Cladosporium fulvum. Process Biochem 44:612–618
Zhao X et al (2009b) Highly selective biotransformation of ginsenoside Rb1 to Rd by the phytopathogenic fungus Cladosporium fulvum (syn. Fulvia fulva). J Ind Microbiol 36:721–726
Acknowledgments
The authors wish to acknowledge the financial support provided by the National Basic Research Program of China (“973” Program: 2012CB721105) and the Ministry of Science and Technology of China (“863” Program: 2012AA02A701).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, F., Guo, X. & Lu, W. Efficient biotransformation of ginsenoside Rb1 to Rd by isolated Aspergillus versicolor, excreting β-glucosidase in the spore production phase of solid culture. Antonie van Leeuwenhoek 108, 1117–1127 (2015). https://doi.org/10.1007/s10482-015-0565-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-015-0565-5