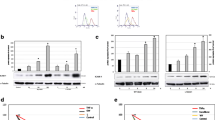
Abstract
Cathepsin B has been shown to be important in angiogenesis; therefore, understanding its regulation in endothelial cells should provide fundamental information that will aid in the development of new treatment options. Peroxisome proliferator-activated receptors (PPARs) have been shown to have anti-inflammatory, anti-angiogenic and anti-tumorigenic properties. We explored the influence of a PPARα agonist on cathepsin B expression in human endothelial cells. The PPARα agonist, Wy14643, was found to inhibit cathepsin B protein expression. Further studies demonstrated the Wy14643-dependent but PPARα-independent suppression of cathepsin B. This has been previously described for other PPAR agonists. Wy14643 suppressed the accumulation of cathepsin B mRNA, which was accompanied by the selective suppression of a 5′-alternative splice variant. Consistent with these results, luciferase promoter assays and electrophoretic mobility shift analysis demonstrated that the suppression was facilitated by reduced binding of the transcription factors USF1/2 to an E-box within the cathepsin B promoter. Additionally, Wy14643 treatment resulted in a reduction in cathepsin B half-life, suggesting a posttranslational regulatory mechanism. Overall, our results suggest that the PPARα-dependent anti-angiogenic action of Wy14643 seems to be mediated, in part, by Wy14643-dependent but PPARα-independent regulation of cathepsin B expression.






Similar content being viewed by others
Abbreviations
- COX-2:
-
Cyclooxygenase-2
- DMSO:
-
Dimethylsulfoxide
- EMSA:
-
Electromobility shift assay
- HUVEC:
-
Human umbilical vein endothelial cell
- IκBα:
-
Inhibitor of kappa Bα
- LPS:
-
Lipopolysaccharide
- NF-κB:
-
Nuclear factor-kappa B
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- USF:
-
Upstream stimulatory factor
References
Daynes RA, Jones DC (2002) Emerging roles of PPARs in inflammation and immunity. Nat Rev Immunol 2:748–759
Chinetti G, Fruchart JC, Staels B (2000) Peroxisome proliferator-activated receptors (PPARs): nuclear receptors at the crossroads between lipid metabolism and inflammation. Inflamm Res 49:497–505
Torra IP, Chinetti G, Duval C, Fruchart JC, Staels B (2001) Peroxisome proliferator-activated receptors: from transcriptional control to clinical practice. Curr Opin Lipidol 12:245–254
Michalik L, Desvergne B, Wahli W (2004) Peroxisome-proliferator-activated receptors and cancers: complex stories. Nat Rev Cancer 4:61–70
Gross B, Staels B (2007) PPAR agonists: multimodal drugs for the treatment of type-2 diabetes. Best Pract Res Clin Endocrinol Metab 21:687–710
Reilly SM, Lee CH (2008) PPAR delta as a therapeutic target in metabolic disease. FEBS Lett 582:26–31
Gervois P, Fruchart JC, Staels B (2007) Drug Insight: mechanisms of action and therapeutic applications for agonists of peroxisome proliferator-activated receptors. Nat Clin Pract Endocrinol Metab 3:145–156
Friedmann PS, Cooper HL, Healy E (2005) Peroxisome proliferator-activated receptors and their relevance to dermatology. Acta Derm Venereol 85:194–202
Straus DS, Glass CK (2007) Anti-inflammatory actions of PPAR ligands: new insights on cellular and molecular mechanisms. Trends Immunol 28:551–558
Xin X, Yang S, Kowalski J, Gerritsen ME (1999) Peroxisome proliferator-activated receptor gamma ligands are potent inhibitors of angiogenesis in vitro and in vivo. J Biol Chem 274:9116–9121
Huang W, Andras IE, Rha GB, Hennig B, Toborek M (2011) PPARα and PPARγ protect against HIV-1-induced MMP-9 overexpression via caveolae-associated ERK and Akt signaling. FASEB J 25(11):3979–3988
Park BC, Thapa D, Lee JS, Park SY, Kim JA (2009) Troglitazone inhibits vascular endothelial growth factor-induced angiogenic signaling via suppression of reactive oxygen species production and extracellular signal-regulated kinase phosphorylation in endothelial cells. J Pharmacol Sci 111:1–12
Mahmood DF, Jguirim-Souissi I, Khadija E, Blondeau N, Diderot V, Amrani S, Slimane MN, Syrovets T, Simmet T, Rouis M (2011) Peroxisome proliferator-activated receptor gamma induces apoptosis and inhibits autophagy of human monocyte-derived macrophages via induction of cathepsin L: potential role in atherosclerosis. J Biol Chem 286:28858–28866
Nakken B, Varga T, Szatmari I, Szeles L, Gyongyosi A, Illarionov PA, Dezso B, Gogolak P, Rajnavolgyi E, Nagy L (2011) Peroxisome proliferator-activated receptor gamma-regulated cathepsin D is required for lipid antigen presentation by dendritic cells. J Immunol 187:240–247
Banfi C, Auwerx J, Poma F, Tremoli E, Mussoni L (2003) Induction of plasminogen activator inhibitor I by the PPARalpha ligand, Wy-14,643, is dependent on ERK1/2 signaling pathway. Thromb Haemost 90(4):611–619
Liangpunsakul S, Wou SE, Wineinger KD, Zeng Y, Cyganek I, Jayaram HN, Crabb DW (2009) Effects of WY-14,643 on the phosphorylation and activation of AMP-dependent protein kinase. Arch Biochem Biophys 485(1):10–15
Conus S, Simon HU (2010) Cathepsins and their involvement in immune responses. Swiss Med Wkly 140:w13042
Turk V, Turk B, Turk D (2001) Lysosomal cysteine proteases: facts and opportunities. EMBO J 20:4629–4633
Saftig P, Hunziker E, Wehmeyer O, Jones S, Boyde A, Rommerskirch W, Moritz JD, Schu P, Von FK (1998) Impaired osteoclastic bone resorption leads to osteopetrosis in cathepsin-K-deficient mice. Proc Natl Acad Sci USA 95:13453–13458
Shi GP, Villadangos JA, Dranoff G, Small C, Gu L, Haley KJ, Riese R, Ploegh HL, Chapman HA (1999) Cathepsin S required for normal MHC class II peptide loading and germinal center development. Immunity 10:197–206
Mohamed MM, Sloane BF (2006) Cysteine cathepsins: multifunctional enzymes in cancer. Nat Rev Cancer 6:764–775
Roth W, Deussing J, Botchkarev VA, Pauly-Evers M, Saftig P, Hafner A, Schmidt P, Schmahl W, Scherer J, Anton-Lamprecht I, Von FK, Paus R, Peters C (2000) Cathepsin L deficiency as molecular defect of furless: hyperproliferation of keratinocytes and pertubation of hair follicle cycling. FASEB J 14:2075–2086
Joyce JA, Baruch A, Chehade K, Meyer-Morse N, Giraudo E, Tsai FY, Greenbaum DC, Hager JH, Bogyo M, Hanahan D (2004) Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis. Cancer Cell 5:443–453
Wang B, Sun J, Kitamoto S, Yang M, Grubb A, Chapman HA, Kalluri R, Shi GP (2006) Cathepsin S controls angiogenesis and tumor growth via matrix-derived angiogenic factors. J Biol Chem 281:6020–6029
Zavasnik-Bergant T, Turk B (2007) Cysteine proteases: destruction ability versus immunomodulation capacity in immune cells. Biol Chem 388:1141–1149
Conus S, Simon HU (2008) Cathepsins: key modulators of cell death and inflammatory responses. Biochem Pharmacol 76:1374–1382
Cavallo-Medved D, Mai J, Dosescu J, Sameni M, Sloane BF (2005) Caveolin-1 mediates the expression and localization of cathepsin B, pro-urokinase plasminogen activator and their cell-surface receptors in human colorectal carcinoma cells. J Cell Sci 118:1493–1503
Jane DT, Morvay L, Dasilva L, Cavallo-Medved D, Sloane BF, Dufresne MJ (2006) Cathepsin B localizes to plasma membrane caveolae of differentiating myoblasts and is secreted in an active form at physiological pH. Biol Chem 387:223–234
Werle B, Kraft C, Lah TT, Kos J, Schanzenbacher U, Kayser K, Ebert W, Spiess E (2000) Cathepsin B in infiltrated lymph nodes is of prognostic significance for patients with nonsmall cell lung carcinoma. Cancer 89:2282–2291
Iacobuzio-Donahue CA, Shuja S, Cai J, Peng P, Murnane MJ (1997) Elevations in cathepsin B protein content and enzyme activity occur independently of glycosylation during colorectal tumor progression. J Biol Chem 272:29190–29199
Sinha AA, Quast BJ, Wilson MJ, Reddy PK, Gleason DF, Sloane BF (1998) Codistribution of procathepsin B and mature cathepsin B forms in human prostate tumors detected by confocal and immunofluorescence microscopy. Anat Rec 252:281–289
Foekens JA, Kos J, Peters HA, Krasovec M, Look MP, Cimerman N, Meijer-van Gelder ME, Henzen-Logmans SC, van Putten WL, Klijn JG (1998) Prognostic significance of cathepsins B and L in primary human breast cancer. J Clin Oncol 16:1013–1021
Krueger S, Haeckel C, Buehling F, Roessner A (1999) Inhibitory effects of antisense cathepsin B cDNA transfection on invasion and motility in a human osteosarcoma cell line. Cancer Res 59:6010–6014
Mohanam S, Jasti SL, Kondraganti SR, Chandrasekar N, Lakka SS, Kin Y, Fuller GN, Yung AW, Kyritsis AP, Dinh DH, Olivero WC, Gujrati M, Ali-Osman F, Rao JS (2001) Down-regulation of cathepsin B expression impairs the invasive and tumorigenic potential of human glioblastoma cells. Oncogene 20:3665–3673
Yanamandra N, Gumidyala KV, Waldron KG, Gujrati M, Olivero WC, Dinh DH, Rao JS, Mohanam S (2004) Blockade of cathepsin B expression in human glioblastoma cells is associated with suppression of angiogenesis. Oncogene 23:2224–2230
Strojnik T, Kos J, Zidanik B, Golouh R, Lah T (1999) Cathepsin B immunohistochemical staining in tumor and endothelial cells is a new prognostic factor for survival in patients with brain tumors. Clin Cancer Res 5:559–567
Premzl A, Turk V, Kos J (2006) Intracellular proteolytic activity of cathepsin B is associated with capillary-like tube formation by endothelial cells in vitro. J Cell Biochem 97:1230–1240
Hashimoto Y, Kakegawa H, Narita Y, Hachiya Y, Hayakawa T, Kos J, Turk V, Katunuma N (2001) Significance of cathepsin B accumulation in synovial fluid of rheumatoid arthritis. Biochem Biophys Res Commun 283:334–339
Baici A, Horler D, Lang A, Merlin C, Kissling R (1995) Cathepsin B in osteoarthritis: zonal variation of enzyme activity in human femoral head cartilage. Ann Rheum Dis 54:281–288
Cataldo AM, Nixon RA (1990) Enzymatically active lysosomal proteases are associated with amyloid deposits in Alzheimer brain. Proc Natl Acad Sci USA 87:3861–3865
Bever CT Jr, Garver DW (1995) Increased cathepsin B activity in multiple sclerosis brain. J Neurol Sci 131:71–73
Gelb BD, Shi GP, Chapman HA, Desnick RJ (1996) Pycnodysostosis, a lysosomal disease caused by cathepsin K deficiency. Science 273:1236–1238
Gocheva V, Joyce JA (2007) Cysteine cathepsins and the cutting edge of cancer invasion. Cell Cycle 6:60–64
Meissner M, Pinter A, Michailidou D, Hrgovic I, Kaprolat N, Stein M, Holtmeier W, Kaufmann R, Gille J (2008) Microtubule-targeted drugs inhibit VEGF receptor-2 expression by both transcriptional and post-transcriptional mechanisms. J Invest Dermatol 128:2084–2091
Meissner M, Reichert TE, Kunkel M, Gooding W, Whiteside TL, Ferrone S, Seliger B (2005) Defects in the human leukocyte antigen class I antigen processing machinery in head and neck squamous cell carcinoma: association with clinical outcome. Clin Cancer Res 11:2552–2560
Berardi S, Lang A, Kostoulas G, Horler D, Vilei EM, Baici A (2001) Alternative messenger RNA splicing and enzyme forms of cathepsin B in human osteoarthritic cartilage and cultured chondrocytes. Arthritis Rheum 44:1819–1831
Yan S, Berquin IM, Troen BR, Sloane BF (2000) Transcription of human cathepsin B is mediated by Sp1 and Ets family factors in glioma. DNA Cell Biol 19(2):79–91
Urbich C, Stein M, Reisinger K, Kaufmann R, Dimmeler S, Gille J (2003) Fluid shear stress-induced transcriptional activation of the vascular endothelial growth factor receptor-2 gene requires Sp1-dependent DNA binding. FEBS Lett 535:87–93
Meissner M, Michailidou D, Stein M, Hrgovic I, Kaufmann R, Gille J (2009) Inhibition of Rac1 GTPase downregulates vascular endothelial growth factor receptor-2 expression by suppressing Sp1-dependent DNA binding in human endothelial cells. Exp Dermatol 18:863–869
Yan S, Jane DT, Dufresne MJ, Sloane BF (2003) Transcription of cathepsin B in glioma cells: regulation by an E-box adjacent to the transcription initiation site. Biol Chem 384:1421–1427
Ren S, Xin C, Beck KF et al (2005) PPARalpha activation upregulates nephrin expression in human embryonic kidney epithelial cells and podocytes by a dual mechanism. Biochem Biophys Res Commun 338(4):1818–1824
Jana M, Jana A, Liu X et al (2007) Involvement of phosphatidylinositol 3-kinase-mediated up-regulation of I kappa B alpha in anti-inflammatory effect of gemfibrozil in microglia. J Immunol 179(6):4142–4152
Cabezas F, Lagos J, Céspedes C et al (2011) Megalin/LRP2 expression is induced by peroxisome proliferator-activated receptor -alpha and -gamma: implications for PPARs’ roles in renal function. PLoS ONE 6(2):e16794
Premzl A, Zavasnik-Bergant V, Turk V, Kos J (2003) Intracellular and extracellular cathepsin B facilitate invasion of MCF-10A neoT cells through reconstituted extracellular matrix in vitro. Exp Cell Res 283:206–214
Kim S, Huang W, Mottillo EP, Sohail A, Ham YA, Conley-Lacomb MK, Kim CJ, Tzivion G, Kim HR, Wang S, Chen YQ, Fridman R (2010) Posttranslational regulation of membrane type 1-matrix metalloproteinase (MT1-MMP) in mouse PTEN null prostate cancer cells: enhanced surface expression and differential O-glycosylation of MT1-MMP. Biochim Biophys Acta 1803:1287–1297
Baici A, Muntener K, Willimann A, Zwicky R (2006) Regulation of human cathepsin B by alternative mRNA splicing: homeostasis, fatal errors and cell death. Biol Chem 387:1017–1021
Meissner M, Stein M, Urbich C, Reisinger K, Suske G, Staels B, Kaufmann R, Gille J (2004) PPARalpha activators inhibit vascular endothelial growth factor receptor-2 expression by repressing Sp1-dependent DNA binding and transactivation. Circ Res 94:324–332
Marx N, Kehrle B, Kohlhammer K, Grub M, Koenig W, Hombach V, Libby P, Plutzky J (2002) PPAR activators as antiinflammatory mediators in human T lymphocytes: implications for atherosclerosis and transplantation-associated arteriosclerosis. Circ Res 90:703–710
Sawai H, Liu J, Reber HA, Hines OJ, Eibl G (2006) Activation of peroxisome proliferator-activated receptor-gamma decreases pancreatic cancer cell invasion through modulation of the plasminogen activator system. Mol Cancer Res 4:159–167
Bility MT, Devlin-Durante MK, Blazanin N, Glick AB, Ward JM, Kang BH, Kennett MJ, Gonzalez FJ, Peters JM (2008) Ligand activation of peroxisome proliferator-activated receptor beta/delta (PPAR beta/delta) inhibits chemically induced skin tumorigenesis. Carcinogenesis 29:2406–2414
Bility MT, Zhu B, Kang BH, Gonzalez FJ, Peters JM (2010) Ligand activation of peroxisome proliferator-activated receptor-beta/delta and inhibition of cyclooxygenase-2 enhances inhibition of skin tumorigenesis. Toxicol Sci 113:27–36
Cavallo-Medved D, Rudy D, Blum G, Bogyo M, Caglic D, Sloane BF (2009) Live-cell imaging demonstrates extracellular matrix degradation in association with active cathepsin B in caveolae of endothelial cells during tube formation. Exp Cell Res 315:1234–1246
Araki H, Tamada Y, Imoto S, Dunmore B, Sanders D, Humphrey S, Nagasaki M, Doi A, Nakanishi Y, Yasuda K, Tomiyasu Y, Tashiro K, Print C, Charnock-Jones DS, Kuhara S, Miyano S (2009) Analysis of PPARalpha-dependent and PPARalpha-independent transcript regulation following fenofibrate treatment of human endothelial cells. Angiogenesis 12:221–229
Palakurthi SS, Aktas H, Grubissich LM, Mortensen RM, Halperin JA (2001) Anticancer effects of thiazolidinediones are independent of peroxisome proliferator-activated receptor gamma and mediated by inhibition of translation initiation. Cancer Res 61:6213–6218
Yamashita M (2008) PPARalpha/gamma-independent effects of PPARalpha/gamma ligands on cysteinyl leukotriene production in mast cells. PPAR Res 2008:293538
Kim J, Ahn JH, Kim JH, Yu YS, Kim HS, Ha J, Shinn SH, Oh YS (2007) Fenofibrate regulates retinal endothelial cell survival through the AMPK signal transduction pathway. Exp Eye Res 84:886–893
Guo D, Sarkar J, Suino-Powell K, Xu Y, Matsumoto K, Jia Y, Yu S, Khare S, Haldar K, Rao MS, Foreman JE, Monga SP, Peters JM, Xu HE, Reddy JK (2007) Induction of nuclear translocation of constitutive androstane receptor by peroxisome proliferator-activated receptor alpha synthetic ligands in mouse liver. J Biol Chem 282(50):36766–36776
Kim DJ, Murray IA, Burns AM, Gonzalez FJ, Perdew GH, Peters JM (2005) Peroxisome proliferator-activated receptor-beta/delta inhibits epidermal cell proliferation by down-regulation of kinase activity. J Biol Chem 280:9519–9527
Pozzi A, Ibanez MR, Gatica AE, Yang S, Wei S, Mei S, Falck JR, Capdevila JH (2007) Peroxisomal proliferator-activated receptor-alpha-dependent inhibition of endothelial cell proliferation and tumorigenesis. J Biol Chem 282:17685–17695
Madej A, Okopien B, Kowalski J, Zielinski M, Wysocki J, Szygula B, Kalina Z, Herman ZS (2007) Effects of fenofibrate on plasma cytokine concentrations in patients with atherosclerosis and hyperlipoproteinemia IIb. Int J Clin Pharmacol Ther 36(6):345–349
Staels B, Koenig W, Habib A, Merval R, Lebret M, Torra IP, Delerive P, Fadel A, Chinetti G, Fruchart JC, Najib J, Maclouf J, Tedgui A (1998) Activation of human aortic smooth-muscle cells is inhibited by PPARalpha but not by PPARgamma activators. Nature 393(6687):790–793
Goya K, Sumitani S, Xu X, Kitamura T, Yamamoto H, Kurebayashi S, Saito H, Kouhara H, Kasayama S, Kawase I (2004) Peroxisome proliferator-activated receptor alpha agonists increase nitric oxide synthase expression in vascular endothelial cells. Arterioscler Thromb Vasc Biol 24(4):658–663
Thuillier P, Anchiraico GJ, Nickel KP, Maldve RE, Gimenez-Conti I, Muga SJ, Liu KL, Fischer SM, Belury MA (2000) Activators of peroxisome proliferator-activated receptor-alpha partially inhibit mouse skin tumor promotion. Mol Carcinog 29(3):134–142
Panigrahy D, Kaipainen A, Huang S, Butterfield CE, Barnés CM, Fannon M, Laforme AM, Chaponis DM, Folkman J, Kieran MW (2008) PPARalpha agonist fenofibrate suppresses tumor growth through direct and indirect angiogenesis inhibition. Proc Natl Acad Sci USA 105(3):985–990
Acknowledgments
The study was supported by the Wilhelm Sander Foundation (MM) and the Berlin Foundation for Dermatology (MM) and by a grant from the University of Frankfurt (GR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reichenbach, G., Starzinski-Powitz, A., Sloane, B.F. et al. PPARα agonist Wy14643 suppresses cathepsin B in human endothelial cells via transcriptional, post-transcriptional and post-translational mechanisms. Angiogenesis 16, 223–233 (2013). https://doi.org/10.1007/s10456-012-9314-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-012-9314-9