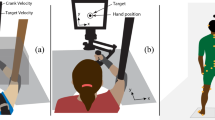
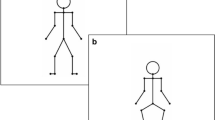
Abstract
Our objective was to investigate how manipulating sensory input through mastoid vibration (MV) could affect dynamic postural control during walking, with and without simultaneous manipulation of the visual and the somatosensory systems. We used three levels of MV (none, unilateral, and bilateral) via vibrating elements placed on the mastoid processes. We combined this with the six conditions of the Locomotor Sensory Organization Test (LSOT) paradigm to challenge the visual and somatosensory systems. We hypothesized that MV would affect both amount and temporal structure measures of sway variability during walking and that, in combination with manipulations of the visual and the somatosensory inputs, MV would augment the effects previously observed. The results confirmed that MV produced a significant increase in the amount of sway variability in both anterior–posterior and medial–lateral directions. Significant changes in the temporal structure of sway variability were only observed in the anterior–posterior direction. Bilateral MV produced larger effects than unilateral stimulation. We concluded that sensory input while walking could be affected using MV. Combining MV with manipulations of visual and somatosensory input could allow us to better understand the contributions of the sensory systems during locomotion.





Similar content being viewed by others
Abbreviations
- MV:
-
Mastoid vibration
- LSOT:
-
Locomotor Sensory Organization Test
- netCOP:
-
NET Center of Pressure
- SampEn:
-
Sample Entropy
References
Agrawal, Y., J. P. Carey, C. C. Della Santina, M. C. Schubert, and L. B. Minor. Disorders of balance and vestibular function in US adults. Arch. Intern. Med. 169(10):938–944, 2009.
Biguer, B., I. M. Donaldson, A. Hein, and M. Jeannerod. Neck muscle vibration modifies the representation of visual motion and direction in man. Brain 111:1405–1424, 1988.
Bottini, G., H. O. Karnath, G. Vallar, R. Sterzi, C. D. Frith, R. S. Frackowiak, and E. Paulesu. Cerebral representation for egocentric space: functional-anatomical evidence from caloric vestibular stimulation and neck vibration. Brain 124:1182–1196, 2001.
Bove, M., G. Brichetto, G. Abbruzzese, R. Marchese, and M. Schieppati. Neck proprioception and spatial orientation in cervical dystonia. Brain 127:2764–2778, 2004.
Bove, M., G. Courtine, and M. Schieppati. Neck muscle vibration and spatial orientation during stepping in place in humans. J. Neurophysiol. 88(5):2232–2241, 2002.
Bove, M., M. Diverio, T. Pozzo, and M. Schieppati. Neck muscle vibration disrupts steering of locomotion. J. Appl. Physiol. (1985) 91(2):581–588, 2001.
Brandt, T. Cervical vertigo—reality or fiction? Audiol Neurootol 1:187–196, 1996.
Brandt, T., and A. M. Bronstein. Cervical vertigo. J. Neurol. Neurosurg. Psychiatry 71(2):8–12, 2001.
Brandt, T., J. Dichgans, and W. Buchle. Motion habituation: inverted self-motion perception and optokinetic after-nystagmus. Exp. Brain Res. 21(4):337–352, 1974.
Brandt, T., M. Strupp, J. Benson, and M. Dieterich. Vestibulopathic gait. Walking and running. Adv. Neurol. 87:165–172, 2001.
Chan, Y. S., J. Kasper, and V. J. Wilson. Dynamics and directional sensitivity of neck muscle spindle responses to head rotation. J. Neurophysiol. 57:1716–1729, 1987.
Chien, J. H., D. J. Eikema, M. Mukherjee, and N. Stergiou. Locomotor sensory organization test: a novel paradigm for the assessment of sensory contributions in gait. Ann. Biomed. Eng. 42(12):2512–2523, 2014.
Chien, J. H., M. Mukherjee, K. C. Siu, and N. Stergiou. Locomotor sensory organization test: how sensory conflict affects the temporal structure of sway variability during gait. Ann. Biomed. Eng. 2015. doi:10.1007/s10439-015-1440-2.
Cohen, H. S. Vestibular disorders and impaired path integration along a linear trajectory. J. Vestib. Res. 10(1):7–15, 2000.
Deshpande, N., L. Ferrucci, J. Metter, K. A. Faulkner, E. Strotmeyer, S. Satterfield, A. Schwartz, and E. Simonsick. Association of lower limb cutaneous sensitivity with gait speed in the elderly: the health ABC study. Am. J. Phys. Med. Rehabil. 87(11):921–928, 2008.
Deshpande, N., E. J. Metter, and L. Ferrucci. Validity of clinically derived cumulative somatosensory impairment index. Arch. Phys. Med. Rehabil. 91(2):226–232, 2010.
Deshpande, N., and A. E. Patla. Postural responses and spatial orientation to neck proprioceptive and vestibular inputs during locomotion in young and older adults. Exp. Brain Res. 167(3):468–474, 2005.
Dilda, V., H. G. MacDougall, and S. T. Moore. Tolerance to extended galvanic vestibular stimulation: optimal exposure for astronaut training. Aviat. Space Environ. Med. 82(8):770–774, 2011.
Ernst, M. O., and M. S. Banks. Humans integrate visual and haptic information in a statistically optimal fashion. Nature 415(6870):429–433, 2002.
Fetsch, C. R., A. H. Turner, G. C. DeAngelis, and D. E. Angelaki. Dynamic reweighting of visual and vestibular cues during self-motion perception. J. Neurosci. 29(49):15601–15612, 2009.
Fife, T. D., R. J. Tusa, J. M. Furman, D. S. Zee, E. Frohman, R. W. Baloh, T. Hain, J. Goebel, J. Demer, and L. Eviatar. Assessment: vestibular testing techniques in adults and children: report of the Therapeutic and Technology Assessment Subcommittee of American Academy Neurology. Neurology 55(10):1431–1441, 2000.
Fitzpatrick, R. C., J. E. Butler, and B. L. Day. Resolving head rotation for human bipedalism. Curr. Biol. 16(15):1509–1514, 2006.
Fitzpatrick, R. C., D. L. Wardman, and J. L. Taylor. Effects of galvanic vestibular stimulation during human walking. J. Physiol. 517:931–939, 1999.
Fransson, P. A., M. Karlberg, T. Sterner, and M. Maqnusson. Direction of galvanically-induced vestibulo-postural responses during active and passive neck torsion. Acta Otolaryngol. 120(4):500–503, 2000.
Goldberg, J. M., C. E. Smith, and C. Fernandez. Relation between discharge regularity and response to externally applied galvanic currents in vestibular nerve afferents of the squirrel monkey. J. Neurophysiol. 51(6):1236–1256, 1984.
Gordon, C. R., D. Tal, N. Gadoth, and A. Shupak. Prolonged optokinetic stimulation generates podokinetic after rotation. Ann. N. Y. Acad. Sci. 297–302:2003, 1004.
Ivanenko, Y. P., R. Grasso, and F. Lacquaniti. Neck muscle vibration makes walking humans accelerate in the direction gaze. J. Physiol. 525(pt 3):803–814, 2000.
Jacobson, G. P., and C. W. Newman. The development of the Dizziness Handicap Inventory. Arch Otolanryngol Head Surg 116:424–427, 1990.
Jahn, K., M. Strupp, E. Schneider, M. Dieterich, and T. Brandt. Differential effects of vestibular stimulation on walking and running. NeuroReport 11(8):1745–1748, 2000.
Jordan, K., J. H. Challis, and K. M. Newell. Walking speed influences on gait cycle variability. Gait Posture. 26(1):128–134, 2007.
Jordan, K., and K. M. Newell. The structure of variability in human walking and running is speed-dependent. Exerc. Sport Sci. Rev. 36(4):200–204, 2008.
Karlberg, M., S. Aw, R. Black, M. Todd, H. MacDougall, and M. Halamgyi. Vibration-induced ocular torsion and nystagmus after unilateral vestibular deafferentation. Brain 126:956–964, 2003.
Kavounoudias, A., J. C. Gilhodes, R. Roll, and J. P. Roll. From balance regulation to body orientation: two goals for muscle proprioceptive information processing? Exp. Brain Res. 124(1):80–88, 1999.
Kubo, T., H. Kumakura, Y. Hirokawa, K. Yamamoto, and E. Hirasaki. 3D analysis of human locomotion before and after caloric stimulation. Acta Otolaryngol. 117(2):143–148, 1997.
Lekhel, H., K. Popov, A. Bronstein, and M. Gresty. Postural reponses to vibration of neck muscles in patients with uni- and bilateral vestibular loss. Gait Posture 7:228–236, 1998.
Mawase, F., T. Haizler, S. Bar-Haim, and A. Karniel. Kinetic adaptation during locomotion on a split-belt treadmill. J. Neurophysiol. 109:2216–2227, 2013.
Neuhauser, H. K., A. Radtke, and M. von Brevern. Burden of dizziness and vertigo in the community. Arch. Intern. Med. 168(19):2118–2124, 2008.
Nuti, D., and M. Mandala. Sensitivity and specificity of mastoid vibration test in detection of effects of vestibular neuritis. Acta Otorhinolarynqol Ital. 25(5):271–276, 2005.
Pettorossi, V. E., and M. Schieppati. Neck proprioception shapes body orientation and perception of motion. Front Hum Neurosci. 8:895, 2014.
Proctor, L. R. Clinical experience with a short-acting caloric test. Laryngoscope 95(1):75–80, 1985.
Souman, J. L., I. Frissen, M. N. Sreenivasa, and M. O. Ernst. Walking straight into circles. Curr. Biol. 19(18):1538–1542, 2009.
St George, R. J., R. C. Fitzpatrick, and RJ. The sense of self-motion, orientation and balance explored by vestibular stimulation. J. Physiol. 589(pt 4):807–813, 2011.
Strupp, M., V. Arbusow, M. Dieterich, and T. Brandt. Perceptual and oculomotor effects of neck muscle vibration in vestibular neuritis: ipsilateral somatosensory substitution of vestibular function. Brain 121:677–685, 1998.
Wurdeman, S. R., N. B. Huben, and N. Stergiou. Variability of gait is dependent on direction of progression: implication of active control. J. Biomech. 45(4):653–659, 2012.
Wurdeman, S. R., and N. Stergiou. Temporal structure of variability reveals similar control mechanisms during lateral stepping and forward walking. Gait Posture 38(1):73–78, 2013.
Yentes, J. M., N. Hunt, K. K. Schmid, J. P. Kaipust, and D. McGrath. Stergiou N. The appropriate use of approximate entropy and sample entropy with short data sets. Ann. Biomed. Eng. 41(2):349–365, 2013.
Acknowledgments
This work was supported by the Center for Research in Human Movement Variability of the University of Nebraska Omaha and the NIH (P20GM109090 and R01AG034995). Additional support was provided by NASA EPSCoR NNX11AM06A.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Thurmon E. Lockhart oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Chien, J.H., Mukherjee, M. & Stergiou, N. Mastoid Vibration Affects Dynamic Postural Control During Gait. Ann Biomed Eng 44, 2774–2784 (2016). https://doi.org/10.1007/s10439-016-1556-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-016-1556-z