Abstract
Background
Neuroendocrine carcinoma (NEC) of the esophagus is a rare histologic type that is aggressive and grows rapidly. Immunohistochemistry with conventional antibodies, such as synaptophysin, chromogranin A, and CD56, is widely performed. Several novel neuroendocrine markers, such as mammalian achaete-scute homolog 1 (mASH1), NeuroD1, and PGP9.5, were recently proposed.
Methods
From 1115 consecutive cases with esophageal cancer, 10 NEC cases (0.9 %) were enrolled and the clinicopathologic parameters, including prognosis, were compared with those of 32 squamous cell carcinomas (SCC) and 30 basaloid squamous carcinomas (BSC). Immunohistochemistry for mASH1, NeuroD1, PGP9.5 and for conventional antibodies was performed. Both monoclonal and polyclonal antibodies for NeuroD1 and PGP9.5 were evaluated.
Results
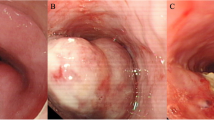
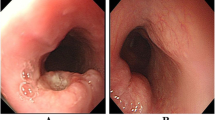
Six NEC cases were diagnosed as T1b (submucosal invasion), four of which were endoscopically resected. The remaining four cases with T3 or T4 died of the disease within 37 months. Of the six patients with T1b, three are alive with no evidence of disease for more than 50 months, although the prognosis is worse for NEC than for SCC and BSC. The mASH1, NeuroD1-monoclonal, NeuroD1-polyclonal, PGP9.5-monoclonal, and PGP9.5-polyclonal immunohistochemical markers were positive in 8, 8, 1, 7, and 6 NEC cases, respectively. The number of mASH1-positive cases and synaptophysin-positive cases was the same. mASH1 had good concordance with chromogranin A and CD56, and was superior to other novel antibodies. mASH1 had excellent specificity and sensitivity for NEC.
Conclusion
Esophageal NEC has a worse prognosis than SCC and BSC, even in T1b patients. mASH1 is an excellent novel marker for diagnosing esophageal NEC.





Similar content being viewed by others
References
Japan Esophageal Society. Comprehensive registry of esophageal cancer in Japan (1995, 1996, 1997). 2nd ed. 2001. http://esophagus.jp/crec.html. Accessed 7 Jan 2014.
Japan Esophageal Society. Comprehensive registry of esophageal cancer in Japan (1998, 1999) and long-term results of esophagectomy in Japan (1988–1997). 3rd ed. 2002. http://esophagus.jp/crec.html. Accessed 7 Jan 2014.
Ide H. Macroscopic findings of non-squamous cell carcinoma of the esophagus. Stomach Intest. 2005;40:279–87 (in Japanese with English Abstract).
Bosman FT, Carneiro F, Hruban RH, Theise ND, editors. WHO classification of tumours of the digestive system. Geneva: WHO Press; 2010.
Yamamoto J, Ohshima K, Ikeda S, Iwashita A, Kikuchi M. Primary esophageal small cell carcinoma with concomitant invasive squamous cell carcinoma or carcinoma in situ. Hum Pathol. 2003;34:1108–15.
Yun JP, Zhang MF, Hou JH, Tian QH, Fu J, Liang XM, et al. Primary small cell carcinoma of the esophagus: clinicopathological and immunohistochemical features of 21 cases. BMC Cancer. 2007;7:38.
Kuo CH, Hsieh CC, Chan ML, Li AF, Huang MH, Hsu WH, et al. Small cell carcinoma of the esophagus: a report of 16 cases from a single institution and literature review. Ann Thorac Surg. 2011;2:373–8.
Lu J, Xue LY, Lu N, Zou SM, Liu XY, Wen P. Superficial primary small cell carcinoma of the esophagus: clinicopathological and immunohistochemical analysis of 15 cases. Dis Esophagus. 2010;23:153–9.
Noguchi T, Takeno S, Kato T, Wada S, Noguchi T, Uchida Y, et al. Small cell carcinoma of the esophagus; clinicopathological and immunohistochemical analysis of six cases. Dis Esophagus. 2003;16:252–8.
Japan Esophageal Society. Japanese classification of esophageal cancer, tenth edition: parts II and III. Esophagus. 2009;6:71–94.
Hudson E, Powell J, Mukherjee S, Crosby TD, Brewster AE, Maughan TS, et al. Small cell oesophageal carcinoma: an institutional experience and review of the literature. Br J Cancer. 2007;96:708–11.
Ku GY, Minsky BD, Rusch VW, Bains M, Kelsen DP, Ilson DH. Small-cell carcinoma of the esophagus and gastroesophageal junction: review of the Memorial Sloan-Kettering experience. Ann Oncol. 2008;19:533–7.
Chin K, Baba S, Hosaka H, Ishiyama A, Mizunuma N, Shinozaki E, et al. Irinotecan plus cisplatin for therapy of small-cell carcinoma of the esophagus: report of 12 cases from single institution experience. Jpn J Clin Oncol. 2008;38:426–31.
Serrano MF, El-Mofty SK, Gnepp DR, Lewis JS Jr. Utility of high molecular weight cytokeratins, but not p63, in the differential diagnosis of neuroendocrine and basaloid carcinomas of the head and neck. Hum Pathol. 2008;39:591–8.
Maru DM, Khurana H, Rashid A, Correa AM, Anandasabapathy S, Krishnan S, et al. Retrospective study of clinicopathologic features and prognosis of high-grade neuroendocrine carcinoma of the esophagus. Am J Surg Pathol. 2008;32:1404–11.
Li AF, Li AC, Hsu CY, Li WY, Hsu HS, Chen JY. Small cell carcinomas in gastrointestinal tract: immunohistochemical and clinicopathological features. J Clin Pathol. 2010;63:620–5.
Shida T, Furuya M, Nikaido T, Kishimoto T, Koda K, Oda K, et al. Aberrant expression of human achaete-scute homologue gene 1 in the gastrointestinal neuroendocrine carcinomas. Clin Cancer Res. 2005;11:450–8.
Rapa I, Ceppi P, Bollito E, Rosas R, Cappia S, Bacillo E, et al. Human ASH1 expression in prostate cancer with neuroendocrine differentiation. Mod Pathol. 2008;21:700–7.
Shida T, Furuya M, Kishimoto T, Nikaido T, Tanizawa T, Koda K, et al. The expression of NeuroD and mASH1 in the gastroenteropancreatic neuroendocrine tumors. Mod Pathol. 2008;21:1363–70.
Fujii A, Kamiakito T, Takayashiki N, Fujii T, Tanaka A. Neuroendocrine tissue-specific transcription factor, BETA2/NeuroD, in gastric carcinomas: a comparison with chromogranin A and synaptophysin expressions. Pathol Res Pract. 2003;199:513–9.
Sasaki H, Yukiue H, Moriyama S, Kobayashi Y, Nakashima Y, Kaji M, et al. Expression of the protein gene product 9.5, PGP9.5, is correlated with T-status in non-small cell lung cancer. Jpn J Clin Oncol. 2001;31:532–5.
Takase T, Hibi K, Yamazaki T, Nakayama H, Taguchi M, Kasai Y, et al. PGP9.5 overexpression in esophageal squamous cell carcinoma. Hepatogastroenterology. 2003;50:1278–80.
Arai T, Aida J, Nakamura K, Ushio Y, Takubo K. Clinicopathologic characteristics of basaloid squamous carcinoma of the esophagus. Esophagus. 2011;8:169–77.
Lv J, Liang J, Wang J, Wang L, He J, Xiao Z, et al. Primary small cell carcinoma of the esophagus. J Thorac Oncol. 2008;3:1460–5.
Takubo K, Nakamura K, Sawabe M, Arai T, Esaki Y, Miyashita M, et al. Primary undifferentiated small cell carcinoma of the esophagus. Hum Pathol. 1999;30:216–21.
Koide N, Saito H, Suzuki A, Sato T, Koiwai K, Nakamura N, et al. Clinicopathologic features and histochemical analyses of proliferative activity and angiogenesis in small cell carcinoma of the esophagus. J Gastroenterol. 2007;42:932–8.
Chen SB, Yang JS, Yang WP, Weng HR, Li H, Liu DT, et al. Treatment and prognosis of limited disease primary small cell carcinoma of esophagus. Dis Esophagus. 2011;24:114–9.
Huang Q, Wu H, Nie L, Shi J, Lebenthal A, Chen J, et al. Primary high-grade neuroendocrine carcinoma of the esophagus: a clinicopathologic and immunohistochemical study of 42 resection cases. Am J Surg Pathol. 2013;37:467–83.
Vos B, Rozema T, Miller RC, Hendlisz A, Van Laethem JL, Khanfir K, et al. Small cell carcinoma of the esophagus: a multicentre Rare Cancer Network study. Dis Esophagus. 2011;24:258–64.
Shia J, Tang LH, Weiser MR, Brenner B, Adsay NV, Stelow EB, et al. Is nonsmall cell type high-grade neuroendocrine carcinoma of the tubular gastrointestinal tract a distinct disease entity? Am J Surg Pathol. 2008;32:719–31.
Marchevsky AM, Gal AA, Shah S, Koss MN. Morphometry confirms the presence of considerable nuclear size overlap between “small cells” and “large cells” in high-grade pulmonary neuroendocrine neoplasms. Am J Clin Pathol. 2001;116:466–72.
Hiroshima K, Iyoda A, Shida T, Shibuya K, Iizasa T, Kishi H, et al. Distinction of pulmonary large cell neuroendocrine carcinoma from small cell lung carcinoma: a morphological, immunohistochemical, and molecular analysis. Mod Pathol. 2006;19:1358–68.
Helpap B, Köllermann J. Immunohistochemical analysis of the proliferative activity of neuroendocrine tumors from various organs. Are there indications for a neuroendocrine tumor-carcinoma sequence? Virchows Arch. 2001;438:86–91.
Howe MC, Chapman A, Kerr K, Dougal M, Anderson H, Hasleton PS. Neuroendocrine differentiation in non-small cell lung cancer and its relation to prognosis and therapy. Histopathology. 2005;46:195–201.
Jiang SX, Kameya T, Asamura H, Umezawa A, Sato Y, Shinada J, et al. hASH1 expression is closely correlated with endocrine phenotype and differentiation extent in pulmonary neuroendocrine tumors. Mod Pathol. 2004;17:222–9.
Oyama K, Sanno N, Teramoto A, Osamura RY. Expression of neuro D1 in human normal pituitaries and pituitary adenomas. Mod Pathol. 2001;14:892–9.
Larsen CN, Krantz BA, Wilkinson KD. Substrate specificity of deubiquitinating enzymes: ubiquitin C-terminal hydrolases. Biochemistry. 1998;37:3358–68.
Hibi K, Westra WH, Borges M, Goodman S, Sidransky D, Jen J. PGP9.5 as a candidate tumor marker for non-small-cell lung cancer. Am J Pathol. 1999;155:711–5.
Campbell LK, Thomas JR, Lamps LW, Smoller BR, Folpe AL. Protein gene product 9.5 (PGP 9.5) is not a specific marker of neural and nerve sheath tumors: an immunohistochemical study of 95 mesenchymal neoplasms. Mod Pathol. 2003;16:963–9.
Wilson PO, Barber PC, Hamid QA, Power BF, Dhillon AP, Rode J, et al. The immunolocalization of protein gene product 9.5 using rabbit polyclonal and mouse monoclonal antibodies. Br J Exp Pathol. 1988;69:91–104.
Tezel E, Hibi K, Nagasaka T, Nakao A. PGP9.5 as a prognostic factor in pancreatic cancer. Clin Cancer Res. 2000;6:4764–7.
Yamazaki T, Hibi K, Takase T, Tezel E, Nakayama H, Kasai Y, et al. PGP9.5 as a marker for invasive colorectal cancer. Clin Cancer Res. 2002;8:192–5.
Acknowledgments
The authors gratefully acknowledge Dr. Toichiro Takizawa, former professor of Tokyo Medical and Dental University, for providing both the opportunity for this study and his sincere encouragement.
Ethical Statement
About the submission of our manuscript titled “Neuroendocrine carcinoma of the esophagus: Clinicopathologic study of 10 cases and verification of the diagnostic utility of mASH1, NeuroD1, and PGP9.5”, all of authors here declare that this study was performed in accordance with the World Medical Association and the Declaration of Helsinki. The ethics committee of Tokyo Medical and Dental University approved the protocols (Registration No. 1026).
Conflict of interest
There are no financial or other relations that could lead to a conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akazawa, N., Kawachi, H., Kitagaki, K. et al. Neuroendocrine carcinoma of the esophagus: clinicopathologic study of 10 cases and verification of the diagnostic utility of mASH1, NeuroD1, and PGP9.5. Esophagus 11, 245–257 (2014). https://doi.org/10.1007/s10388-014-0444-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10388-014-0444-6