Abstract
Purpose
The 2 most common causative genes for achromatopsia (ACHM) are CNGA3 and CNGB3; other genes including GNAT2 account for only a small portion of ACHM cases. The cone mosaics in eyes with CNGA3 and CNGB3 variants are severely disrupted; the cone mosaics in patients with GNAT2-associated ACHM; however, have been reported to show a contiguous pattern in adaptive optics (AO) retinal images. The purpose of this study was to analyze the cone mosaic of another case of GNAT2-associated ACHM.
Patient and methods
The patient was a 17-year-old Japanese boy. Comprehensive ocular examinations including fundus photography, electroretinography (ERGs), optical coherence tomography (OCT), and whole-exome analysis were performed. The cone mosaic was recorded with a flood-illuminated AO fundus camera, and the cone density was compared with those of 10 normal control eyes.
Results
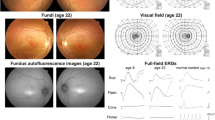
The patient had the typical phenotype of ACHM, and a novel homozygous variant, c.730_743del, in GNAT2 was identified. The fundus did not show any specific abnormalities, and the OCT images showed the presence of the ellipsoid zone. The AO fundus image showed a clearly defined cone mosaic around the fovea. The cone density at 500 μm from the fovea was reduced by 15–30 % as compared with those of the normal eyes.
Conclusions
This is the first description of a Japanese patient with ACHM with a novel GNAT2 variant. The eyes of this patient had a preserved cone structure with loss of function.




Similar content being viewed by others
References
Michaelides M, Aligianis IA, Holder GE, Simunovic MP, Mollon JD, Maher ER, et al. Cone dystrophy phenotype associated with a frameshift mutation (M280fsX291) in the alpha-subunit of cone specific transducin (GNAT2). Br J Ophthalmol. 2003;87:1317–20.
Andreasson S, Tornqvist K. Electroretinograms in patients with achromatopsia. Acta Ophthalmol. 1991;69:711–6.
Sundin OH, Yang JM, Li YY, Zhu DP, Hurd JN, Mitchell TN, et al. Genetic basis of total colourblindness among the Pingelapese islanders. Nat Genet. 2000;25:289–93.
Wissinger B, Jagle H, Kohl S, Broghammer M, Baumann B, Hanna DB, et al. Human rod monochromacy: linkage analysis and mapping of a cone photoreceptor expressed candidate gene on chromosome 2q11. Genomics. 1998;51:325–31.
Johnson S, Michaelides M, Aligianis IA, Ainsworth JR, Mollon JD, Maher ER, et al. Achromatopsia caused by novel mutations in both CNGA3 and CNGB3. J Med Genet. 2004;41:e20.
Kohl S, Varsanyi B, Antunes GA, Baumann B, Hoyng CB, Jagle H, et al. CNGB3 mutations account for 50 % of all cases with autosomal recessive achromatopsia. Eur J Hum Genet. 2005;13:302–8.
Saqib MA, Awan BM, Sarfraz M, Khan MN, Rashid S, Ansar M. Genetic analysis of four Pakistani families with achromatopsia and a novel S4 motif mutation of CNGA3. Jpn J Ophthalmol. 2011;55:676–80.
Kuniyoshi K, Muraki-Oda S, Ueyama H, Toyoda F, Sakuramoto H, Ogita H, et al. Novel mutations in the gene for alpha-subunit of retinal cone cyclic nucleotide-gated channels in a Japanese patient with congenital achromatopsia. Jpn J Ophthalmol. 2016;60:187–97.
Kohl S, Baumann B, Rosenberg T, Kellner U, Lorenz B, Vadala M, et al. Mutations in the cone photoreceptor G-protein alpha-subunit gene GNAT2 in patients with achromatopsia. Am J Hum Genet. 2002;71:422–5.
Kohl S, Coppieters F, Meire F, Schaich S, Roosing S, Brennenstuhl C, et al. A nonsense mutation in PDE6H causes autosomal-recessive incomplete achromatopsia. Am J Hum Genet. 2012;91:527–32.
Thiadens AAHJ, Somervuo V, van den Born LI, Roosing S, van Schooneveld MJ, Kuijpers RWAM, et al. Progressive loss of cones in achromatopsia: an imaging study using spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2010;51:5952–7.
Thomas MG, Kumar A, Kohl S, Proudlock FA, Gottlob I. High-resolution in vivo imaging in achromatopsia. Ophthalmology. 2011;118:882–7.
Sundaram V, Wilde C, Aboshiha J, Cowing J, Han C, Langlo CS, et al. Retinal structure and function in achromatopsia: implications for gene therapy. Ophthalmology. 2014;121:234–45.
Roorda A, Williams DR. The arrangement of the three cone classes in the living human eye. Nature. 1999;397:520–2.
Kitaguchi Y, Bessho K, Yamaguchi T, Nakazawa N, Mihashi T, Fujikado T. In vivo measurements of cone photoreceptor spacing in myopic eyes from images obtained by an adaptive optics fundus camera. Jpn J Ophthalmol. 2007;51:456–61.
Bessho K, Fujikado T, Mihashi T, Yamaguchi T, Nakazawa N, et al. Photoreceptor images of normal eyes and of eyes with macular dystrophy obtained in vivo with an adaptive optics fundus camera. Jpn J Ophthalmol. 2008;52:380–5.
Carroll J, Choi SS, Williams DR. In vivo imaging of the photoreceptor mosaic of a rod monochromat. Vis Res. 2008;48:2564–8.
Scoles D, Sulai YN, Langlo CS, Fishman GA, Curcio CA, Carroll J, et al. In vivo imaging of human cone photoreceptor inner segments. Invest Ophthalmol Vis Sci. 2014;55:4244–51.
Dubis AM, Cooper RF, Aboshiha J, Langlo CS, Sundaram V, Liu B, et al. Genotype-dependent variability in residual cone structure in achromatopsia: toward developing metrics for assessing cone health. Invest Ophthalmol Vis Sci. 2014;55:7303–11.
McCulloch DL, Marmor MF, Brigell MG, Hamilton R, Holder GE, Tzekov R, et al. ISCEV Standard for full-field clinical electroretinography (2015 update). Doc Ophthalmol. 2015;130:1–12.
Nakanishi A, Ueno S, Kawano K, Ito Y, Kominami T, Yasuda S, et al. Pathologic changes of cone photoreceptors in eyes with occult macular dystrophy. Invest Ophthalmol Vis Sci. 2015;56:7243–9.
Ueno S, Kawano K, Ito Y, Ra E, Nakanishi A, Nagaya M, et al. Near-infrared reflectance imaging in eyes with acute zonal occult outer retinopathy. Retina. 2015;35:1521–30.
Katagiri S, Yoshitake K, Akahori M, Hayashi T, Furuno M, Nishino J, et al. Whole-exome sequencing identifies a novel ALMS1 mutation (p. Q2051X) in two Japanese brothers with Alstrom syndrome. Mol Vis. 2013;19:2393–406.
Curcio CA, Sloan KR, Kalina RE, Hendrickson AE. Human photoreceptor topography. J Comp Neurol. 1990;292:497–523.
Lombardo M, Serrao S, Ducoli P, Lombardo G. Variations in image optical quality of the eye and the sampling limit of resolution of the cone mosaic with axial length in young adults. J Cataract Refract Surg. 2012;38:1147–55.
Rosenberg T, Baumann B, Kohl S, Zrenner E, Jorgensen AL, Wissinger B. Variant phenotypes of incomplete achromatopsia in two cousins with GNAT2 gene mutations. Invest Ophthalmol Vis Sci. 2004;45:4256–62.
Ouechtati F, Merdassi A, Bouyacoub Y, Largueche L, Derouiche K, Ouragini H, et al. Clinical and genetic investigation of a large Tunisian family with complete achromatopsia: identification of a new nonsense mutation in GNAT2 gene. J Hum Genet. 2011;56:22–8.
Hasegawa T, Ueda T, Okamoto M, Ogata N. Presence of foveal bulge in optical coherence tomographic images in eyes with macular edema associated with branch retinal vein occlusion. Am J Ophthalmol. 2014;157:390–6.
Bidaut Garnier M, Flores M, Debellemaniere G, Puyraveau M, Tumahai P, Meillat M, et al. Reliability of cone counts using an adaptive optics retinal camera. Clin Exp Ophthalmol. 2014;42:833–40.
Biel M, Seeliger M, Pfeifer A, Kohler K, Gerstner A, Ludwig A. Selective loss of cone function in mice lacking the cyclic nucleotide-gated channel CNG3. Proc Natl Acad Sci USA. 1999;96:7553–7.
Michalakis S, Geiger H, Haverkamp S, Hofmann F, Gerstner A, Biel M. Impaired opsin targeting and cone photoreceptor migration in the retina of mice lacking the cyclic nucleotide-gated channel CNGA3. Invest Ophthalmol Vis Sci. 2005;46:1516–24.
Chang B, Dacey MS, Hawes NL, Hitchcock PF, Milam AH, Atmaca-Sonmez P, et al. Cone photoreceptor function loss-3: a novel mouse model of achromatopsia due to a mutation in Gnat2. Invest Ophthalmol Vis Sci. 2006;47:5017–21.
Acknowledgments
We thank Professor Duco Hamasaki of the Bascom Palmer Eye Institute for discussions and for editing the final version of the manuscript. This work was supported in part by a Scientific Research C Grant (25462709 SU) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
S. Ueno, Grants (Santen), Lecture fees (Novartis, Pfizer); A. Nakanishi, None; T. Kominami, None; Y. Ito, Lecture fees (Aichi Ophthalmologists Association, Alcon, Bayer Health Care, Canon Life Care Solutions, Carl Zeiss Meditec, Kowa, Nidek, Novartis, Okazaki City Medical Association, Pfizer, Santen); T. Hayashi, None; K. Yoshitake, None; Y. Kawamura, None; K. Tsunoda, None; T. Iwata, None; H. Terasaki, Grants (Canon, Nidek), Lecture fees (Carl Zeiss Meditec, Nidek).
About this article
Cite this article
Ueno, S., Nakanishi, A., Kominami, T. et al. In vivo imaging of a cone mosaic in a patient with achromatopsia associated with a GNAT2 variant. Jpn J Ophthalmol 61, 92–98 (2017). https://doi.org/10.1007/s10384-016-0484-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-016-0484-7