Abstract
Purpose
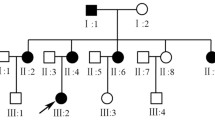
To report three types of heterozygous mutations in the OPA1 gene in five patients from three families with autosomal dominant optic atrophy (ADOA, MIM#165500).
Methods
DNA was extracted from the leukocytes of the peripheral blood. For mtDNA, mutations were examined at positions 11778, 3460 and 14484. For the OPA1 gene, the exons were amplified by PCR and mutations were detected by restriction enzymes or the dye terminator method.
Results
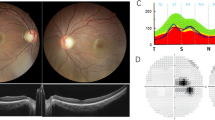
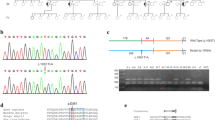
We detected three types of OPA1 mutation but no mtDNA mutations. In the OPA1 gene, heterozygous frameshift mutations from codon 903 due to a four-base pair deletion in exon 27 were detected in three patients from one family (c.2708_2711delTTAG, p.V903GfsX905). A heterozygous mutation due to a three-base pair deletion in exon 17, leading to a one-amino acid deletion (c.1618_1620delACT, p.T540del), and a heterozygous mutation due to a one-base substitution in exon 11, leading to a stop codon (c.1084G>T, p.E362X), were detected in sporadic cases.
Conclusion
OPA1 mutations existed in three Japanese families with ADOA. After a detailed clinical assessment of the proband, the screening of the OPA1 gene may be helpful for precise diagnosis of ADOA, provided the relevant information of the family members is limited.



Similar content being viewed by others
References
Kjer P. Infantile optic atrophy with dominant mode of inheritance: a clinical and genetic study of 19 Danish families. Acta Ophthalmol Scand. 1959;37(suppl 54):1–146.
Johnston PB, Gaster RN, Smith VC, Tripathi RC. A clinicopathological study of autosomal dominant optic atrophy. Am J Ophthalmol. 1979;88:868–75.
Hoyt CS. Autosomal dominant optic atrophy: a spectrum of disability. Ophthalmology. 1980;87:245–51.
Kerrison JB. Hereditary optic neuropathies. Ophthalmol Clin North Am. 2001;14:99–107.
Cohn AC, Toomes C, Potter C, Towns K, Hewitt AW, Inglehearn CF, et al. Autosomal dominant optic atrophy: penetrance and expressivity in patients with OPA1 mutations. Am J Ophthalmol. 2007;143:656–62.
Delettre C, Lenaers G, Griffoin JM, Gigarel N, Lorenzo C, Belenguer P, et al. Nuclear gene OPA1, encoding a mitochondrial dynamin-related protein, is mutated in dominant optic atrophy. Nat Genet. 2000;26:207–10.
Alexander C, Votruba M, Pesch UEA, Thiselton DL, Mayer S, Moore A, et al. OPA1, encoding a dynamin-related GTPase, is mutated in autosomal dominant optic atrophy linked to chromosome 3q28. Nat Genet. 2000;26:211–5.
Ferré M, Amati-Bonneau P, Tourmen Y, Malthiery Y, Reynier P. eOPA1: an online database for OPA1 mutations. Hum Mutat. 2005;25:423–8.
Toomes C, Marchbank NJ, Mackey DA, Craig JE, Newbury-Ecob RA, Bennett CP, et al. Spectrum, frequency and penetrance of OPA1 mutations in dominant optic atrophy. Hum Mol Genet. 2001;10:1369–78.
Puomila A, Huoponen K, Mäntyjärvi M, Hämäläinen P, Paananen R, Sankila EM, et al. Dominant optic atrophy: correlation between clinical and molecular genetic studies. Acta Ophthalmol Scand. 2005;83:337–46.
Kjer B, Eiberg H, Kjer P, Rosenberg T. Dominant optic atrophy mapped to chromosome 3q region. II. Clinical and epidemiological aspects. Acta Ophthalmol Scand. 1996;74:333–9.
Man PYW, Griffiths PG, Brown TD, Howell N, Turnbull DM, Chinnery PF. The epidemiology of Leber hereditary optic neuropathy in the North East of England. Am J Hum Genet. 2003;72:333–9.
Puomila A, Hämäläinen P, Kivioja S, Savontaus ML, Koivumäki S, Huoponen K, et al. Epidemiology and penetrance of Leber hereditary optic neuropathy in Finland. Eur J Hum Genet. 2007;15:1079–89.
Leber T. Ueber hereditaer und congenital angelegte Sehnervenleiden. Graefes Arch Ophthalmol. 1871;17:249–91.
Eiberg H, Kjer B, Kjer P, Rodenberg T. Dominant optic atrophy (OPA1) mapped to chromosome 3q region. I. Linkage analysis. Hum Mol Genet. 1994;3:977–80.
Lunkes A, Hartung U, Magarino C, Rodriguez M, Palmero A, Rodriguez L, et al. Refinement of the OPA1 gene locus on chromosome 3q28–q29 to a region of 2–8 cM, in one Cuban pedigree with autosomal dominant optic atrophy type Kjer. Am J Hum Genet. 1995;57:968–70.
Delettre C, Griffoin JM, Kaplan J, DollfusH Lorenz B, Faivre L, et al. Mutation spectrum and splicing variants in the OPA1 gene. Hum Genet. 2001;109:584–9.
Okamoto MP, Tripet B, Litowski J, Hodges RS, Vallee RB. Multiple distinct coiled-coils are involved in dynamin self-assembly. J Biol Chem. 1999;274:10277–86.
Smirnova E, Shurland DL, Newman-Smith ED, Pishvaee B, van der Blieki AM. A model for dynamin self-assembly based on binding between three different protein domains. J Biol Chem. 1999;274:14942–7.
Olichon A, Baricault L, Gas N, Guillou E, Valette A, Belenguer P, et al. Loss of OPA1 perturbates the mitochondrial inner membrane structure and integrity, leading to cytochrome c release and apoptosis. J Biol Chem. 2003;278:7743–6.
Ferré M, Bonneau D, Milea D, Chevrollier A, Verny C, Dollfus H, et al. Molecular screening of 980 cases of suspected hereditary optic neuropathy with a report on 77 novel OPA1 mutations. Hum Mutat. 2009;30:692–705.
Amati-Bonneau P, Guichet A, Olichon A, Chevrollier A, Viala F, Miot S, et al. OPA1 R445H mutation in optic atrophy associated with sensorineural deafness. Ann Neurol. 2005;58:958–63.
Shimizu S, Mori N, Kishi M, Sugata H, Tsuda A, Kubota N, et al. A novel mutation of the type 1 optic atrophy (OPA1) gene in a Japanese family with OPA1. Nippon Ganka Gakkai Zasshi. 2002;106:398–403.
Nakamura M, Lin J, Ueno S, Asaoka R, Hirai T, Hotta Y, et al. Novel mutations in the OPA1 gene and associated clinical features in Japanese patients with optic atrophy. Ophthalmology. 2006;113:483–8.
Amati-Bonneau P, Milea D, Bonneau D, Chevrollier A, Ferré M, Guillet V, et al. OPA1-associated disorders: phenotypes and pathophysiology. Int J Biochem Cell Biol. 2009;41:1855–65.
Pesch UEA, Leo-Kottler B, Mayer S, Jurklies B, Kellner U, Apfelstedt-Sylla E, et al. OPA1 mutations in patients with autosomal dominant optic atrophy and evidence for semi-dominant inheritance. Hum Mol Genet. 2001;10:1359–68.
Schimpf S, Fuhrmann N, Schaich S, Wissinger B. Comprehensive cDNA study and quantitative transcript analysis of mutant OPA1 transcripts containing premature termination codons. Hum Mutat. 2008;29:106–12.
Olichon A, Landes T, Arnauné-Pelloquin L, Emorine LJ, Mils V, Guichet A, et al. Effects of OPA1 mutations on mitochondrial morphology and apoptosis: relevance to ADOA pathogenesis. J Cell Physiol. 2007;211:423–30.
Alavi MV, Bette S, Schimpf S, Schuettauf F, Schraermeyer U, Wehrl H, et al. A splice site mutation in the murine Opa1 gene features pathology of autosomal dominant optic atrophy. Brain. 2007;130:1029–42.
Davies VJ, Hollins AJ, Piechota MJ, Yip W, Davies J, White KE, et al. Opa1 deficiency in a mouse model of autosomal dominant optic atrophy impairs mitochondrial morphology, optic nerve structure and visual function. Hum Mol Genet. 2007;16:1307–18.
Acknowledgments
This study was supported in part by a Grant in Aid, no. 20261901, from the Research Grant from the Study Group on Chorioretinal Degeneration and Optic Atrophy, The Ministry of Health, Labor and Welfare, Japan. The authors indicate no financial conflict of interest. The authors thank Dr. Miyuki Yoshikawa for the patients she introduced to this study.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hamahata, T., Fujimaki, T., Fujiki, K. et al. OPA1 mutations in Japanese patients suspected to have autosomal dominant optic atrophy. Jpn J Ophthalmol 56, 91–97 (2012). https://doi.org/10.1007/s10384-011-0096-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-011-0096-1