Abstract
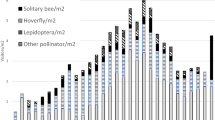
Larvae of common green lacewings (Chrysoperla carnea species-complex) are important generalist predators, and can serve as valuable biological control agents. Since only larvae are predatory, oviposition is of crucial importance. Recently, a ternary floral bait was described, attractive to females that would then lay their eggs in the vicinity of the baits. Earlier studies have shown that females prefer to lay their eggs on spines and hairs of plants. In the present study, we tested whether the number of eggs laid could be increased by combining chemical, visual and tactile stimuli. Common green lacewings were caught equally well in baited funnel traps covered with white, yellow, fluorescent yellow, light blue or black sheets, showing no preference to any of the colours. In further tests the field effect of the ternary floral bait on the oviposition site choice of common green lacewings was confirmed, as in the absence of the bait only negligible numbers of eggs were recorded on egg-laying plates. In the presence of the ternary floral bait, lacewings laid more eggs on spiny than on smooth surfaces. Removal of previously laid eggs did not affect the number of eggs laid afterwards. Our studies demonstrated that by combining the ternary floral bait and an appropriate spiny surface, oviposition of common green lacewings can be increased and concentrated to a relatively small area. Possible practical applications are discussed.



Similar content being viewed by others
References
Atlihan R, Kaydan B, Özgökce MS (2004) Feeding activity and life history characteristics of the generalist predator, Chrysoperla carnea (Neuroptera: Chrysopidae) at different prey densities. J Pest Sci 77:17–21
Blackmer JL, Byers JA, Rodriguez-Saona CR (2008) Evaluation of color traps for monitoring Lygus spp.: design, placement, height, time of day, and non-target effects. Crop Prot 27:171–181
Bozsik A (1992) Natural adult food of some important Chrysopa species (Planipennia: Chrysopidae). Acta Phytopathol Entomol Hung 27:141–146
Canard M (2001) Natural food and feeding habits of lacewings. In: McEwen PK, New TR, Whittington A (eds) Lacewings in the crop environment. Cambridge University Press, Cambridge, pp 116–128
Capinera JL (1980) Visual responses of some sugarbeet insects to sticky traps of various yellow and orange hues positioned at two heights. Southwest Entomol 5:76–79
Capinera JL, Walmsley MR (1978) Visual responses of some sugarbeet insects to sticky traps and water pan traps of various colors. J Econ Entomol 71:926–927
Duelli P (2001) Lacewings in field crops. In: McEwen PK, New TR, Whittington A (eds) Lacewings in the crop environment. Cambridge University Press, Cambridge, pp 158–171
Flint HM, Salter SS, Walters S (1979) Caryophyllene: an attractant for the green lacewing. Environ Entomol 8:1123–1125
Fréchette B, Coderre D, Lucas É (2006) Chrysoperla rufilabris (Neuroptera: Chrysopidae) females do not avoid ovipositing in the presence of conspecific eggs. Biol Control 37:354–358
Hagley EAC (1989) Release of Chrysoperla carnea Stephens (Neuroptera: Chrysopidae) for control of the green apple aphid, Aphis pomi deGeer (Homoptera: Aphididae). Can Entomol 121:309–314
Henry CS, Brooks SJ, Thierry D, Duelli P, Johnson JB (2001) The common green lacewing (Chrysoperla carnea s. lat.) and the sibling species problem. In: McEwen PK, New TR, Whittington A (eds) Lacewings in the crop environment. Cambridge University Press, Cambridge, pp 29–42
Jaastad G, Hatleli L, Knudsen GK, Tóth M (2010) Volatiles initiate egg laying in common green lacewings. IOBC/wprs Bull 54:77–82
Jenser G, Szita É, Szénási Á, Vörös G, Tóth M (2010) Monitoring the population of vine thrips (Drepanothrips reuteri Uzel) (Thysanoptera: Thripidae) by using fluorescent yellow sticky traps. Acta Phytopathol Entomol Hung 45:329–335
Koczor S, Szentkirályi F, Birkett MA, Pickett JA, Voigt E, Tóth M (2010) Attraction of Chrysoperla carnea complex and Chrysopa spp. lacewings (Neuroptera: Chrysopidae) to aphid sex pheromone components and a synthetic blend of floral compounds in Hungary. Pest Manag Sci 66:1374–1379
Koczor S, Knudsen GK, Hatleli L, Szentkirályi F, Tóth M (2015) Manipulation of oviposition and overwintering site choice of common green lacewings with synthetic lure (Neuroptera: Chrysopidae). J Appl Entomol 139:201–206
Kral K, Stelzl M (1998) Daily visual sensitivity pattern in the green lacewing Chrysoperla carnea (Neuroptera: Chrysopidae). Eur J Entomol 95:327–333
Maisonneuve JC, Marrec C (1999) The potential of Chrysoperla lucasina for IPM programmes in greenhouses. IOBC/wprs Bull 22:165–168
Maredia KM, Gage SH, Landis DA, Wirth TM (1992) Visual response of Coccinella septempunctata (L.), Hippodamia parenthesis (Say), (Coleoptera: Coccinellidae), and Chrysoperla carnea (Stephens), (Neuroptera: Chrysopidae) to colors. Biol Control 2:253–256
McEwen PK, Shuja A, Senior L (1998) Conservation of the common green lacewing (Chrysoperla carnea s.l.) (Neuroptera, Chrysopidae) to control crop pests. Acta Zool Fenn 209:153–156
Obrycki JJ, Hamid MN, Sajap AS, Lewis LC (1989) Suitability of corn insect pests for development and survival of Chrysoperla carnea and Chrysopa oculata (Neuroptera: Chrysopidae). Environ Entomol 18:1126–1130
Pappas ML, Broufas GD, Koveos DS (2011) Chrysopid predators and their role in biological control. J Entomol 8:301–326
Prokopy RJ, Owens ED (1983) Visual detection of plants by herbivorous insects. Annu Rev Entomol 28:337–364
R Core Team (2012) R A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL: http://www.R-project.org/
Ruzicka Z (1996) Oviposition-deterring pheromone in Chrysopidae (Neuroptera): intra- and interspecific effects. Eur J Entomol 93:161–166
Sattar M, Abro GH (2011) Mass rearing of Chrysoperla carnea (Stephens) (Neuroptera: Chrysopidae) adults for integrated pest management programmes. Pak J Zool 43:483–487
Schmera D, Tóth M, Subchev M, Sredkov I, Szarukán I, Jermy T, Szentesi Á (2004) Importance of visual and chemical cues in the development of an attractant trap for Epicometis (Tropinota) hirta Poda (Coleoptera: Scarabaeidae). Crop Prot 23:939–944
Sprent P, Smeeton NC (2007) Applied nonparametric statistical methods. Chapman & Hall/CRC, Boca Raton
Szentkirályi F (2001a) Ecology and habitat relationships. In: McEwen PK, New TR, Whittington A (eds) Lacewings in the crop environment. Cambridge University Press, Cambridge, pp 82–115
Szentkirályi F (2001b) Lacewings in fruit and nut crops. In: McEwen PK, New TR, Whittington A (eds) Lacewings in the crop environment. Cambridge University Press, Cambridge, pp 172–238
Szentkirályi F (2001c) Lacewings in vegetables, forests, and other crops. In: McEwen PK, New TR, Whittington A (eds) Lacewings in the crop environment. Cambridge University Press, Cambridge, pp 239–291
Tóth M, Bozsik A, Szentkirályi F, Letardi A, Tabilio MR, Verdinelli M, Zandigiacomo P, Jekisa J, Szarukán I (2006) Phenylacetaldehyde: a chemical attractant for common green lacewings (Chrysoperla carnea s.l., Neuroptera: Chrysopidae). Eur J Entomol 103:267–271
Tóth M, Szentkirályi F, Vuts J, Letardi A, Tabilio MR, Jaastad G, Knudsen GK (2009) Optimization of a phenylacetaldehyde-based attractant for common green lacewings (Chrysoperla carnea s.l., Neuroptera: Chrysopidae). J Chem Ecol 35:449–458
Udayagiri S, Mason CE, Pesek JD (1997) Coleomegilla maculata, Coccinella septempunctata (Coleoptera: Coccinellidae), Chrysoperla carnea (Neuroptera: Chrysopidae), and Macrocentrus grandii (Hymenoptera: Braconidae) trapped on colored sticky traps in corn habitats. Environ Entomol 26:983–988
Vas J, Ábrahám L, Markó V (1999) Study of nocturnal and diurnal activities of lacewings (Neuropteroidea: Raphidioptera, Neuroptera) by suction trap. Acta Phytopathol Entomol Hung 34:149–152
Zaki FN, El-Shaarawy MF, Farag NA (1999) Release of two predators and two parasitoids to control aphids and whiteflies. J Pest Sci 72:19–20
Zhu J, Cossé AA, Obrycki JJ, Boo KS, Baker TC (1999) Olfactory reactions of the twelve-spotted lady beetle, Coleomegilla maculata and the green lacewing, Chrysoperla carnea to semiochemicals released from their prey and host plant: electroantennogram and behavioral responses. J Chem Ecol 25:1163–1177
Acknowledgments
This research was partially supported by the National Research Development and Innovation Office (NKFIH) grant PD 115938 of the Hungarian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human and animals
All applicable international, national guidelines for the care and use of animals were followed.
Informed consent
Informed consent does not apply to these studies.
Additional information
Communicated by P. G. Becher.
Rights and permissions
About this article
Cite this article
Koczor, S., Szentkirályi, F., Fekete, Z. et al. Smells good, feels good: oviposition of Chrysoperla carnea-complex lacewings can be concentrated locally in the field with a combination of appropriate olfactory and tactile stimuli. J Pest Sci 90, 311–317 (2017). https://doi.org/10.1007/s10340-016-0785-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-016-0785-0