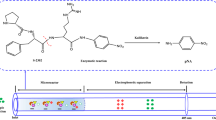
Abstract
In this work, a new immobilized enzyme reactor (IMER) containing gelatinase enzyme (a mixture of MMP-2 and -9) was developed and then inserted into a HPLC system to evaluate the inhibitory potency of novel compounds towards gelatinase. The gelatinase was immobilized on an epoxy-silica column by in situ technique. To monitor the performance of the newly developed method, the kinetic constant (K m) of immobilized gelatinase as well as the inhibition constant (IC50) of a known inhibitor, i.e., LY52, were determined and these results were compared with those obtained by classical succinyl-gelatin assay. The IMER-HPLC system was subsequently applied to the screening of 39 synthetic compounds. The inhibitory potential of these inhibitors (expressed in IC50) was found to be in agreement with the literature.






Similar content being viewed by others
References
Roy R, Yang J, Moses MA (2009) Matrix metalloproteinases as novel biomarkers and potential therapeutic targets in human cancer. J Clin Oncol 27:5287–5297
Hu JL, Van den Steen PE, Sang QXA, Opdenakker G (2007) Matrix metalloproteinase inhibitors as therapy for inflammatory and vascular diseases. Nat Rev Drug Discov 6:480–498
MacFadyen RJ (2007) Can matrix metalloproteinase inhibitors provide a realistic therapy in cardiovascular medicine? Curr Opin Pharmacol 7:171–178
Polette M, Nawrocki-Raby B, Gilles C, Clavel C, Birembaut P (2004) Tumour invasion and matrix metalloproteinases. Crit Rev Oncol Hemat 49:179–186
Fink K, Boratynski J (2012) The role of metalloproteinases in modification of extracellular matrix in invasive tumor growth, metastasis and angiogenesis. Postep Hig Med Dosw 66:609–628
Krstic J, Santibanez JF (2014) Transforming growth factor-beta and matrix metalloproteinases: functional interactions in tumor stroma-infiltrating myeloid cells. Sci World J 2014:521754
Bourboulia D, Stetler-Stevenson WG (2010) Matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs): positive and negative regulators in tumor cell adhesion. Semin Cancer Biol 20:161–168
Park SY, Shin HW, Lee KB, Lee MJ, Jang JJ (2010) Differential expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in thioacetamide-induced chronic liver injury. J Korean Med Sci 25:570–576
Koivunen E, Arap W, Valtanen H, Rainisalo A, Medina OP, Heikkila P et al (1999) Tumor targeting with a selective gelatinase inhibitor. Nat Biotechnol 17:768–774
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174
Lee M, Celenza G, Boggess B, Blase J, Shi Q, Toth M et al (2009) A potent gelatinase inhibitor with anti-tumor-invasive activity and its metabolic disposition. Chem Biol Drug Des 73:189–202
Auge F, Hornebeck W, Laronze JY (2004) A novel strategy for designing specific gelatinase A inhibitors: potential use to control tumor progression. Crit Rev Oncol Hemat 49:277–282
Kleifeld O, Kotra LP, Gervasi DC, Brown S, Bernardo MM, Fridman R et al (2001) X-ray absorption studies of human matrix metalloproteinase-2 (MMP-2) bound to a highly selective mechanism-based inhibitor. comparison with the latent and active forms of the enzyme. J Biol Chem 276:17125–17131
Baragi VM, Shaw BJ, Renkiewicz RR, Kuipers PJ, Welgus HG, Mathrubutham M et al (2000) A versatile assay for gelatinases using succinylated gelatin. Matrix Biol 19:267–273
Bartolini M, Cavrini V, Andrisano V (2004) Monolithic micro-immobilized-enzyme reactor with human recombinant acetylcholinesterase for online inhibition studies. J Chromatogr A 1031:27–34
de Oliveira KB, Mischiatti KL, Fontana JD, de Oliveira BH (2014) Tyrosinase immobilized enzyme reactor: development and evaluation. J Chromatogr B 945:10–16
Li Y, Chen Y, Xiao C, Chen D, Xiao Y, Mei Z (2014) Rapid screening and identification of alpha-amylase inhibitors from Garcinia xanthochymus using enzyme-immobilized magnetic nanoparticles coupled with HPLC and MS. J Chromatogr B 960:166–173
Bartolini M, Greig NH, Yu QS, Andrisano V (2009) Immobilized butyrylcholinesterase in the characterization of new inhibitors that could ease Alzheimer’s disease. J Chromatogr A 1216:2730–2738
Spross J, Sinz A (2010) A capillary monolithic trypsin reactor for efficient protein digestion in online and offline coupling to ESI and MALDI mass spectrometry. Anal Chem 82:1434–1443
Ma X, Chan EC (2010) On-line chromatographic screening of matrix metalloproteinase inhibitors using immobilized MMP-9 enzyme reactor. J Chromatogr B 878:1777–1783
Mazzini F, Nuti E, Petri A, Rossello A (2011) Immobilization of matrix metalloproteinase 8 (MMP-8) for online drug screening. J Chromatogr B 879:756–762
Cheng XC, Wang Q, Fang H, Tang W, Xu WF (2008) Design, synthesis and preliminary evaluation of novel pyrrolidine derivatives as matrix metalloproteinase inhibitors. Eur J Med Chem 43:2130–2139
Cheng XC, Wang Q, Fang H, Tang W, Xu WF (2008) Design, synthesis and evaluation of novel sulfonyl pyrrolidine derivatives as matrix metalloproteinase inhibitors. Bioorgan Med Chem 16:5398–5404
Cheng XC, Wang Q, Fang H, Tang W, Xu WF (2008) Synthesis of new sulfonyl pyrrolidine derivatives as matrix metalloproteinase inhibitors. Bioorgan Med Chem 16:7932–7938
Calleri E, Massolini G, Lubda D, Temporini C, Loiodice F, Caccialanza G (2004) Evaluation of a monolithic epoxy silica support for penicillin G acylase immobilization. J Chromatogr A 1031:93–100
Liu YQ, Lipponen K, Cilpa-Karhu G, Oorni K, Riekkola ML (2012) Plasma low-density lipoprotein immobilized silica as stationary phase in nano-liquid chromatography. J Chromatogr A 1270:104–110
Yuan YX, Xu WF, Liu J, Chen MH, Meng H, Qu XJ (2006) Inhibitory effects of matrix metalloproteinase (MMP) inhibitor LY52 on expression of MMP-2 and MMP-9 and invasive ability of human ovarian carcinoma cell line SKOV3. Chin J Cancer 25:663–670
Acknowledgments
This work was financially supported by the Shandong Provincial Natural Science Foundation of China (2009ZRB02230).
Conflict of interest
The authors have declared no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liang, W., Hou, Z., Wang, H. et al. Immobilized Enzyme Reactor Chromatography for Online Gelatinase Inhibitors Screening. Chromatographia 78, 763–773 (2015). https://doi.org/10.1007/s10337-015-2904-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-015-2904-0