Abstract
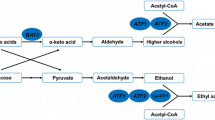
Acetate esters and higher alcohols greatly influence the quality and flavor profiles of Chinese Baijiu (Chinese liquor). Various mutants have been constructed to investigate the interactions of ATF1 overexpression, IAH1 deletion, and BAT2 deletion on the production of acetate esters and higher alcohols. The results showed that the overexpression of ATF1 under the control of the PGK1 promoter with BAT2 and IAH1 double-gene deletion led to a higher production of acetate esters and a lower production of higher alcohols than the overexpression of ATF1 with IAH1 deletion or overexpression of ATF1 with BAT2 deletion. Moreover, deletion of IAH1 in ATF1 overexpression strains effectively increased the production of isobutyl acetate and isoamyl acetate by reducing the hydrolysis of acetate esters. The decline in the production of higher alcohol by the ATF1 overexpression strains with BAT2 deletion is due to the interaction of ATF1 overexpression and BAT2 deletion. Mutants with varying abilities of producing acetate esters and higher alcohols were developed by genetic engineering. These strains have great potential for industrial application.





Similar content being viewed by others
References
Derrick S, Large PJ (1993) Activities of the enzymes of the Ehrlich pathway and formation of branched-chain alcohols in Saccharomyces cerevisiae and Candida utilis grown in continuous culture on valine or ammonium as sole nitrogen source. J Gen Microbiol 139:2783–2792. doi:10.1099/00221287-139-11-2783
Dickinson JR, Norte V (1993) A study of branched-chain amino acid aminotransferase and isolation of mutations affecting the catabolism of branched-chain amino acids in Saccharomyces cerevisiae. FEBS Lett 326:29–32. doi:10.1016/0014-5793(93)81754-N
Dickinson JR, Salgado LEJ, Hewlins MJE (2003) The catabolism of amino acids to long chain and complex alcohols in Saccharomyces cerevisiae. J Biol Chem 278:8028–8034. doi:10.1074/jbc.M211914200
Eden A, Nedervelde LV, Drukker M, Benvenisty N, Debourg A (2001) Involvement of branched-chain amino acid aminotransferases in the production of fusel alcohols during fermentation in yeast. Appl Microbiol Biotechnol 55:296–300. doi:10.1007/s002530000506
Fan W, Qian MC (2005) Headspace solid phase microextraction and gas chromatography-olfactometry dilution analysis of young and aged Chinese “Yanghe Daqu” liquors. J Agric Food Chem 53:7931–7938. doi:10.1021/jf051011k
Fan W, Qian MC (2006) Characterization of aroma compounds of Chinese “Wuliangye” and “Jiannanchun” liquors by aroma extract dilution analysis. J Agric Food Chem 54:2695–2704. doi:10.1021/jf052635t
Fukuda K, Kuwahata O, Kiyokawa Y, Yanagiuchi T, Wakai Y, Kitamoto K, Inoue Y, Kimura A (1996) Molecular cloning and nucleotide sequence of the isoamyl acetate-hydrolyzing esterase gene (EST2) from Saccharomyces cerevisiae. J Ferment Bioeng 82:8–15. doi:10.1016/0922-338X(96)89447-5
Fukuda K, Yamamoto N, Kiyokawa Y, Yanagiuchi T, Wakai Y, Kitamoto K, Inoue Y, Kimura A (1998) Balance of activities of alcohol acetyltransferase and esterase in Saccharomyces cerevisiae is important for production of isoamyl acetate. Appl Environ Microbiol 64:4076–4078. doi:10.1016/0003-2697(71)90136-9
Gietz RD, Woods RA (2002) Transformation of yeast by lithium acetate/single-stranded carrier DNA/polyethylene glycol method. Methods Enzymol 350:87–96. doi:10.1016/S0076-6879(02)50957-5
Gueldener U, Heinisch J, Koehler GJ, Voss D, Hegemann JH (2002) A second set of loxP marker cassettes for Cre-mediated multiple gene knockouts in budding yeast. Nucl Acids Res 30:88–94. doi:10.1093/nar/30.6.e23
Güldener U, Heck S, Fielder T, Beinhauer J, Hegemann JH (1996) A new efficient gene disruption cassette for repeated use in budding yeast. Nucl Acids Res 24:2519–2524. doi:10.1093/nar/24.13.2519
Hirosawa I, Aritomi K, Hoshida H, Kashiwagi S, Nishizawa Y, Akada R (2004) Construction of a self-cloning sake yeast that overexpresses alcohol acetyltransferase gene by a two-step gene replacement protocol. Appl Microbiol Biotechnol 65:68–73. doi:10.1007/s00253-004-1563-0
Kispal G, Steiner H, Court DA, Rolinski B, Lill R (1996) Mitochondrial and cytosolic branched-chain amino acid transaminases from yeast, homologs of the myc oncogene-regulated Eca39 protein. J Biol Chem 271:24458–24464. doi:10.1074/jbc.271.40.24458
Kobayashi M, Shimizu H, Shioya S (2008) Beer volatile compounds and their application to low-malt beer fermentation. J Biosc Bioeng 106:317–323. doi:10.1263/jbb.106.317
Lilly M, Bauer FF, Lambrechts MG, Swiegers JH, Cozzolino D, Pretorius IS (2006) The effect of increased yeast alcohol acetyltransferase and esterase activity on the flavour profiles of wine and distillates. Yeast 23:641–659. doi:10.1002/yea.1382
Lilly M, Bauer FF, Styger G, Lambrechts MG, Pretorius IS (2006) The effect of increased branched-chain amino acid transaminase activity in yeast on the production of higher alcohols and on the flavour profiles of wine and distillates. FEMS Yeast Res 6:726–743. doi:10.1111/j.1567-1364.2006.00057.x
Lilly M, Lambrechts MG, Pretorius IS (2000) Effect of increased yeast alcohol acetyltransferase activity on flavor profiles of wine and distillates. Appl Environ Microbiol 66:744–753. doi:10.1128/AEM.66.2.744-753.2000
Lu J, Dong J, Wu D, Chen Y, Guo X, Shi Y, Sun X, Xiao D (2012) Construction of recombinant industrial brewer’s yeast with lower diacetyl production and proteinase A activity. Eur Food Res Technol 235:951–961. doi:10.1007/s00217-012-1821-9
Ma J, Lu Q, Yuan Y, Ge H, Li K, Zhao W, Gao Y, Niu L, Teng M (2011) Crystal structure of isoamyl acetate-hydrolyzing esterase from Saccharomyces cerevisiae reveals a novel active site architecture and the basis of substrate specificity. Proteins 79:662–668. doi:10.1002/prot.22865
Meng X, Wu Q, Wang L, Wang D, Chen L, Xu Y (2015) Improving flavor metabolism of Saccharomyces cerevisiae by mixed culture with Bacillus licheniformis for Chinese maotai-flavor liquor making. J Ind Microbiol Biotechnol 42:1–8. doi:10.1007/s10295-015-1647-0
Nordström K (1962) Formation of ethyl acetate in fermentation with brewer’s yeast. III. Participation of coenzyme A. J I Brewing 68:398–407. doi:10.1002/j.2050-0416.1962.tb01882.x
Nordström K (1963) Formation of ethyl acetate in fermentation with brewer’s yeast: IV. Metabolism of acetyl-coenzyme A. J Inst Brew 69:142–153. doi:10.1002/j.2050-0416.1963.tb01910.x
Nordström K (1964) Formation of esters from alcohols by brewer’s yeast. J Inst Brew 70:328–336. doi:10.1002/j.2050-0416.1964.tb01999.x
Stribny J, Querol A, Pérez-Torrado R (2016) Differences in enzymatic properties of the Saccharomyces kudriavzevii and Saccharomyces uvarum alcohol acetyltransferases and their impact on aroma-active compounds production. Front Microbiol 7:897. doi:10.3389/fmicb.2016.00897
Swiegers JH, Pretorius IS (2005) Yeast modulation of wine flavor. Adv Appl Microbiol 57:131–175. doi:10.1016/S0065-2164(05)57005-9
Verstrepen KJ, Van Laere SD, Vanderhaegen BM, Derdelinckx G, Dufour JP, Pretorius IS, Winderickx J, Thevelein JM, Delvaux FR (2003) Expression levels of the yeast alcohol acetyltransferase genes ATF1, Lg-ATF1, and ATF2 control the formation of a broad range of volatile esters. Appl Environ Microbiol 69:5228–5237. doi:10.1128/AEM.69.9.5228-5237.2003
Wu Q, Kong Y, Xu Y (2015) Flavor profile of Chinese liquor is altered by interactions of intrinsic and extrinsic microbes. Appl Environ Microbiol 82:422–430. doi:10.1128/AEM.02518-15
Wu XH, Zheng XW, Han BZ, Vervoort J, Nout MJ (2009) Characterization of Chinese liquor starter, “Daqu”, by flavor type with 1H NMR-based nontargeted analysis. J Agric Food Chem 57:11354–11359. doi:10.1021/jf902881p
Xiao M, Wang G, Liu F (1996) Study on higher alcohols production by microbes in the course of sorghum solid-state Chinese-liquor fermentation. Food Ferment Ind 19:65–85 (in Chinese)
Xiao ZB, Yu D, Niu YW, Chen F, Song SQ, Zhu JC, Zhu GY (2014) Characterization of aroma compounds of Chinese famous liquors by gas chromatography-mass spectrometry and flash GC electronic-nose. J Chromatogr B 945–946:92–100. doi:10.1016/j.jchromb.2013.11.032
Yang D, Luo X, Wang X (2014) Characteristics of traditional Chinese shanlan wine fermentation. J Biosci Bioeng 117:203–207. doi:10.1016/j.jbiosc.2013.07.010
Zhang CY, Liu YL, Qi YN, Zhang JW, Dai LH, Lin X, Xiao DG (2013) Increased esters and decreased higher alcohols production by engineered brewer’s yeast strains. Eur Food Res Technol 236:1009–1014. doi:10.1007/s00217-013-1966-1
Zhang CY, Qi YN, Ma HX, Li W, Dai LH, Xiao DG (2015) Decreased production of higher alcohols by Saccharomyces cerevisiae, for Chinese rice wine fermentation by deletion of Bat aminotransferases. J Ind Microbiol Biotechnol 42:617–625. doi:10.1007/s10295-015-1583-z
Zhang JW, Zhang CY, Dai LH, Dong J, Liu YL, Guo XW, Xiao DG (2012) Effects of overexpression of the alcohol acetyltransferase-encoding gene ATF1 and disruption of the esterase-encoding gene IAH1 on the flavour profiles of Chinese yellow rice wine. Int J Food Sci Technol 47:2590–2596. doi:10.1111/j.1365-2621.2012.03140.x
Zheng XW, Tabrizi MR, Nout MJR, Han BZ (2011) Daqu—a traditional Chinese liquor fermentation starter. J Inst Brew 117:82–90. doi:10.1002/j.2050-0416.2011.tb00447.x
Zheng XW, Yan Z, Nout MJR, Boekhout T, Han BZ, Zwietering MH, Eddy JS (2014) Characterization of the microbial community in different types of Daqu samples as revealed by 16s rRNA and 26s rRNA gene clone libraries. World J Microbiol Biotechnol 31:199–208. doi:10.1007/s11274-014-1776-z
Zhu J, Lin J, Palomec L, Wheeldon I (2015) Microbial host selection affects intracellular localization and activity of alcohol-o-acetyltransferase. Microb Cell Fact 14:1–10. doi:10.1186/s12934-015-0221-9
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (31471724) and the Major Project of Research Program on Applied Fundamentals and Advanced Technologies of Tianjin (14JCZDJC32900).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, W., Wang, JH., Zhang, CY. et al. Regulation of Saccharomyces cerevisiae genetic engineering on the production of acetate esters and higher alcohols during Chinese Baijiu fermentation. J Ind Microbiol Biotechnol 44, 949–960 (2017). https://doi.org/10.1007/s10295-017-1907-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-017-1907-2