Abstract
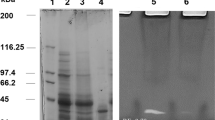
In this study, we detail the specificity of an aspartic peptidase from Rhizomucor miehei and evaluate the effects of this peptidase on clotting milk using the peptide sequence of k-casein (Abz-LSFMAIQ-EDDnp) and milk powder. Molecular mass of the peptidase was estimated at 37 kDa, and optimum activity was achieved at pH 5.5 and 55 °C. The peptidase was stable at pH values ranging from 3 to 5 and temperatures of up 45 °C for 60 min. Dramatic reductions in proteolytic activity were observed with exposure to sodium dodecyl sulfate, and aluminum and copper (II) chloride. Peptidase was inhibited by pepstatin A, and mass spectrometry analysis identified four peptide fragments (TWSISYGDGSSASGILAK, ASNGGGGEYIFGGYDSTK, GSLTTVPIDNSR, and GWWGITVDRA), similar to rhizopuspepsin. The analysis of catalytic specificity showed that the coagulant activity of the peptidase was higher than the proteolytic activity and that there was a preference for aromatic, basic, and nonpolar amino acids, particularly methionine, with specific cleavage of the peptide bond between phenylalanine and methionine. Thus, this peptidase may function as an important alternative enzyme in milk clotting during the preparation of cheese.



Similar content being viewed by others
References
Aoki K, Matsubara S, Umeda M, Tachibanac S, Doi M, Takenaka S (2013) Aspartic protease from Aspergillus (Eurotium) repens strain MK82 is involved in the hydrolysis and decolourisation of dried bonito (Katsuobushi). J Sci Food Agric 93:1349–1355
Bersanetti PA, Park HY, Bae KS, Son KH, Shin DH, Hirata IY, Juliano MA, Carmona AK, Juliano L (2005) Characterization of arazyme, an exocellular metalloprotease isolated from Serratia proteamaculans culture medium. Enzyme Microb Tech 37:574–581
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein-dye binding. Anal Biochem 72:248–254
Brinkworth RI, Prociv P, Loukas A, Brindley PJ (2001) Hemoglobin-degrading, aspartic proteases of blood-feeding parasites: substrate specificity revealed by homology models. J Biol Chem 42:38844–38851
Da Silva RR, Caetano RC, Okamoto DN, de Oliveira LCG, Bertolin TC, Juliano MA, Juliano L, Oliveira AHC, Rosa JC, Cabral H (2014) The identification and biochemical properties of the catalytic specificity of a serine peptidase secreted by Aspergillus fumigatus Fresenius. Protein Pept Lett 21:663–671
De Oliveira AHC, Cruz AK, Greene LJ, Rosa JC, Ward RJ (2006) Subproteomic analysis of soluble proteins of the microsomal fraction from two Leishmania species. Comp Biochem Physiol Part D 3:300–308
Dunn BM (1989) Determination of protease mechanism. In: Beynon RJ, Bond JS (eds) Proteolytic enzymes: a practical approach. IRL Press, NY, pp 57–81
El-Enshasy HA (2007) Filamentous fungal cultures–process characteristics, products, and applications. In: Shang-Tian Y (ed) Bioprocessing for Value-Added Products from Renewable Resources. Elsevier BV, pp 225–262
Graminho ER, Silva RR, Cabral TPF, Arantes EC, da Rosa NG, Juliano L, Okamoto DN, Oliveira LCG, Kondo MY, Juliano MA, Cabral H (2013) Purification, characterization, and specificity determination of a new serine protease secreted by Penicillium waksmanii. Appl Biochem Biotechnol 169:201–214
Hajji M, Kanoun S, Nasri M, Gharsallah N (2007) Purification and characterization of an alkaline serine-protease produced by a new isolated Aspergillus clavatus ES1. Process Biochem 42:791–797
Hirata IY, Cezari MHS, Nakaie CR, Boschcov P, Ito AS, Juliano MA, Juliano L (1994) Internally quenched fluorogenic protease substrates: Solid-phase synthesis and fluorescence spectroscopy of peptides containing ortho-aminobenzoyl/dinitrophenyl groups as donor-acceptor pairs. Lett Pept Sci 1:299–308
Hsiao N-W, Chen Y, Kuan Y-C, Lee Y-C, Lee S-K, Chan H-H, Kao C-H (2014) Purification and characterization of an aspartic protease from the Rhizopus oryzae protease extract, Peptidase R. Electron J Biotechnol 17:89–94
Klemencic I, Carmona AK, Cezari MHS, Juliano MA, Juliano L, Guncar G, Turk D, Krizaj I, Turk V, Turk B (2000) Biochemical characterization of human cathepsin X revealed that the enzyme is an exopeptidase, acting as carboxymonopeptidase or carboxydipeptidase. Eur J Biochem 267:5404–5412
Kumar A, Grover S, Sharma J, Batish VK (2010) Chymosin and other milk coagulants: sources and biotechnological interventions. Crit Rev Biotechnol 30:243–258
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Merheb-Dini C, Gomes E, Boscolo M, da Silva R (2010) Production and characterization of a milk-clotting protease in the crude enzymatic extract from the newly isolated Thermomucor indicae-seudaticae N31 (Milk-clotting protease from the newly isolated Thermomucor indicae-seudaticae N31). Food Chem 120:87–93
Merheb-Dini C, Cabral H, Leite RSR, Zanphorlin LM, Okamoto DN, Rodriguez GOB, Juliano L, Arantes EC, Gomes E, Da Silva R (2009) Biochemical and functional characterization of a metalloprotease from the thermophilic fungus Thermoascus aurantiacus. J Agr Food Chem 57:9210–9217
Meyers SP, Ahearn DG (1977) Extracellular proteolysis by Candida lipolytica. Mycologia 69:646–651
Neto YAAH, de Oliveira LCG, de Oliveira AHC, Rosa JC, Juliano MA, Juliano L, Rodrigues A, Cabral H (2015) Determination of specificity and biochemical characteristics of neutral protease isolated from Myceliophthora thermophila. Protein Pept Lett 22:972–982
Silva RR, Angelo T, Cabral H (2013) Comparative evaluation of peptidases produced by Penicillium corylophilum and Penicillium waksmanii in solid state fermentation using agro-industrial residues. J Agric Sci Technol B 3:230–237
Ramão A, Gimenez M, Laure HJ, Izumi C, Vida RC, Oba-Shinjo S, Marie SK, Rosa JC (2012) Changes in the expression of proteins associated with aerobic glycolysis and cell migration are involved in tumorigenic ability of two glioma cell lines. Proteome Sci 53:1–12. doi:10.1186/1477-5956-10-53
Rao S, Mizutani O, Hirano T, Masaki K, Lefuji H (2011) Purification and characterization of a novel aspartic protease from basidiomycetous yeast Cryptococcus sp. S-2. J Biosci Bioeng 112:441–446
Rao MB, Tanksale AM, Ghatge MS, Deshpande VV (1998) Molecular and biotechnological aspects of microbial proteases. Microbiol Mol Biol Rev 62:597–635
Sarath G, de la Motte RS, Wagner FW (1996) Protease assay methods. In: Beynon RJ, Bond JS (eds) Proteolytic enzymes: a practical approach. IRL Press, NY, pp 25–55
Schechter I, Berger A (1967) On the size of the active site in proteases. I. Papain. Biochem Bioph Res Co 27:157–162
See YS, Jackowski G (1989) Estimating molecular weights of polypeptides by SDS gel electrophoresis. In: Creigton TE (ed) Protein structure a practical approach. Oxford University, New York, pp 1–19
Silva RR, Cabral TPF, Rodrigues A, Cabral H (2013) Production and partial characterization of serine and metallo peptidases secreted by Aspergillus fumigatus Fresenius in submerged and solid state fermentation. Braz J Microbiol 44:235–243
Turk B, Kriz Ïaj I, Kralj B, Dolenc I, Popovic ÏT, Bieth JG, Turk V (1993) Bovine stefin C, a new member of the stefin family. J Biol Chem 268:7323–7329
Yegin S, Fernandez-Lahore M, Salgado AJG, Guvenc U, Goksungur Y, Tari C (2011) Aspartic proteinases from Mucor spp. in cheese manufacturing. Appl Microbiol Biotechnol 89:949–960
Acknowledgments
The authors would like to acknowledge the financial support provided by Fundação de Amparo à Pesquisa do Estado de São Paulo-FAPESP (2011/06986-0 and 2012/24703-8) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (308078/2012-8).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
da Silva, R.R., Souto, T.B., de Oliveira, T.B. et al. Evaluation of the catalytic specificity, biochemical properties, and milk clotting abilities of an aspartic peptidase from Rhizomucor miehei . J Ind Microbiol Biotechnol 43, 1059–1069 (2016). https://doi.org/10.1007/s10295-016-1780-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-016-1780-4