Abstract
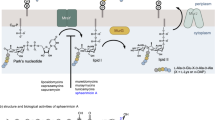
Natural products represent a major source of approved drugs and still play an important role in supplying chemical diversity. Consistently, 2014 has seen new, natural product-derived antibiotics approved for human use by the US Food and Drug Administration. One of the recently approved second-generation glycopeptides is dalbavancin, a semi-synthetic derivative of the natural product A40,926. This compound inhibits bacterial growth by binding to lipid intermediate II (Lipid II), a key intermediate in peptidoglycan biosynthesis. Like other recently approved antibiotics, dalbavancin has a complex history of preclinical and clinical development, with several companies contributing to different steps in different years. While our work on dalbavancin development stopped at the previous company, intriguingly our current pipeline includes two more Lipid II-binding natural products or derivatives thereof. In particular, we will focus on the properties of NAI-107 and related lantibiotics, which originated from recent screening and characterization efforts.




Similar content being viewed by others
References
Alduina R, Lo Piccolo L, D’Alia D, Ferraro C, Gunnarsson N, Donadio S, Puglia AM (2007) Phosphate-controlled regulator for the biosynthesis of the dalbavancin precursor A40926. J Bacteriol 189:8120–8129
Arnison PG et al (2013) Ribosomally synthesized and post-translationally modified peptide natural products: overview and recommendations for a universal nomenclature. Nat Prod Rep 30:108–160
Breukink E, de Kruijff B (2006) Lipid II as a target for antibiotics. Nat Rev Drug Discov 5:321–332
Bugg TD, Braddick D, Dowson CG, Roper DI (2011) Bacterial cell wall assembly: still an attractive antibacterial target. Trends Biotechnol 29:167–173
Ciabatti R et al (2007) Synthesis and preliminary biological characterization of new semisynthetic derivatives of ramoplanin. J Med Chem 50:3077–3085
Cruz JCS, Iorio M, Monciardini P, Simone M, Gaspari E, Maffioli S, Wellington E, Sosio M, Brunati C, Donadio S (2015) A brominated variant of the lantibiotic NAI-107 with enhanced antibacterial potency. J Nat Prod (accepted)
Dischinger J, Basi Chipalu S, Bierbaum G (2014) Lantibiotics: promising candidates for future applications in health care. Int J Med Microbiol 304:51–62
Donadio S, Sosio M, Serina S, Mercorillo D (2009) Genes and proteins for the biosynthesis of the lantibiotic 107891. PCT/IB2007/002270
Errington J (2013) L-form bacteria, cell walls and the origins of life. Open Biol 3:120143
Fabbretti A, He CG, Gaspari E, Maffioli S, Brandi L, Spurio R, Sosio M, Jabes D, Donadio S (2015) A derivative of the thiopeptide GE2270A highly selective against Propionibacterium acnes. Antimicrob Agents Chemother 59:4560–4568
Foulston LC, Bibb MJ (2010) Microbisporicin gene cluster reveals unusual features of lantibiotic biosynthesis in actinomycetes. Proc Natl Acad Sci USA 107:13461–13466
Iorio M et al (2014) A glycosylated, labionin-containing lanthipeptide with marked antinociceptive activity. ACS Chem Biol 9:398–404
Jabés D, Brunati C, Candiani G, Riva S, Romanó G, Donadio S (2011) Efficacy of the new lantibiotic NAI-107 in experimental infections induced by multidrug-resistant Gram-positive pathogens. Antimicrob Agents Chemother 55:1671–1676
Jabes D, Brunati C, Candiani G, Riva S, Romanó G, Maffioli S, Rossi R, Simone M, Gaspari E, Donadio S (2014) Pharmacological properties of NAI-603, a well-tolerated semisynthetic derivative of ramoplanin. Antimicrob Agents Chemother 58:1922–1929
Jabes D, Donadio S (2010) Strategies for the isolation and characterization of antibacterial lantibiotics. Methods Mol Biol 618:31–45
Kahne D, Leimkuhler C, Lu W, Walsh C (2005) Glycopeptide and lipoglycopeptide antibiotics. Chem Rev 105:425–448
Koehn FE (2008) New strategies and methods in the discovery of natural product anti-infective agents: the mannopeptimycins. J Med Chem 51:2613–2617
Lancini G (2006) Forty years of antibiotic research at Lepetit: a personal journey. SIM News 56:192–212
Lazzarini A, Gastaldo L, Candiani G, Ciciliato I, Losi D, Marinelli F, Selva E, Parenti F (2005) Antibiotic 107891, its factors A1 and A2, pharmaceutically acceptable salts and compositions, and use thereof. WO/2005/014628
Lepak AJ, Marchillo K, Craig WA, Andes DR (2015) In vivo pharmacokinetics and pharmacodynamics of the lantibiotic NAI-107 in a neutropenic murine thigh infection model. Antimicrob Agents Chemother 59:1258–1264
Ling LL et al (2015) A new antibiotic kills pathogens without detectable resistance. Nature 517:455–459
Lo Grasso L, Maffioli S, Sosio M, Bibb M, Puglia AM, Alduina R (2015) Two master switch regulators trigger A40926 biosynthesis in Nonomuraea sp. strain ATCC 39727. J Bacteriol 197:2536–2544
Maffioli SI, Iorio M, Sosio M, Monciardini P, Gaspari E, Donadio S (2014) Characterization of the congeners in the lantibiotic NAI-107 complex. J Nat Prod 77:79–84
Maffioli SI, Monciardini P, Catacchio B, Mazzetti C, Münch D, Brunati C, Sahl HG, Donadio S (2015) Family of class I lantibiotics from actinomycetes and improvement of their antibacterial activities. ACS Chem Biol 10:1034–1042
Maffioli SI, Potenza D, Vasile F, De Matteo M, Sosio M, Marsiglia B, Rizzo V, Scolastico C, Donadio S (2009) Structure revision of the lantibiotic 97518. J Nat Prod 72:605–607
Malabarba A, Ciabatti R, Scotti R, Goldstein BP, Ferrari P, Kurz M, Andreini BP, Denaro M (1995) New semisynthetic glycopeptides MDL 63,246 and MDL 63,042, and other amide derivatives of antibiotic A-40,926 active against highly glycopeptide-resistant VanA enterococci. J Antibiot 48:869–883
Malabarba A, Goldstein BP (2005) Origin, structure, and activity in vitro and in vivo of dalbavancin. J Antimicrob Chemother 55(Suppl S2):ii15–ii20
Malabarba A, Pallanza R, Berti M, Cavalleri B (1990) Synthesis and biological activity of some amide derivatives of the lantibiotic actagardine. J Antibiot 43:1089–1097
Monciardini P, Iorio M, Maffioli S, Sosio M, Donadio S (2014) Discovering new bioactive compounds from microbial sources. Microb Biotechnol 7:209–220
Müller A, Münch D, Schmidt Y, Reder-Christ K, Schiffer G, Bendas G, Gross H, Sahl HG, Schneider T, Brötz-Oesterhelt H (2012) The lipodepsipeptide empedopeptin inhibits cell wall biosynthesis through Ca2+-dependent complex formation with peptidoglycan precursors. J Biol Chem 287:20270–20280
Münch D, Müller A, Schneider T, Kohl B, Wenzel M, Bandow JE, Maffioli S, Sosio M, Donadio S, Wimmer R, Sahl HG (2014) The lantibiotic NAI-107 binds to bactoprenol-bound cell wall precursors and impairs membrane functions. J Biol Chem 289:12063–12076
Pozzi R, Coles M, Linke D, Kulik A, Nega M, Wohlleben W, Stegmann E (2015) Distinct mechanisms contribute to immunity in the lantibiotic NAI-107 producer strain Microbispora ATCC PTA-5024. Environ Microbiol
Schneider T et al (2010) Plectasin, a fungal defensin, targets the bacterial cell wall precursor Lipid II. Science 328:1168–1172
Schneider T, Sahl HG (2011) An oldie but a goodie—cell wall biosynthesis as antibiotic target pathway. Int J Med Microbiol 300:161–169
Simone M, Monciardini P, Gaspari E, Donadio S, Maffioli SI (2013) Isolation and characterization of NAI-802, a new lantibiotic produced by two different Actinoplanes strains. J Antibiot 66:73–78
Sosio M, Canavesi A, Stinchi S, Donadio S (2010) Improved production of A40926 by Nonomuraea sp. through deletion of a pathway-specific acetyltransferase. Appl Microbiol Biotechnol 87:1633–1638
Sosio M, Stinchi S, Beltrametti F, Lazzarini A, Donadio S (2003) The gene cluster for the biosynthesis of the glycopeptide antibiotic A40926 by Nonomuraea species. Chem Biol 10:541–549
Walker S, Chen L, Hu Y, Rew Y, Shin D, Boger DL (2005) Chemistry and biology of ramoplanin: a lipoglycodepsipeptide with potent antibiotic activity. Chem Rev 105:449–476
Acknowledgments
This work was partially supported by the European Community’s Seventh Framework Programme (FP7/2007–2013) under grant agreements 289285 (TRAIN-ASAP) and 245066 (LAPTOP) and by a joint grant from Regione Lombardia and Italian Ministry of Education, University and Research (ProjectID 30190679).
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue: Natural Product Discovery and Development in the Genomic Era. Dedicated to Professor Satoshi Ōmura for his numerous contributions to the field of natural products.
Rights and permissions
About this article
Cite this article
Maffioli, S.I., Cruz, J.C.S., Monciardini, P. et al. Advancing cell wall inhibitors towards clinical applications. J Ind Microbiol Biotechnol 43, 177–184 (2016). https://doi.org/10.1007/s10295-015-1703-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-015-1703-9