Abstract
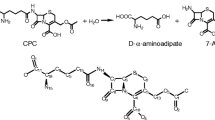
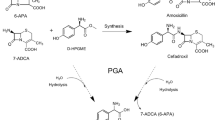
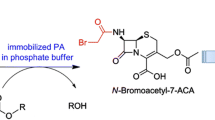
Cephalosporin C (CPC) acylase is important for the one-step production of 7-aminocephalosporanic acid (7-ACA), a key intermediate for cephalosporin antibiotics. However, its application is hampered by the low activity, substrate inhibition, and product inhibition. In this study, two rounds of combinatorial active-site saturation testing (CASTing) were carried out on the CPC acylase acyII from Pseudomonas SE83, and one mutant H57βA/H70βY with no substrate inhibition was obtained. For further engineering to reduce the product inhibition, a quick pH indicator assay was developed, allowing for real-time monitoring of the product inhibition in the presence of added 7-ACA. The utility of the assay was demonstrated by screening six libraries of site-directed saturation mutagenesis libraries of H57βA/H70βY. A new mutant H57βA/H70βY/I176βN was obtained, which showed a k cat 3.26-fold and a K IP 3.08-fold that of the wild type, respectively. Given the commercial value of the enzyme, both this pH indicator assay and the triple mutant should be useful for further engineering of the enzyme to increase the specific activity and to decrease the product inhibition.




Similar content being viewed by others
References
Adam Y, Tayer N, Rotem D, Schreiber G, Schuldiner S (2007) The fast release of sticky protons: kinetics of substrate binding and proton release in a multidrug transporter. Proc Natl Acad Sci USA 104:17989–17994
Aramori I, Fukagawa M, Tsumura M, Iwami M, Ono H, Ishitani Y, Kojo H, Kohsaka M, Ueda Y, Imanaka H (1992) Comparative characterization of new glutaryl 7-ACA and cephalosporin C acylases. J Ferment Bioeng 73:185–192
Aramori I, Fukagawa M, Tsumura M, Iwami M, Yokota Y, Kojo H, Kohsaka M, Ueda Y, Imanaka H (1991) Isolation of soil strains producing new cephalosporin acylases. J Ferment Bioeng 72:227–231
Banerjee A, Kaul P, Sharma R, Banerjee U (2003) A high-throughput amenable colorimetric assay for enantioselective screening of nitrilase-producing microorganisms using pH sensitive indicators. J Biomol Screen 8:559–565
Conlon HD, Baqai J, Baker K, Shen YQ, Wong BL, Noiles R, Rausch CW (1995) Two-step immobilized enzyme conversion of cephalosporin C to 7-aminocephalosporanic acid. Biotechnol Bioeng 46:510–513
Deng C, Chen RR (2004) A pH-sensitive assay for galactosyltransferase. Anal Biochem 330:219–226
Emily G, Rachel P, Wan JT, Anandhi A, Gavin F, Gianluca M, Elena R, Loredano P, Alice V (2013) Structure of a class III engineered cephalosporin acylase: comparisons to class I acylase and implications for differences in substrate specificity and catalytic activity. Biochem J 451:217–226
Gasteiger E, Gattiker A, Hoogland C, Ivanyi I, Appel RD, Bairoch A (2003) Expasy: the proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res 31:3784–3788
Gibbons BH, Edsall JT (1963) Rate of hydration of carbon dioxide and dehydration of carbonic acid at 25°. J Biol Chem 238:3502–3507
John GT, Heinzle E (2001) Quantitative screening method for hydrolases in microplates using pH indicators: determination of kinetic parameters by dynamic pH monitoring. Biotechnol Bioeng 72:620–627
Li Y, Chen J, Jiang W, Mao X, Zhao G, Wang E (1999) In vivo post-translational processing and subunit reconstitution of cephalosporin acylase from Pseudomonas sp. 130. Eur J Biochem 262:713–719
Martínez-Martínez I, Montoro-García S, Lozada-Ramírez JD, Sánchez-Ferrer Á, García-Carmona F (2007) A colorimetric assay for the determination of acetyl xylan esterase or cephalosporin C acetyl esterase activities using 7-amino cephalosporanic acid, cephalosporin C, or acetylated xylan as substrate. Anal Biochem 369:210–217
Matsuda A, Matsuyama K, Yamamoto K, Ichikawa S, Komatsu K (1987) Cloning and characterization of the genes for two distinct cephalosporin acylases from a Pseudomonas strain. J Bacteriol 169:5815–5820
Matsuda A, Toma K, Komatsu K-l (1987) Nucleotide sequences of the genes for two distinct cephalosporin acylases from a Pseudomonas strain. J Bacteriol 169:5821–5826
Oh B, Kim M, Yoon J, Chung K, Shin Y, Lee D, Kim Y (2003) Deacylation activity of cephalosporin acylase to cephalosporin C is improved by changing the side-chain conformations of active-site residues. Biochem Biophys Res Commun 310:19–27
Otten L, Sio C, Reis C, Koch G, Cool R, Quax W (2007) A highly active adipyl-cephalosporin acylase obtained via rational randomization. FEBS J 274:5600–5610
Otten LG, Sio CF, Vrielink J, Cool RH, Quax WJ (2002) Altering the substrate specificity of cephalosporin acylase by directed evolution of the beta-subunit. J Biol Chem 277:42121–42127
Pan J-L, Syu M-J (2005) Kinetic study on substrate and product inhibitions for the formation of 7-amino-3-deacetoxy cephalosporanic acid from cephalosporin G by immobilized penicillin G acylase. Biochem Eng J 23:203–210
Patett F, Fischer L (2006) Spectrophotometric assay for quantitative determination of 7-aminocephalosporanic acid from direct hydrolysis of cephalosporin C. Anal Biochem 350:304–306
Persson M, Palcic MM (2008) A high-throughput pH indicator assay for screening glycosyltransferase saturation mutagenesis libraries. Anal Biochem 378:1–7
Pollegioni L, Lorenzi S, Rosini E, Marcone GL, Molla G, Verga R, Cabri W, Pilone MS (2005) Evolution of an acylase active on cephalosporin C. Protein Sci 14:3064–3076
Pollegioni L, Rosini E, Molla G (2013) Cephalosporin C acylase: dream and (/or) reality. Appl Microbiol Biotechnol 97:2341–2355
Reetz MT, Bocola M, Carballeira JD, Zha D, Vogel A (2005) Expanding the range of substrate acceptance of enzymes: combinatorial active-site saturation test. Angew Chem Int Ed 44:4192–4196
Reetz MT, Kahakeaw D, Lohmer R (2008) Addressing the numbers problem in directed evolution. ChemBioChem 9:1797–1804
Reyes F, Martinez MJ, Soliveri J (1989) Determination of cephalosporin-C amidohydrolase activity with fluorescamine. J Pharm Pharmacol 41:136–137
Salazar O, Sun L (2003) Evaluating a screen and analysis of mutant libraries. In: Arnold FH, Georgiou G (eds) Methods in molecular biology, vol 230. Humana, Totowa, pp 85–97
Shin YC, Jeon JY, Jung KH, Park MR, Kim Y (2007) Cephalosporin C acylase mutant and method for preparing 7-ACA using same. US Patent 7592168
Sio CF, Otten LG, Cool RH, Quax WJ (2003) Analysis of a substrate specificity switch residue of cephalosporin acylase. Biochem Biophys Res Commun 312:755–760
Tischer W, Giesecke U, Lang G, Röder A, Wedekind F (1992) Biocatalytic 7-aminocephalosporanic acid production. Ann NY Acad Sci 672:502–509
Wang Y, Yu H, Song W, An M, Zhang J, Luo H, Shen Z (2012) Overexpression of synthesized cephalosporin C acylase containing mutations in the substrate transport tunnel. J Biosci Bioeng 113:36–41
Yu K, Hu S, Huang J, Mei LH (2011) A high-throughput colorimetric assay to measure the activity of glutamate decarboxylase. Enzyme Microb Technol 49:272–276
Zhao H (2003) A pH-indicator-based screen for hydrolytic haloalkane dehalogenase. In: Arnold FH, Georgiou G (eds) Directed enzyme evolution: screening and selection methods, vol 230. Humana, Totowa, pp 213–221
Acknowledgments
This work was supported by grants from the National Basic Research Program of China (2009CB724704 and 2013CB733902), and a grant from the National High-Tech Program of China (2012AA022205).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xiao, Y., Huo, X., Qian, Y. et al. Engineering of a CPC acylase using a facile pH indicator assay. J Ind Microbiol Biotechnol 41, 1617–1625 (2014). https://doi.org/10.1007/s10295-014-1501-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1501-9