Abstract
Objectives
The twice-weekly administration of 25 mg of etanercept (TW) has been shown to be effective in patients with rheumatoid arthritis (RA). However, the once-weekly administration of 25 mg of etanercept (OW) was tried in order to address the economic burden of anti-rheumatic biologics. We evaluated the clinical and radiographic results from a 2-year follow-up study of patients receiving OW or TW.
Methods
Sixty-three biologics-naive patients with RA were randomly assigned to receive either OW (n = 42) or TW (n = 21).
Results
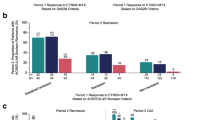
From baseline to year 2, rates of clinical remission, according to the Disease Activity Score of 28 joints (DAS-28) (based on C-reactive protein; CRP)–with clinical remission being regarded as a DAS-28 (CRP) score of <2.3–were significantly improved in the OW group (from 1.6 to 39.0%) and in the TW group (from 9.5 to 47.6%), but no significant between-group difference was observed at year 2. Radiographic joint damage, quantified with the modified Sharp score, was significantly progressive in the OW group in contrast to findings in the TW group. Thus, among patients receiving TW therapy, the progression of joint damage may have been inhibited or may have shown remission.
Conclusions
These results suggest that, in terms of DAS-28 remission, OW therapy can efficiently substitute for TW therapy in biologics-naive patients with RA. However, TW therapy was indispensable in preventing the worsening of joint damage.




Similar content being viewed by others
References
Cole P, Rabasseda X. The soluble tumor necrosis factor receptor etanercept. Drugs Today. 2004;40:281–324.
Weinblatt ME, Kremer JM, Bankhurst AD, Bulpitt KJ, Fleischmann RM, Fox RI, et al. A trial of etanercept, a recombinant tumor necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N Engl J Med. 1999;340:253–9.
van der Heijde D, Klareskog L, Landewé R, Bruyn GA, Cantagrel A, Durez P, et al. Disease remission and sustained halting of radiographic progression with combination etanercept and methotrexate in patients with rheumatoid arthritis. Arthritis Rheum. 2007;56:3928–39.
Emery P, Breedveld FC, Hall S, Durez P, Chang DJ, Robertson D, et al. Comparison of methotrexate monotherapy with a combination of methotrexate and etanercept in active, early, moderate to severe rheumatoid arthritis (COMET): a randomised, double-blind, parallel treatment trial. Lancet. 2008;372:375–82.
Emery P, Kvlein TK, Combe B, Foehl J, Pobertson D, Pedersen R, et al. Very early (<4 months) treatment with combination etanercept and methotrexate produces significantly better remission rates: results from the COMET study. Ann Rheum Dis. 2010;69(Suppl 3):57–8.
Koike T, Harigai M, Inokuma A, Inoue K, Ishiguro N, Ryu J, et al. Postmarketing surveillance of the safety and effectiveness of etanercept in Japan. J Rheumatol. 2009;36:898–906.
Felson DT, Anderson JJ, Boers M, Bombardier C, Furst D, Goldsmith C, et al. American College of Rheumatology preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum. 1995;38:727–35.
PMDA. New drug application and complete response letter of etanercept (53039601_21700AMY). http://www.info.pmda.go.jp/approvalSrch/ApprovalSrch.
Botsios C, Furlan A, Ostuni P, Striso P, Todesco S, Punzi L. Effects of low-dose etanercept in maintaining DAS-remission previously achieved with standard-dose in patients with rheumatoid arthritis. Ann Rheum Dis. 2007;66(Suppl II):54.
Prevoo ML, van’t Hof MA, Kuper HH, van Leeuwen MA,van de Putte LB, van Riel PL, et al. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995;38:44–8.
van der Heijde D. How to read radiographs according to the Sharp/van der Heijde method. J Rheumatol. 2000;27:261–3.
van der Heijde D, Simon L, Smolen J, Strand V, Sharp J, Boers M, et al. How to report radiographic data in randomized clinical trial in rheumatoid arthritis: guidelines from a roundtable discussion. Arthritis Rheum. 2002;47:215–8.
van Gestel AM, Prevoo ML, van’t Hof MA, van Rijswijk MH, van de Putte LB, van Riel PL, et al. Development and validation of the European League Against Rheumatism response criteria for rheumatoid arthritis. Comparison with the preliminary American College of Rheumatology and the World Health Organization/International League Against Rheumatism Criteria. Arthritis Rheum. 1996;39:34–40.
Schoonjans F, Zalata A, Depuydt CE, Comhaire FH. MedCalc: a new computer program for medical statistics. Comput Methods Programs Biomed. 1995;48:257–62.
Klarenbeek NB, Koevoets R, van der Heijde DMFM, Gerards AH, ten Wolde S, Kerstens PJSM, et al. Association with joint damage and physical functioning of nine composite indices and the 2011. ACR/EULAR remission criteria in rheumatoid arthritis. Ann Rheum Dis. 2011;2011(70):1815–21.
Inoue E, Yamanaka H, Hara M, Tomotsu T, Komatani N. Comparison of disease activity score (DAS)28-erythrocyte sedimentation rate and DAS28-C-reactive protein threshold values. Ann Rheum Dis. 2007;66:407–9.
Blumenauer B, Judd M, Cranney A, Burls A, Coyle D, Hochberg M, et al. Etanercept for the treatment of rheumatoid arthritis. Cochrane Database Syst Rev. 2003; CD004525.
Miyasaka N. Clinical investigation in highly disease-affected rheumatoid arthritis patients in Japan with adalimumab applying standard and general evaluation: the CHANGE study. Mod Rheumatol. 2008;18:252–62.
Keystone C, Genovese MC, Klareskog L, Hsia EC, Hall ST, Miranda PC, et al. Golimumab, a human antibody to tumour necrosis factor α given by monthly subcutaneous injections, in active rheumatoid arthritis despite methotrexate therapy: the GO-FORWARD Study. Ann Rheum Dis. 2009;68:789–96.
Taylor PC. Pharmacology of TNF blockade in rheumatoid arthritis and other chronic inflammatory disease. Curr Opin Pharmacol. 2010;10:308–15.
Salmon-Ceron D, Tubach F, Lortholary O, Chosidow O, Bretagne S, Nicolas N, et al. Drug-specific risk of non-tuberculosis opportunistic infections in patients receiving anti-TNF therapy reported to the 3-year prospective French RATIO registry. Ann Rheum Dis. 2010 (Epub ahead of print).
Crum NF, Lederman ER, Wallace NR. Infections associated with tumor necrosis factor-alpha antagonists. Medicine. 2005;84:291–302.
Bartelds GM, Wijbrandts CA, Nurmohamed MT, Stapel S, Lems WF, Aarden L. Clinical response to adalimumab: relationship to anti-adalimumab antibodies and serum adalimumab concentrations in rheumatoid arthritis. Ann Rheum Dis. 2007;66:921–6.
Klareskog L, Wajdula J, Yeh P, Fatenejad S. Low autoantibody and anti-etanercept antibody formation and lack of impact on clinical outcomes following 5 years of treatment with Enbrel in patients with rheumatoid arthritis. Arthritis Rheum. 2005;52(Suppl 9):S348.
Dore RK, Mathews S, Schechtman J, Surbeck W, Mandel D, Patel A, et al. The immunogenicity, safety, and efficacy of etanercept liquid administered once weekly in patients with rheumatoid arthritis. Clin Exp Rheumatol. 2007;25(1):40–6.
Hetland RF, Christensen IJ, Tarp U, Dreyer L, Hansen A, Hansen IT, et al. Direct comparison of treatment responses, remission rate, and drug adherence in patients with rheumatoid arthritis treated with adalimumab, etanercept, or infliximab. Arthritis Rheum. 2010;62:22–32.
Smolen JS, Wells GA, Zhang B, van Tuyl LHD, Funovits J, Boers M. Predictive validity of the new preliminary ACR/EULAR definitions for remission in rheumatoid arthritis. Arthritis Rheum. 2010;62(Suppl):S882.
Felson DT, Smolen JS, Wells G, Zhang B, van Tuyl LHD, et al. American College of Rheumatology/European League Against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Arthritis Rheum. 2011;63:573–86.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Wada, T., Son, Y., Ozaki, Y. et al. Clinical and radiographic results from a 2-year comparison of once-weekly versus twice-weekly administration of etanercept in biologics-naive patients with rheumatoid arthritis. Mod Rheumatol 22, 824–830 (2012). https://doi.org/10.1007/s10165-011-0591-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10165-011-0591-8