Abstract
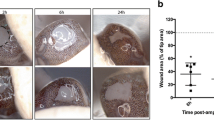
To provide quantitative information on arm regeneration in cuttlefish, the regenerating arms of two cuttlefish species, Sepia officinalis and Sepia pharaonis, were observed at regular intervals after surgical amputation. The third right arm of each individual was amputated to ~10–20 % starting length. Arm length, suction cup number, presence of chromatophores, and behavioral measures were collected every 2–3 days over a 39-day period and compared to the contralateral control arm. By day 39, the regenerating arm reached a mean 95.5 ± 0.3 % of the control for S. officinalis and 94.9 ± 1.3 % for S. pharaonis. The process of regeneration was divided into five separate stages based on macroscopic morphological events: Stage I (days 0–3 was marked by a frayed leading edge; Stage II (days 4–15) by a smooth hemispherical leading edge; Stage III (days 16–20) by the appearance of a growth bud; Stage IV (days 21–24) by the emergence of an elongated tip; and Stage V (days 25–39) by a tapering of the elongated tip matching the other intact arms. Behavioral deficiencies in swimming, body postures during social communication, and food manipulation were observed immediately after arm amputation and throughout Stages I and II, returning to normal by Stage III. New chromatophores and suction cups in the regenerating arm were observed as early as Stage II and by Stage IV suction cup number equaled that of control arms. New chromatophores were used in the generation of complex body patterns by Stage V. These results show that both species of cuttlefish are capable of fully regenerating lost arms, that the regeneration process is predictable and consistent within and across species, and provide the first quantified data on the rate of arm lengthening and suction cup addition during regeneration.





Similar content being viewed by others
References
Aldrich FA, Aldrich MM (1968) On regeneration of the tentacular arm of the giant squid Architeuthis dux Steenstrup (Decapoda, Architeuthidae). Can J Zool 46:845–847
Barbosa-Sabanero K, Hoffmann A, Judge C, Lightcap N, Tsonis PA, Del Rio-Tsonis K (2012) Lens and retina regeneration: new perspectives from model organisms. Biochem J 447:321–334
Domingues P, Bettencourt V, Guerra à (2006) Growth of Sepia officinalis in captivity and in nature. Vie Milieu Life Environ 56:109–120
Duval P, Chichery MP, Chichery R (1984) Prey capture by the cuttlefish (Sepia-officinalis L)—an experimental-study of 2 strategies. Behav Process 9:13–21
Feral J (1977) La regeneration des bras de Sepia officinalis L. (Mollusque, Cephalopode, Coleoide). Inaug Diss Univ Paris
Feral J (1978) La regeneration des bras de la seiche Sepia officinalis L. (Cephalopoda: Sepioidea) I. Etude morphologique. Cah Biol Mar 355–361
Feral J (1979) La regeneration des bras de la seiche Sepia officinalis L. (Cephalopoda: Sepioidea) II. Etude histologique et cytologique. Cah Biol Mar 29–42
Florey E (1969) Ultrastructure and function of cephalopod chromatophores. Am Zool 9:429–442
Gaston MR, Tublitz NJ (2006) Central distribution and three-dimensional arrangement of fin chromatophore motoneurons in the cuttlefish Sepia officinalis. Invertebr Neurosci IN 6:81–93
Hanlon R (2007) Cephalopod dynamic camouflage. Curr Biol CB 17:R400–R404
Kier WM, Schachat FH (2008) Muscle specialization in the squid motor system. J Exp Biol 211:164–169
Kier WM, Stella MP (2007) The arrangement and function of octopus arm musculature and connective tissue. J Morphol 268:831–843
Lange MM (1920) On the regeneration and fine structure of the arms of the cephalopods. J Exp Zool 31:1–57
Loi PK, Tublitz NJ (1998) Long term rearing of cuttlefish in a small scale facility. Aquar Sci Conserv 2:135–143
Matzner H, Gutfreund Y, Hochner B (2000) Neuromuscular system of the flexible arm of the octopus: physiological characterization. J Neurophysiol 83:1315–1328
Moffett SB (1995) Neural regeneration in gastropod molluscs. Prog Neurobiol 46:289–330
Murata M, Ishii M, Osako M (1981) On the regeneration of tentacle of oceanic squid, Ommastephes bartrami (Lesueur). Bull Hokkaido Reg Fish Res Lab 46:1–14
Rohrbach B, Schmidtberg H (2006) Sepia arms and tentacles: model systems for studying the regeneration of brachial appendages. Vie et Milieu Life Environ 56:175–190
Sicard RE (1985) Regulation of vertebrate limb regeneration. Oxford University Press, New York
Sumbre G, Fiorito G, Flash T, Hochner B (2005) Neurobiology: motor control of flexible octopus arms. Nature 433:595–596
Yacob J, Lewis AC, Gosling A, St Hilaire DH, Tesar L, McRae M, Tublitz NJ (2011) Principles underlying chromatophore addition during maturation in the European cuttlefish, Sepia officinalis. J Exp Biol 214:3423–3432
Yekutieli Y, Sagiv-Zohar R, Aharonov R, Engel Y, Hochner B, Flash T (2005a) Dynamic model of the octopus arm. I. Biomechanics of the octopus reaching movement. J Neurophysiol 94:1443–1458
Yekutieli Y, Sagiv-Zohar R, Hochner B, Flash T (2005b) Dynamic model of the octopus arm. II. Control of reaching movements. J Neurophysiol 94:1459–1468
Yoshinari N, Kawakami A (2011) Mature and juvenile tissue models of regeneration in small fish species. Biol Bull 221:62–78
Acknowledgments
This material is based upon work supported by the Air Force Office of Scientific Research under Award No. FA9550-09-1-0395.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tressler, J., Maddox, F., Goodwin, E. et al. Arm regeneration in two species of cuttlefish Sepia officinalis and Sepia pharaonis . Invert Neurosci 14, 37–49 (2014). https://doi.org/10.1007/s10158-013-0159-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10158-013-0159-8