Abstract
Background
To confirm the usefulness of a combination therapy of interleukin-2 (IL-2) and interferon-α (IFN-α) against metastatic renal cell carcinoma, the recommended dose of IFN-α to use in combination with low-dose IL-2 was determined (phase 1). Efficacy and safety at this dose was evaluated (phase 2).
Methods
In phase 1, the dose of IL-2 was fixed at 0.7 × 106 Japan reference unit (JRU)/person for 5 days a week. The dose of IFN-α was increased from 3 × 106 IU for 3 days a week (level I) to 6 × 106 IU for 3 days a week (level II) and to 6 × 106 IU for 5 days a week (level III).
Results
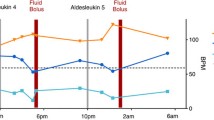
In phase 1, 10 patients were registered, with 9 (3 at each level) able to be evaluated. Because grade 3 and grade 4 neutropenia were observed at level III in 1 patient each, level II was found to be the recommended regimen. The response rate in phase 1 was 44.4% (4/9). In phase 2, 46 patients were registered, with a response rate in 37 patients, classified as per protocol set (PPS), of 21.6% (8/37). Toxicities observed were primarily flu-like symptoms due to cytokines, and gastrointestinal symptoms. Leukocyte abnormalities were observed, but they were milder and tolerable.
Conclusion
In the 46 patients evaluated in phase 1 and phase 2, the response rate was 26.1% (12/46), being highest in 38.7% (12/31) of those who were nephrectomized, and with only lung metastases.
Similar content being viewed by others
References
MP Wirth (1993) ArticleTitleImmunotherapy for metastatic renal cell carcinoma Urol Clin North Am 20 283–295 Occurrence Handle7684167 Occurrence Handle1:STN:280:ByyB283ntVY%3D
JS Horoszewicz GP Murphy (1989) ArticleTitleAn assessment of the current use of human interferons in therapy of urological cancers J Urol 142 1173–1180 Occurrence Handle2478725 Occurrence Handle1:STN:280:By%2BD2c%2FpsFw%3D
RM Bukowski (1997) ArticleTitleNatural history and therapy of metastatic renal cell carcinoma: the role of interleukin-2 Cancer 80 1198–1220 Occurrence Handle9317170 Occurrence Handle10.1002/(SICI)1097-0142(19971001)80:7<1198::AID-CNCR3>3.0.CO;2-H Occurrence Handle1:CAS:528:DyaK2sXms1ShtLo%3D
Physicians' Desk Reference (1994) Thomson Healthcare, United States, pp 801–804
S-M Liang B Allet K Rose et al. (1985) ArticleTitleCharacterization of human interleukin 2 derived from Escherichia coli Biochem J 229 429–439 Occurrence Handle3899106 Occurrence Handle1:CAS:528:DyaL2MXltVehsLo%3D
Y Aso H Tazaki T Umeda et al. (1989) ArticleTitleA phase II study of S-6820 (recombinant interleukin 2) on renal cell carcinoma Biotherapy 3 999–1007
Y Aso Y Homma H Tazaki et al. (1995) ArticleTitleA phase II trial of S-6820 (recombinant interleukin-2) on renal cell carcinoma refractory to interferon Hinyokigeka 8 75–86
T Tsuchida (1994) ArticleTitleIndication of activated units in recombinant interleukin-2. What is true or false? Biotherapy Today 1 41–43
JA Hank J Surfus J Gan et al. (1999) ArticleTitleDistinct clinical and laboratory activity of two recombinant interleukin-2 preparations Clin Cancer Res 5 281–289 Occurrence Handle10037176 Occurrence Handle1:CAS:528:DyaK1MXhsVWrt7w%3D
WHO Handbook for reporting results of cancer treatment (1979) WHO offset publication No.48. World Health Organization, Geneva
TR Fleming (1982) ArticleTitleOne-sample multiple testing procedure for phase II clinical trials Biometrics 38 143–151 Occurrence Handle7082756 Occurrence Handle10.2307/2530297 Occurrence Handle1:STN:280:Bi2B3cnptlY%3D
InstitutionalAuthorNameJapanese Urological Association and Japanese Society of Pathology (1992) ArticleTitleResponse criteria for urological cancer treatment Jpn J Urol 83 447–472
S Negrier B Escudier C Lasset et al. (1998) ArticleTitleRecombinant human interleukin-2, recombinant human interferon alpha-2a, or both in metastatic renal-cell carcinoma N Engl J Med 338 1272–1278 Occurrence Handle9562581 Occurrence Handle10.1056/NEJM199804303381805 Occurrence Handle1:CAS:528:DyaK1cXivF2qt74%3D
G Fyfe RI Fisher SA Rosenberg et al. (1995) ArticleTitleResults of treatment of 255 patients with metastatic renal cell carcinoma who received high dose recombinant interleukin-2 therapy J Clin Oncol 13 688–696 Occurrence Handle7884429 Occurrence Handle1:STN:280:ByqC1M%2FltF0%3D
T Umeda T Niijima (1986) ArticleTitlePhase II study of alpha interferon on renal cell carcinoma: summary of three collaborative trials Cancer 58 1231–1235 Occurrence Handle3742449 Occurrence Handle10.1002/1097-0142(19860915)58:6<1231::AID-CNCR2820580610>3.0.CO;2-# Occurrence Handle1:STN:280:BimB1MfgslU%3D
J-M Tourani C Pfister N Tubiana et al. (2003) ArticleTitleSubcutaneous interleukin-2 and interferon alfa administration in patients with metastatic renal cell carcinoma: final results of SCAPP III, a large, multicenter, phase II, nonrandomized study with sequential analysis design J Clin Oncol 21 3987–3994 Occurrence Handle14581421 Occurrence Handle10.1200/JCO.2003.02.073 Occurrence Handle1:CAS:528:DC%2BD2cXpsVajsbs%3D
S Dirk R Heike A Ralf et al. (2005) ArticleTitlePhase I clinical and pharmacokinetic study of the novel raf kinase and vascular endothelial growth factor receptor inhibitor BAY 43-9006 in patients with advanced refractory solid tumors J Clin Oncol 23 965–972
J Robert M Motzer M Dror et al. (2006) ArticleTitleActivity of SU11248, a multitargeted inhibitor of vascular endothelial growth factor receptor and platelet-derived growth factor receptor, in patients with metastatic renal cell carcinoma J Clin Oncol 24 16–24 Occurrence Handle10.1200/JCO.2005.04.1764
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Akaza, H., Tsukamoto, T., Onishi, T. et al. A low-dose combination therapy of interleukin-2 and interferon-α is effective for lung metastasis of renal cell carcinoma: a multicenter open study. Int J Clin Oncol 11, 434–440 (2006). https://doi.org/10.1007/s10147-006-0596-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-006-0596-z