Abstract
The relative importance of exploitation rate and environmental variability in generating fluctuations of harvested populations is a key issue in academic ecology as well as population management. We studied how the eastern Baltic cod (Gadus morhua) is affected by fishing and environmental variation by using a newly developed single species state-space model. Survey data and auxiliary environmental data were used to estimate the model parameters. The model was then used to predict future development of the eastern Baltic cod under different fishing mortalities and abiotic conditions. Abiotic condition was represented by an index: reproductive volume which is the volume of water suitable (in terms of salinity and oxygen content) for the successful development of the early life stages of Baltic cod. The model included direct density dependence, fishing, and a lagged effect of reproductive volume. Our analysis showed that fishing rate is approximately three times more important than reproductive volume in explaining the population dynamics. Furthermore, our model suggests either under- or over-compensatory dynamics depending on the reproductive volume and long term catch levels. It follows that fishing can either reduce or increase temporal oscillations of the cod stock depending on whether the dynamics is over- or undercompensatory, respectively. The sustainable level of fishing rate is however dependent on reproductive volume. Our model predicts a dual role of fishing rate, stabilizing when reproductive volume is high and destabilizing when it is low. Exploitation rate may therefore increase or decrease the risk of the population of cod dropping below a given biomass reference point depending on the environmental conditions, which has practical implications for fisheries management.




Similar content being viewed by others
References
Anderson CNK, Hsieh C, Sandin SA, Hewitt R, Hollowed A, Beddington J May, Sugihara RM (2008) Why fishing magnifies fluctuations in fish abundance. Nature 452:835–839
Berryman A (1999) Principles of population dynamics and their application. Stanley Thornes, Cheltenham
Box GEP, Jenkins MG (1976) Times series analysis: Forecasting and control. Holden day, San Francisco
Brook SP, Gelman P (1998) General methods for monitoring convergence of iterative simulations. J Comp Graph Stat 7:434–455
Cardinale M, Arrhenius F (2000) The influence of stock structure and environmental conditions on the recruitment process of Baltic cod estimated using a generalized additive model. Can J Fish Aquat Sci 12:2402–2409
Casini M, Lovgren J, Hjelm J, Cardinale M Molinero, Kornilovs JC (2008) Multi-level trophic cascades in a heavily exploited open marine ecosystem. Proc R Soc B-Biol Sci 275:1793–1801
Casini M, Hjelm J, Molinero J-C, Lövgren J, Cardinale M, Valerio B, Belgrano A, Kornilovs G (2009) Trophic cascades promote threshold-like shifts in pelagic marine ecosystems. Proc Natl Acad Sci USA 106:197–202
Dennis B, Kemp WP, Taper ML (1998) Joint density dependence. Ecology 79:426–444
Dennis B, Ponciano JM, Lele SR, Taper ML, Staples DF (2006) Estimating density dependence, process noise, and observation error. Ecol Monogr 76:323–341
Durbin J, Koopman SJ (2001) Time series analysis by state-space methods. Oxford University Press, Oxford
Eero M, Koster FW, MacKenzie BR (2008) Reconstructing historical stock development of Atlantic cod (Gadus morhua) in the eastern Baltic Sea before the beginning of intensive exploitation. Can J Fish Aquat Sci 65:2728–2741
Freeman SN, Kirkwood GP (1995) On a structural time series method for estimating stock biomass and recruitment from catch and effort data. Fish Res 22:77–98
Gamito S (1998) Growth models and their usage in ecological modeling: an application to a fish population. Ecol Model 113:83–94
Gelman A (2006) Prior distributions for variance parameters in hierarchical models (comment on article by Browne and Draper). Bayesian Analysis 1:515–533
Heikinheimo O (2008) Average salinity as an index for environmental forcing on cod recruitment in the Baltic Sea. Boreal Environ Res 13:457–464
Hilborn R (2012) The evolution of quantitative marine fisheries management 1985-2010. Nat Res Mod 25:122–144
Hilborn R, Walters CJ (1992) Quantitative fisheries stock assessment: choice, dynamics and uncertainty. Chapman & Hall, New York
Hillborn R (2001) Calculation of biomass trend, exploitation rate, and surplus production from survey data and catch. Can J Fish Aquat Sci 58:579–584
Hsieh C, Reiss CS, Hunter JR, Beddington JR, May RM, Sugihara G (2006) Fishing elevates variability in the abundance of exploited species. Nature 443:859–862
ICES (International Council for the Exploration of the Sea) (1990) Stock assessment reports from 1990 to 2010. ICES Headquarters, Copenhagen
ICES (2008) Report of the Baltic Fisheries Assessment Working Group (WGBFAS), 8 -17 April 2008, ICES Headquarters, Copenhagen. ICES CM 2008\ACOM:06
ICES (2009) Report of the ICES/HELCOM Working Group on Integrated Assessments of the Baltic Sea (WGIAB), 16–20 March 2009, Rostock, Germany. ICES Headquarters, Copenhagen. ICES CM 2009/BCC:02
ICES (2010) Report of the Baltic International Fish Survey Working Group (WGBIFS), 22–26 March 2010, Klaipeda, Lithuania. ICES Headquarters, Copenhagen. ICES CM 2010/SSGESST:07
ICES (2011) Report of the Baltic Fisheries Assessment Working Group (WGBFAS), 12-19 April 2011, Copenhagen, Denmark. ICES CM 2011/ACOM:10
Jonzén N, Lundberg P, Cardinale M, Arrhenius F (2001) Variable fishing mortality and the possible commercial extinction of the eastern Baltic cod. Mar Ecol-Prog Ser 210:291–296
Jonzén N, Ripa J, Lundberg P (2002a) A theory of stochastic harvesting in stochastic environments. Am Nat 159:427–437
Jonzén N, Cardinale M, Gardmark A, Arrhenius F, Lundberg P (2002b) Risk of collapse in the eastern Baltic cod fishery. Mar Ecol-Prog Ser 240:225–233
Jonzén N, Pople AR, Grigg GC, Possingham HP (2005) Of sheep and rain: large-scale population dynamics of the red kangaroo. J Anim Ecol 74:22–30
Kaitala V, Ranta E, Lindström J (1996) Cyclic population dynamics and random perturbations. J Anim Ecol 65:249–251
Knape J (2008) Estimability of density dependence in models of time series data. Ecology 89:2994–3000
Knape J, Jonzén N, Sköld M, Sokolov L (2009) Multivariate state-space modelling of bird migration count data. In: Thomson DL, Cooch EG, Conroy MJ (eds) Modeling demographic processes in marked populations. Environmental and ecological statistics, vol. 3. Springer, Berlin, pp 59-79
Köster FW, Möllmann C, Hinrichsen H-H, Tomkiewicz J, Wieland K, Kraus G, Voss R, MacKenzie BR, Schnack D, Makarchouk A, Plikshs M, Beyer JE (2005) Baltic cod recruitment—the impact of climate variability on key processes. ICES J Mar Sci 62:1408–1425
Lindegren M, Moellmann C, Nielsen A, Stenseth NC (2009) Preventing the collapse of the Baltic cod stock through an ecosystem-based management approach. Proc Natl Acad Sci USA 106:14722–14727
Lindegren M, Möllman C, Nielsen A, Brander K, MacKenzie BR, Stenseth NC (2010) Ecological forecasting under climate change: the case of the Baltic cod. Proc R Soc B-Biol Sci 277:2121–2130
Lobón-Cerviá J (2011) Why fishing does not magnify temporal fluctuations in the population abundance of stream-living salmonids. Rev Fish Sci 19:246–256
MacKenzie BR, Hinrichsen H-H, Plikshs K, Wieland K, Zerzera AS (2000) Quantifying environmental heterogeneity: habitat size necessary for successful development of cod Gadus morhua eggs in the Baltic Sea. Mar Ecol-Prog Ser 193:143–156
MacKenzie BR, Gislason H, Möllmann C, Köster FW (2007) Impact of 21st century climate change on the Baltic Sea fish community and fisheries. Global Change Biol 13:1348–1367
McIntosh RP (1985) The background of ecology: concept and theory. Cambridge University Press, Cambridge
Methot RD Jr, Wetzel CR (2013) Stock synthesis: a biological and statistical framework for fish stock assessment and fishery management. Fish Res 142:86–99
Millar B, Meyer R (2000) Bayesian state-space modeling of age-structured data: fitting a model is just the beginning. Can J Fish Aquat Sci 57:43–50
Nissling A, Vallin L (1996) The ability of Baltic cod eggs to maintain neutral buoyancy and the opportunity for survival in fluctuating conditions in the Baltic Sea. J Fish Biol 48:217–227
Plikshs M, Kalejs M, Graumann G (1993) The influence of environmental conditions and reproductive stock size on the year-class strength of the Eastern Baltic cod. ICES document CM 1993/J:22
Quinn TJ, Deriso RB (1999) Quantitative fishery dynamics. Oxford University Press, New York
Shelton AO, Mangel M (2011a) Fluctuations of fish populations and the magnifying effects of fishing. Proc Natl Acad Sci USA 108:7075–7080
Shelton OA, Mangel M (2011b) Reply to Sugihara et al.: the biology of variability in fish populations. Proc Natl Acad Sci USA 108:E1226–E1226
SMHI (2010) Swedish metrological and hydrological institute. Folkborgsvägen 1
Spiegelhalter DJ, Thomas A, Best N, Lunn D (2007) WINBUGS 1.4.3, Bayesian inference using Gibbs sampling. Manual. MRC Biostatistics Unit, Cambridge, UK
Sugihara G, Beddington J, Hsieh C-H, Deyle E, Fogarty M, Glaser SM, Hewitt R, Hollowed A, May RM, Munch SB, Perretti C, Rosenberg AA, Sandin S, Ye H (2011) Are exploited fish populations stable? Proc Natl Acad Sci USA 108:E1224–E1225
Thorsen AOS (1996) Physiological mechanisms of buoyancy in eggs from brackish water cod. J Fish Biol 48:457–477
Vainikka A, Gaardmark A, Bland B, Hjelm J (2009) Two- and three-dimensional maturation reaction norms for the eastern Baltic cod, Gadus morhua. ICES J Mar Sci 66:248–257
Walters CJ, Martell SJD (2004) Fisheries ecology and management. Princeton University Press, New Jersey
Wikström A, Ripa J, Jonzén N (2012) The role of harvesting in age-structured populations: disentangling dynamics and age truncation effects. Theor Popul Biol 82:348–354
Acknowledgments
A.W. was financially supported by the Swedish Research Council FORMAS (Grant No 2007-549 to N.J), and N.J. is holding a research fellowship from the Swedish Research Council. Thanks also to Hans-Harald Hinrichsen for his helpful provision of oxygen data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Professor N. Jonzén passed away on 9 May 2015.
Appendix
Appendix
Reproductive volume, dissolved oxygen or salinity as environmental covariate
We examined reproductive volume (RV t ), dissolved oxygen and salinity (from the Gotland basin 80–100 m in depth; SMHI 2010) as possible environmental covariates to explain changes in cod biomass. However, according to Table 1 salinity and dissolved oxygen did not have any predictive power in our dataset and we restricted the analysis to RV t .
A relation between index and total stock biomass
The SSB threshold level for the Eastern Baltic cod is estimated as the lowest level of SSB under which recruitment is considered impaired (ICES 2010). The latest available threshold value (SSB threshold) was in 2008 estimated by ICES at around 230,000 tonnes (ICES 2008). To be able to compare our result with recommendations from the ICES we therefore transformed their estimates of spawning stock biomass to an index according to:
In logarithm form this becomes:
SSB t is total spawning stock biomass, I t is the cod index, q and α are constants. To estimate q and α we regressed loge(I t ) on loge(SSB t ) using data from the time period 1991–2010. As the only available data on SSB t in the Baltic Sea originated from a VPA we used this source despite all potential problems pointed out earlier in this work. The point estimates are: q = 0.18 and α = 0.60.
The minimum sustainable spawning biomass (SSB threshold) for the Eastern Baltic cod is, according to ICES, 230,000 tonnes. Equation 5 with the estimated values of q and α gives the corresponding value: I threshold = 310 (number of cod caught per hour).
Model dynamics
To better understand the dynamic behaviour of Eq. 4 when simulated for different (fixed) values of catch (C t = C) and reproductive volume (RV t = R), we express the model in general terms:
and linearize around the deterministic equilibrium J * evaluated at the expected values of the noise, E(ε t ) = ε * = 0:
Denoting the partial derivatives:
Equation 8 can be written
The parameter a determines the endogenous dynamics, which is under-compensatory if a > 0 and over-compensatory if a < 0. By substituting γ = α 1 + α 3 C + α 4 R we find that f = J t exp(γ + α 2 J t + ε t ), J * = -γ/α 2, a = 1-γ and b = -γ/α 2. Because α 3 < 0 and α 4 > 0 (see Table 3), increased reproductive volume makes the dynamics more over-compensatory, whereas this effect is counteracted by fishing. Equation 11 is a first-order autoregressive process (e.g., Box and Jenkins 1976) its variance equals to:
For small catches, such that:
we have an unstable system and therefore large oscillations.
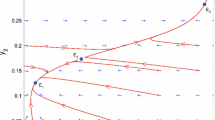
It should however be noticed that the stability limit above is for the deterministic part of the model. A nonlinear model with a noise disturbance can, if the perturbations are large enough, show a stable oscillation in the stable region but near the border of stability (se e.g., Kaitala et al. 1996). Our nonlinear stochastic model is therefore still oscillating for a slightly larger value of C than is postulated by Eq. 13. This can explain why the border of the area with the highest probability (P = 0.87) in Fig. 2b is approximately 10,000 tonnes to the right of the stability border defined by Eq. 13 (see Fig. 3b).
When:
the deterministic equilibrium value is zero (Fig. 4b).
The coefficient of variation is defined by:
The value of C that minimises CV is defined by:
C CVmin also defines the border between under- or over-compensatory endogenous dynamics of the system.
To summarise the result:
0 < C < C stab the system is unstable.
C stab < C < C CVmin the system is stable, over-compensatory and CV is decreased with increased fishing.
C CVmin < C < C zero the system is stable, under-compensatory and CV is increased with increased fishing.
C > C zero the systems equilibrium value is negative which corresponds to an extinct population.
Model convergence
To test model convergence we first run simulations with two different start values for the model parameters: α 1–α 4:
Chain 1: α 1 = 0.72, α 2 = −0.0020, α 3 = −0.41, α 4 = 0.20
Chain 2: α 1 = 2.00, α 2 = −0.0050, α 3 = −1.0, α 4 = 0.10
The resulting chains can be seen in Fig. 5. The two chains look similar for all four parameters (no tendency of divergence).
The second convergence test was a Gelman test (Brook and Gelman 1998). We used three different start values for the model parameters α 1–α 4, σ obs and σ proc. The potential scale reduction factor for the model parameters were:
α 1 = 0.99995, α 2 = 1.0001, α 3 = 1, α 4 = 1, σ obs = 1.0006, σ proc = 1.0003
A value near 1.0 for the potential scale factor indicates that the model has converged.
The third test was to check the sensibility for choice of prior distributions. We tested two additional distributions for σ p and σ m: half-t 5 priors (original) uniform (0, 10) (test1) and inverse gamma (0.001, 0.001) (test2) (Table 2).
Our conclusion is that all three tests indicate that the model has converged.
Rights and permissions
About this article
Cite this article
Wikström, A., Knape, J., Casini, M. et al. Fishing, reproductive volume and regulation: population dynamics and exploitation of the eastern Baltic cod. Popul Ecol 58, 199–211 (2016). https://doi.org/10.1007/s10144-015-0520-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10144-015-0520-3