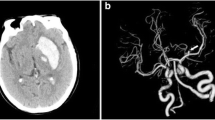
Abstract
Additional space-occupying intracerebral hematoma (ICH) in patients suffering from subarachnoid hemorrhage (SAH) is a known predictor for poor outcome. Emergent clot evacuation might be mandatory. However, data concerning the influence of ICH location on outcome is scarce. Therefore, we analyzed the influence of ICH location on clinical course and outcome in patients with SAH and additional ICH. One hundred seventy-four patients were treated with aneurysmal SAH and additional ICH between September 1999 and May 2012. Information including patient characteristics, treatment, and radiological findings were prospectively entered into a database. Patients were stratified according to ICH location and neurological outcome. Neurological outcome was assessed according to modified Rankin Scale (mRS). ICH location was temporal (58.6 %), frontal (28.7 %), and perisylvian ICH (12.6 %); 63.8 % presented in poor admission status and favorable outcome was achieved in 35.6 %. In the multivariate analysis, favorable outcome was associated with young age, ICH <50 ml, and good admission status. The location of ICH was not associated with outcome. The current data confirms that a significant number of patients with ICH after aneurysm rupture achieve favorable outcome. Prognostic factor for favorable outcome are “age,” “size of the hematoma,” and “admission status.” The location of the ICH seems not to be associated with outcome.


Similar content being viewed by others
References
Abbed KM, Ogilvy CS (2003) Intracerebral hematoma from aneurysm rupture. Neurosurg Focus 15(4):E4
Brandt L et al (1987) Ruptured middle cerebral artery aneurysm with intracerebral hemorrhage in younger patients appearing moribund: emergency operation? Neurosurgery 20(6):925–929
Crompton MR (1962) Intracerebral haematoma complicating ruptured cerebral berry aneurysm. J Neurol Neurosurg Psychiatry 25:378–386
D’Ambrosio AL et al (2005) Decompressive hemicraniectomy for poor-grade aneurysmal subarachnoid hemorrhage patients with associated intracerebral hemorrhage: clinical outcome and quality of life assessment. Neurosurgery 56(1):12–19, discussion 19–20
de los Reyes K et al (2013) Management of subarachnoid hemorrhage with intracerebral hematoma: clipping and clot evacuation versus coil embolization followed by clot evacuation. J Neurointerv Surg 5(2):99–103
Guresir E et al (2008) Subarachnoid hemorrhage and intracerebral hematoma: incidence, prognostic factors, and outcome. Neurosurgery 63(6):1088–1093, discussion 1093–1084
Güresir E et al (2009) Decompressive hemicraniectomy in subarachnoid haemorrhage: the influence of infarction, haemorrhage and brain swelling. J Neurol Neurosurg Psychiatry 80(7):799–801
Güresir E et al (2011) Rapid closure technique in decompressive craniectomy. J Neurosurg 114(4):954–960
Hauerberg J et al (1994) The prognostic significance of intracerebral haematoma as shown on CT scanning after aneurysmal subarachnoid haemorrhage. Br J Neurosurg 8(3):333–339
Hoffmann M et al (2012) Pupil evaluation in addition to Glasgow Coma Scale components in prediction of traumatic brain injury and mortality. Br J Surg 99(Suppl 1):122–130
Kazumata K et al (2010) Poor-grade ruptured middle cerebral artery aneurysm with intracerebral hematoma: bleeding characteristics and management. Neurol Med Chir 50(10):884–892
Liu X, Rinkel GJ (2011) Aneurysmal and clinical characteristics as risk factors for intracerebral haematoma from aneurysmal rupture. J Neurol 258(5):862–865
Nakagawa T et al (2005) Predicting the overall management outcome in patients with a subarachnoid hemorrhage accompanied by a massive intracerebral or full-packed intraventricular hemorrhage: a 15-year retrospective study. Surg Neurol 63(4):329–334, discussion 334–325
Niemann DB et al (2003) Treatment of intracerebral hematomas caused by aneurysm rupture: coil placement followed by clot evacuation. J Neurosurg 99(5):843–847
Niikawa S et al (1998) Significance of acute cerebral swelling in patients with sylvian hematoma due to ruptured middle cerebral artery aneurysm, and its management. Neurol Med Chir (Tokyo) 38(12):844–848, discussion 849–850
Nowak G et al (1998) Intracerebral hematomas caused by aneurysm rupture. Experience with 67 cases. Neurosurg Rev 21(1):5–9
Otani N et al (2008) Surgical outcome following decompressive craniectomy for poor-grade aneurysmal subarachnoid hemorrhage in patients with associated massive intracerebral or Sylvian hematomas. Cerebrovasc Dis 26(6):612–617
Pasqualin A et al (1986) Intracranial hematomas following aneurysmal rupture: experience with 309 cases. Surg Neurol 25(1):6–17
Rosengart AJ et al (2007) Prognostic factors for outcome in patients with aneurysmal subarachnoid hemorrhage. Stroke 38(8):2315–2321
Sakas DE et al (1995) One-year outcome following craniotomy for traumatic hematoma in patients with fixed dilated pupils. J Neurosurg 82(6):961–965
Shim YS et al (2012) Grading of intracerebral hemorrhage in ruptured middle cerebral artery aneurysms. J Korean Neurosurg Soc 51(5):268–271
Shimoda M et al (1997) Surgical indications in patients with an intracerebral hemorrhage due to ruptured middle cerebral artery aneurysm. J Neurosurg 87(2):170–175
Smith ER et al (2002) Proposed use of prophylactic decompressive craniectomy in poor-grade aneurysmal subarachnoid hemorrhage patients presenting with associated large sylvian hematomas. Neurosurgery 51(1):117–124, discussion 124
Tapaninaho A et al (1988) Emergency treatment of cerebral aneurysms with large haematomas. Acta Neurochir (Wien) 91(1–2):21–24
Tawk RG et al (2010) Coiling of ruptured aneurysms followed by evacuation of hematoma. World Neurosurg 74(6):626–631
Tokuda Y et al (1995) Intracerebral hematoma in patients with ruptured cerebral aneurysms. Surg Neurol 43(3):272–277
Yoshimoto Y et al (1999) Intraparenchymal and intrasylvian haematomas secondary to ruptured middle cerebral artery aneurysms: prognostic factors and therapeutic considerations. Br J Neurosurg 13(1):18–24
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Lotfi Hacein-Bey, Sacramento, USA
The article by Bruder et al. in this issue of Neurosurgical Review underscores a number of challenges encountered in managing patients with cerebrovascular hemorrhage. This large series from a recognized academic European center confirms that the best predictors of good outcomes in patients with SAH complicated by ICH are young age, good grade on admission, and absence of complicating signs, i.e., herniation. Conversely, the factors which had the greatest impact on outcome were poor grade upon admission, surgical versus endovascular management of the ruptured aneurysm, and hematoma location (perisylvian location being the worst).
The role of clinical grade upon admission, the effects of SAH, and the differences between treatment techniques have been amply recognized.
However, the impact of cerebral hematomas is less clearly appreciated.
First, hematomas alone may result in cerebral edema, tissue damage, and increased vascularity as macrophage infiltration and astrocyte proliferation around hematomas produce secretory factors such as vascular endothelial growth factor (VEGF), matrix metallopeptidase 9 (MMP-9), various interleukins, tumor necrosis factor-a, and aquaporin water channels. VEGF induces angiogenesis and neo-vascularity in the wall of the hematoma, seen as ring-enhancing lesions on imaging studies. MMPs degrade extracellular matrix proteins, including the neurovascular basal lamina and tight junction proteins of the blood-brain barrier (BBB). BBB disruption results in edema formation. Therefore, hematoma removal not only results in alleviating mass effect, but also reduces brain edema, tissue damage, and increased risk of re-hemorrhage.
Another major fact about hematomas is that they may induce derangements in brain function by causing damage to neural networks. Frontal and temporal hematomas may affect major brain networks such as the default mode network (medial prefrontal cortex, posterior cingulum, and medial temporal lobe) and the central executive network (dorsolateral prefrontal cortex). The observation that perisylvian hemorrhage was associated with particularly poor outcomes in this series may be explained in part by the proximity to deep nuclei and eloquent midline structures. However, an important reason for this phenomenon may be the disruption of structural connectivity between the insula and other brain regions. The insular cortex has been recently described as the “limbic integration cortex” after anatomical connectivity studies have shown major efferent and afferent connections to the amygdala, olfactory cortex, or cingulum. A major neural network, the salience network, which consists of three main cortical areas (the dorsal anterior cingulate cortex, the anterior right insula, and the left insula) plays an important role in the initiation of cognitive control, the implementation of tasks, and the coordination of behavioral responses. As continued progress is made in noninvasive mapping of white matter pathways around the insular cortex, there is no doubt that the importance of the salience network in integrating physiological control and monitoring emotional life will be further elucidated.
This article is a useful and humbling reminder of how difficult the task of a surgeon is and how little we still know about brain functional networks.
Andreas Raabe, Bern, Switzerland
The prognosis of patients with spontaneous intracerebral hemorrhage (ICH) is usually poor, and we would expect that a combination of ICH and subarachnoid hemorrhage (SAH) further deteriorates the chance of a favorable outcome. Patients with ICH and SAH usually present in a poorer clinical admission state and with a more dramatic clinical picture. Thus, we would again expect a rather poor outcome, given the fact, that the initial presentation influences the prognosis considerably. However, the present study demonstrated that a “combined” poor outcome effect of SAH and ICH is less likely. It favors an active management with surgical removal of a space-occupying hematoma and aneurysm clipping instead of a more pessimistic ICH-like approach.
Rights and permissions
About this article
Cite this article
Bruder, M., Schuss, P., Berkefeld, J. et al. Subarachnoid hemorrhage and intracerebral hematoma caused by aneurysms of the anterior circulation: influence of hematoma localization on outcome. Neurosurg Rev 37, 653–659 (2014). https://doi.org/10.1007/s10143-014-0560-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-014-0560-8