Abstract
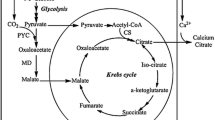
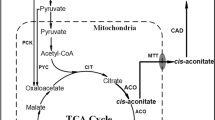
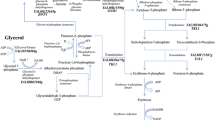
In this study, a pyruvate carboxylase gene (PYC1) from a marine fungus Penicillium rubens I607 was cloned and characterized. ORF of the gene (accession number: KM397349.1) had 3534 bp encoding 1177 amino acids with a molecular weight of 127.531 kDa and a PI of 6.20. The promoter of the gene was located at −1200 bp and contained a TATAA box, several CAAT boxes and a sequence 5′-SYGGRG-3′. The PYC1 deduced from the gene had no signal peptide, was a homotetramer (α4), and had the four functional domains. After expression of the PYC1 gene from the marine fungus in the marine-derived yeast Yarrowia lipolytica SWJ-1b, the transformant PR32 obtained had much higher specific pyruvate carboxylase activity (0.53 U/mg) than Y. lipolytica SWJ-1b (0.07 U/mg), and the PYC1 gene expression (133.8 %) and citric acid production (70.2 g/l) by the transformant PR32 were also greatly enhanced compared to those (100 % and 27.3 g/l) by Y. lipolytica SWJ-1b. When glucose concentration in the medium was 60.0 g/l, citric acid (CA) concentration formed by the transformant PR32 was 36.1 g/l, leading to conversion of 62.1 % of glucose into CA. During a 10-l fed-batch fermentation, the final concentration of CA was 111.1 ± 1.3 g/l, the yield was 0.93 g/g, the productivity was 0.46 g/l/h, and only 1.72 g/l reducing sugar was left in the fermented medium within 240 h. HPLC analysis showed that most of the fermentation products were CA. However, minor malic acid and other unknown products also existed in the culture.








Similar content being viewed by others
References
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL Workspace: a web-based environment for protein structure homology modeling. Bioinformatics 2:195–201
Arzumanov T, Shishkanova N, Finogenova T (2000) Biosynthesis of citric acid by Yarrowia lipolytica repeat-batch culture on ethanol. Appl Microbiol Biotechnol 53(5):525–529
Bologna FP, Andreo CS, Drincovich MF (2007) Escherichia coli malic enzymes: two isoforms with substantial differences in kinetic properties, metabolic regulation, and structure. J Bacteriol 189:5937
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–253
Camp BJ, Farmer L (1967) A rapid spectrophotometric method for the determination of citric acid in blood. Clin Chem 13:501–505
Chi ZM, Liu J, Zhang W (2001) Trehalose accumulation from soluble starch by Saccharomycopsis fibuligera sdu. Enzym Microb Technol 28:240–245
Chi Z, Wang ZP, Wang GY, Khan I, Chi ZM (2014) Microbial biosynthesis and secretion of L-malic acid and its applications. Crit Rev Biotechnol doi:10.3109/07388551.2014.924474
Forster A, Aurich A, Mauersberger S, Barth G (2007a) Citric acid production from sucrose using a recombinant strain of the yeast Yarrowia lipolytica. Appl Microbiol Biotechnol 75:1409–1417
Forster A, Jacobs K, Juretzek T, Mauersberger S, Barth G (2007b) Overexpression of the ICL1 gene changes the product ratio of citric acid production by Yarrowia lipolytica. Appl Microbiol Biotechnol 77:861–869
Gancedo JM (1998) Yeast carbon catabolite repression. Microbiol Mol Biol Rev 62:334–361
Ikram-ul H, Khurshid S, Ali S, Ashraf H, Qadeer MA, Rajoka MI (2001) Mutation of Aspergillus niger for hyperproduction of citric acid from black strap molasses. World J Microbiol Biotechnol 17(1):35–37
Ikram-ul H, Ali S, Qadeer MA, Javed I (2004) Citric acid production by selected mutants of Aspergillus niger from cane molasses. Bioresour Technol 93(2):125–130
Jitrapakdee S, St Maurice M, Rayment I, Cleland WW, Wallace JC, Wallace JC, Attwood PV (2008) Structure, mechanism and regulation of pyruvate carboxylase. Biochem J 413:369–387
Kamzolova SV, Fatykhova AR, Dedyukhina EG, Anastassiadis SG, Golovchenko NP, Morgunov IG (2011) Citric acid production by yeast grown on glycerol-containing waste from biodiesel industry. Food Technol Biotechnol 49(1):65
Karaffa L, Kubicek CP (2003) Aspergillus niger citric acid accumulation: do we understand this well working black box? Appl Microbiol Biotechnol 61:189–196
Khan I, Nazir K, Wang ZP, Liu GL, Chi ZM (2014) Calcium malate overproduction by Penicillium viticola 152 using the medium containing corn steep liquor. Appl Microbiol Biotechnol 98:1539–1546
Lietzan AD, Maurice MS (2013) Insights into the carboxyltransferase reaction of pyruvate carboxylase from the structures of bound product and intermediate analogs. Biochem Biophys Res Commun 441:377–382
Lin H, San KY, Bennett GN (2005) Effect of Sorghum vulgare phosphoenolpyruvate carboxylase and Lactococcus lactis pyruvate carboxylase coexpression on succinate production in mutant strains of Escherichia coli. Appl Microbiol Biotechnol 67:515–523
Liu XY, Chi Z, Liu GL, Wang F, Madzak C, Chi ZM (2010) Inulin hydrolysis and citric acid production from inulin using the surface-engineered Yarrowia lipolytica displaying inulinase. Metab Eng 12:469–476
Liu GL, Wang DS, Wang LF, Zhao SF, Chi ZM (2011) Mig1 is involved in mycelial formation and expression of the genes encoding extracellular enzymes in Saccharomycopsis fibuligera A11. Fungal Genet Biol 48:904–913
Liu XY, Chi Z, Liu GL, Madzak C, Chi ZM (2013) Both decrease in ACL1 gene expression and increase in ICL1 gene expression in marine-derived yeast Yarrowia lipolytica expressing INU1 gene enhance citric acid production from inulin. Mar Biotechnol 15:26–36
Liu X, Lv J, Zhang T, Deng Y (2014) Direct conversion of pretreated straw cellulose into citric acid by co-cultures of Yarrowia lipolytica: SWJ-1b and immobilized Trichoderma reesei mycelium. Appl Biochem Biotechnol 173(2):501–509
Liu X, Lv J, Xu J, Zhang T, Deng Y, He J (2015) Citric acid production in Yarrowia lipolytica: SWJ-1b yeast when grown on waste cooking oil. Appl Biochem Biotechnol 175(5):2347–2356
Makri A, Fakas S, Aggelis G (2010) Metabolic activities of biotechnological interest in Yarrowia lipolytica grown on glycerol in repeated batch cultures. Bioresour Technol 101:2351–2358
Mourya S, Jauhri K (2000) Production of citric acid from starch-hydrolysate by Aspergillus niger. Microbiol Res 155(1):37–44
Ochoa-Estopier A, Guillouet SE (2014) D-stat culture for studying the metabolic shifts from oxidative metabolism to lipid accumulation and citric acid production in Yarrowia lipolytica. J Biotechnol 170:35–41
Otto C, Yovkova V, Aurich A, Mauersberger S, Barth G (2012) Variation of the by-product spectrum during α-ketoglutaric acid production from raw glycerol by overexpression of fumarase and pyruvate carboxylase genes in Yarrowia lipolytica. Appl Microbiol Biotechnol 95:905–917
Papanikolaou S, Galiotou-Panayotou M, Chevalot I, Komaitis M, Marc I, Aggelis G (2006) Influence of glucose and saturated free-fatty acid mixtures on citric acid and lipid production by Yarrowia lipolytica. Curr Microbiol 52(2):134–142
Papanikolaou S, Galiotou-Panayotou M, Fakas S, Komaitis M, Aggelis G (2008) Citric acid production by Yarrowia lipolytica: cultivated on olive-mill wastewater-based media. Biores Technol 99(7):2419–2428
Papanikolaou S, Chatzifragkou A, Fakas S, Galiotou-Panayotou M, Komaitis M, Nicaud JM, Aggelis G (2009) Biosynthesis of lipids and organic acids by Yarrowia lipolytica trains cultivated on glucose. Eur J Lipid Sci Technol 111:1221–1232
Papanikolaou S, Beopoulos A, Koletti A, Thevenieau F, Koutinas AA, Nicaud JM, Aggelis G (2013) Importance of the methyl-citrate cycle on glycerol metabolism in the yeast Yarrowia lipolytica. J Biotechnol 168:303–314
Peksel A, Torres NV, Liu J, Juneau G (2002) 13C-NMR analysis of glucose metabolism during acid production by Aspergillus niger. Appl Microbiol Biotechnol 58:157–163
Peters-Wendisch PG, Schiel B, Wendisch VF, Katsoulidis E, Möckel B, Sahm H, Eikmanns BJ (2001) Pyruvate carboxylase is a major bottleneck for glutamate and lysine production by Corynebacterium glutamicum. J Mol Microbiol Biotechnol 3:295–300
Ratledge C, Wynn JP (2002) The biochemistry and molecular biology of lipid accumulation in oleaginous microorganisms. Adv Appl Microbiol 51:1–51
Reinwald S, Weaver CM, Kester JJ (2008) The health benefits of calcium citrate malate: a review of the supporting science. Adv Food Nutr Res 54:219–346
Rywinska A, Rymowicz W, Zarowska B, Wojtatowicz M (2009) Biosynthesis of citric acid from glycerol by acetate mutants of Yarrowia lipolytica in fed-batch fermentation. Food Technol Biotechnol 47(1):1–6
Rywińska A, Rymowicz W (2010) High-yield production of citric acid by Yarrowia lipolytica: on glycerol in repeated-batch bioreactors. J Ind Microbiol Biotechnol 37(5):431–435
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Beijing, pp 367–370 (Chinese translated edn)
Shojaosadati SA, Babaeipour V (2002) Citric acid production from apple pomace in multi-layer packed bed solid-state bioreactor. Proc Biochem 37(8):909–914
Spiro RG (1966) Analysis of sugars found in glycoproteins. Methods Enzymol 8:3–26
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 24:4876–4882
Tomaszewska L, Rakicka M, Rymowicz W, Rywińska A (2014) A comparative study on glycerol metabolism to erythritol and citric acid in Yarrowia lipolytica yeast cells. FEMS Yeast Res 14(6):966–976
Tran C, Sly L, Mitchell D (1998) Selection of a strain of Aspergillus for the production of citric acid from pineapple waste in solid-state fermentation. World J Microbiol Biotechnol 4(3):399–404
Wang F, Yue LX, Wang L, Madzak C, Li J, Wang XH, Chi ZM (2009) Genetic modification of the marine-derived yeast Yarrowia lipolytica with high-protein content using a GPI-anchor-fusion expression system. Biotechnol Prog 25:1297–1303
Wang ZP, Wang GY, Khan I, Chi ZM (2013a) High-level production of calcium malate from glucose by Penicillium sclerotiorum K302. Bioresour Technol 143:674–677
Wang LF, Wang ZP, Liu XY, Chi ZM (2013b) Citric acid production from extract of Jerusalem artichoke tubers by the genetically engineered yeast Yarrowia lipolytica: strain 30 and purification of citric acid. Bioprocess Biosyst Eng 36(11):1759–1766
Wang GY, Zhang Y, Chi Z, Liu GL, Wang ZP, Chi ZM (2015a) Role of pyruvate carboxylase in accumulation of intracellular lipid of the oleaginous yeast Yarrowia lipolytica ACA-DC 50109. Appl Microbiol Biotechnol 99:1637–1645
Wang L, Zhang J, Cao Z, Wang Y, Gao Q, Zhang J, Wang D (2015b) Inhibition of oxidative phosphorylation for enhancing citric acid production by Aspergillus niger. Microb Cell Fact 14(1):1–12
Xuan JM, Fournier P, Gaillardin C (1988) Cloning of the LYS5 gene encoding saccharopine dehydrogenase. Curr Genet 14:15–21
Yin X, Madzak C, Du G, Zhou J, Chen J (2012) Enhanced alpha-ketoglutaric acid production in Yarrowia lipolytica WSH-Z06 by regulation of the pyruvate carboxylation pathway. Appl Microbiol Biotechnol 96:1527–1537
Zelle RM, de Hulster E, van Winden WA (2008) Malic acid production by Saccharomyces cerevisiae: engineering of pyruvate carboxylation, oxaloacetate reduction, and malate export. Appl Environ Microbiol 74:2766–2777
Zhang F, Wang ZP, Chi Z, Raoufi Z, Abdollahi S, Chi ZM (2013) The changes in Tps1 activity, trehalose content and expression of TPS1 gene in the psychrotolerant yeast Guehomyces pullulans 17-1 grown at different temperatures. Extremophiles 17:241–249
Zhou J, Zhou H, Du G, Liu L, Chen J (2010) Screening of a thiamine-auxotrophic yeast for α-ketoglutaric acid overproduction. Lett Appl Microbiol 51:264–271
Acknowledgments
This research was supported by Hi-Tech Research and Development Program of China (863) (grant no. 2012AA021205).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file
(DOC 533 kb)
Rights and permissions
About this article
Cite this article
Fu, GY., Lu, Y., Chi, Z. et al. Cloning and Characterization of a Pyruvate Carboxylase Gene from Penicillium rubens and Overexpression of the Genein the Yeast Yarrowia lipolytica for Enhanced Citric Acid Production. Mar Biotechnol 18, 1–14 (2016). https://doi.org/10.1007/s10126-015-9665-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-015-9665-5