Abstract
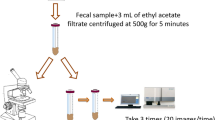
The purpose of this study was to evaluate the level of agreement of the BD Max™ Enteric Parasite Panel (EPP) with microscopy for the detection of Giardia duodenalis, Cryptosporidium spp. and Entamoeba histolytica in stool samples. A total of 372 stool samples (partly collected on the basis of positive microscopy and partly unselected, consecutive sample submitted for parasite investigation) were tested with EPP according to manufacturer’s instructions and also using microscopy according to standard techniques. Discrepant samples were further tested using PCR by the National Parasitology reference laboratory. Levels of agreement and laboratory turnaround times were measured and compared. Overall, positive and negative percent agreement was high between the two methods. However, microscopy resulted in four false positives and one false negative for G. duodenalis and two false positives for Cryptosporidium. Additionally, microscopy could not differentiate between E. histolytica and Entamoeba dispar. Median laboratory turnaround time was 65 hours for microscopy; results from EPP could be available after four hours. Blastocycstis hominis was detected by microscopy in one sample and would have been missed if only EPP was performed. The EPP was a good alternative to microscopy, detecting a small number of additional positives that were missed by microscopy. The assay is significantly faster than microscopy and allows laboratory workflows to be streamlined. The risk of missing parasites that are not included in the EPP appears to be minimal in the studied population; however, there may be certain patient groups who would benefit from microscopic examination of stools.
Similar content being viewed by others
References
van Lieshout L, Roestenberg M (2015) Clinical consequences of new diagnostic tools for intestinal parasites. Clin Microbiol Infect 21:520–8
Halligan E, Edgeworth J, Bisnauthsing K et al (2014) Multiplex molecular testing for management of infectious gastroenteritis in a hospital setting: A comparative diagnostic and clinical utility study. Clin Microbiol Infect 20:O460–7
Zhang H, Morrison S, Tang YW (2015) Multiplex polymerase chain reaction tests for detection of pathogens associated with gastroenteritis. Clin Lab Med 35:461–486
Perry MD, Corden SA, Howe RA (2014) Evaluation of the luminex xTAG gastrointestinal pathogen panel and the Savyon diagnostics gastrointestinal infection panel for the detection of enteric pathogens in clinical samples. J Med Microbiol 63:1419–26
Biswas JS, Al-Ali A, Rajput P, Smith D, Goldenberg SD (2014) A parallel diagnostic accuracy study of three molecular panels for the detection of bacterial gastroenteritis. Eur J Clin Microbiol Infect Dis 33:2075–81
Verweij JJ, Stensvold CR (2014) Molecular testing for clinical diagnosis and epidemiological investigations of intestinal parasitic infections. Clin Microbiol Rev 27:371–418
Binnicker MJ (2015) Multiplex molecular panels for diagnosis of gastrointestinal infection: Performance, result interpretation, and cost-effectiveness. J Clin Microbiol 53:3723–8
Mölling P, Nilsson P, Ennefors T et al (2016) Evaluation of the BD max enteric parasite panel for clinical diagnostics. J Clin Microbiol 54:443–4
Knabl L, Grutsch I, Orth-Höller D (2016) Comparison of the BD MAX® enteric bacterial panel assay with conventional diagnostic procedures in diarrheal stool samples. Eur J Clin Microbiol Infect Dis 35:131–6
Harrington SM, Buchan BW, Doern C et al (2015) Multicenter evaluation of the BD Max enteric bacterial panel PCR assay for rapid detection of Salmonella spp., Shigella spp., Campylobacter spp. (C. Jejuni and C. Coli), and shiga toxin 1 and 2 genes. J Clin Microbiol 53:1639–47
Public Health England (2014) UK Standards for Microbiology Investigations B31: Investigation of specimens other than blood for parasites. Public Health England, London
Verweij JJ, Blangé RA, Templeton K et al (2004) Simultaneous detection of Entamoeba histolytica, Giardia lamblia, and Cryptosporidium parvum in fecal samples by using multiplex real-time PCR. J Clin Microbiol 42:1220–3
Coyle CM, Varughese J, Weiss LM, Tanowitz HB (2012) Blastocystis: To treat or not to treat. Clin Infect Dis 54:105–10
Stensvold CR, Nielsen HV (2011) Comparison of microscopy and PCR for the detection of intestinal parasites in Danish patients supports incentive for molecular screening platforms. J Clin Microbiol 50:540–1
van Lieshout L, Verweij JJ (2010) Newer diagnostic approaches to intestinal protozoa. Curr Opin Infect Dis 23:488–93
Bruijnesteijn van Coppenraet LE, Wallinga JA, Ruijs GJ, Bruins MJ, Verweij JJ (2009) Parasitological diagnosis combining an internally controlled real-time PCR assay for the detection of four protozoa in stool samples with a testing algorithm for microscopy. Clin Microbiol Infect 15:869–74
Schreckenberger PC, McAdam AJ (2015) Point-Counterpoint: Large multiplex PCR panels should be first-line tests for detection of respiratory and intestinal pathogens. J Clin Microbiol 53:3110–5
Acknowledgments
We are grateful to BD who provided consumables and platform free of charge for this evaluation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Consumables were provided free of charge from BD, otherwise no specific funding was obtained for this study. RB receives funding from the NIHR Collaboration for Leadership in Applied Health Research and Care (CLAHRC) South London.
Conflict of interest
Simon Goldenberg reports speakers fees from BD; there were no other conflicts of interest from other authors.
Ethical approval and informed consent
All samples were residual and fully anonymised; research ethics approval and informed consent was not required.
Rights and permissions
About this article
Cite this article
Batra, R., Judd, E., Eling, J. et al. Molecular detection of common intestinal parasites: a performance evaluation of the BD Max™ Enteric Parasite Panel. Eur J Clin Microbiol Infect Dis 35, 1753–1757 (2016). https://doi.org/10.1007/s10096-016-2722-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-016-2722-9