Abstract
Bronchoalveolar lavage (BAL) is a major diagnostic tool in lung diseases, including viral respiratory infections. We aimed to better define the situations where viral tests should be performed on BAL fluid (BALF). We retrospectively studied all cases where viral tests [immunofluorescence, immunocytochemistry, viral culture, and/or polymerase chain reaction (PCR)] were performed on BALF during a period of 1 year (2008) in our institution. We compared the characteristics of patients with virus-positive versus virus-negative BALF. Of the 636 BALF samples sent to the microbiology laboratory, 232 underwent viral tests. Of these, 70 (30 %) were positive and identified 85 viruses: herpes simplex virus (HSV)-1 (n = 27), cytomegalovirus (CMV, n = 23), Epstein–Barr virus (EBV, n = 18), human herpesvirus (HHV)-6 (n = 12), respiratory syncytial virus (RSV, n = 3), rhinovirus (n = 1), and adenovirus (n = 1). The variables associated with positive viral tests on univariate analysis were immunosuppression [human immunodeficiency virus (HIV), corticosteroids >10 mg/day for ≥3 weeks, or other immunosuppressive therapy], ground-glass attenuations on computed tomography (CT) scanning, late-onset ventilator-associated pneumonia (VAP), and durations of (i) hospital stay, (ii) intensive care unit (ICU) stay, and (iii) mechanical ventilation before BAL (p < 0.01 for each comparison). On multivariate analysis, only immunosuppression [odds ratio (OR) 6.4, 95 % confidence interval (CI) [2.8–14.3], p < 0.0001] and ground-glass attenuations (OR 3.7, 95 % CI [1.8–7.7], p = 0.0004) remained associated with virus-positive BAL. None of the viral tests performed on BALF for the initial assessment of diffuse infiltrative lung disease (n = 15) was positive. PCR improved the diagnostic yield of viral tests on BALF by 50 %. Testing for viruses on BALF should be mostly restricted to immunocompromised patients with acute respiratory diseases and/or patients with unexplained ground-glass attenuations on CT scanning.
Similar content being viewed by others

Introduction
Bronchoalveolar lavage (BAL) is a major diagnostic tool for infectious lung diseases, especially in immunocompromised patients [1]. Viral agents play an important role as etiologies of pneumonia in immunocompetent and immunocompromised hosts [2–5], and may be involved in a substantial proportion of asthma and chronic obstructive pulmonary disease (COPD) exacerbations [6–9]. The development of molecular biology techniques, including polymerase chain reaction (PCR), has dramatically increased the yield of viral tests for various respiratory diseases [2, 4, 5, 8]. However, most studies have been performed in specific settings, with highly selected patients. Hence, the yield of viral tests on BAL fluid (BALF) remains poorly characterized in routine practice. A key dilemma with the development of new tests is the relatively high cost associated with their non-discriminated use. To better inform the use of viral tests in patients with respiratory diseases, we performed an observational, retrospective study in our institution, with three aims: (i) to assess the diagnostic value of viral tests on BALF in routine practice; (ii) to analyze the characteristics of patients with virus-positive BALF; and (iii) to identify the factors predictive of positive viral tests in BALF.
Materials and methods
Patients
Pontchaillou University Hospital is a 1,819-bed tertiary-care hospital which serves as a referral center for the area of Rennes, France. All adults (≥18 years of age) who had viral test(s) performed on BALF during the year 2008 were included. Cases were identified through the computerized database in the virology department. Data were extracted from this database and from the medical records using a standardized questionnaire. The protocol was approved by our institutional review board (Rennes University Hospital Ethics Committee, project approval number 12.10), and informed consent was waived.
Data collection
The data collected included demographic information and co-morbidities. Alcohol abuse was defined as daily consumption >3 units/day for men or >2 units/day for women. Patients were classified as immunocompromised if they were infected with human immunodeficiency virus (HIV), were on systemic corticosteroids ≥10 mg/day for at least 3 weeks [10], or were on any immunosuppressive drug on admission. Medical charts were checked for the following signs: fever >38 °C, cough, purulent sputum, hemoptysis, chest pain, dyspnea, and crackles on chest examination. Radiographic patterns prior to BAL were defined by the type of consolidation (alveolar or interstitial, nodules, micronodules, ground-glass attenuations, bronchiectasis, or excavation). Other data collected included any antimicrobial treatment received during the week preceding BAL, mechanical ventilation, intensive care unit (ICU) admission, and in-hospital mortality.
Bronchoalveolar lavage (BAL)
BAL was performed according to the guidelines [11, 12], under local anesthetic (lidocaine spray): 30–50 mL of sterile saline solution was instilled 2–4 times into the distal bronchial tree, either at the site where radiographic abnormalities predominated or in the middle lobe in cases with diffuse radiographic pattern. BALF specimens were aliquoted and immediately transported to laboratories. Appropriate staining was carried out for the direct identification of bacteria, mycobacteria, fungi, and parasites. Cultures for bacterial identification were inoculated under standard aerobic conditions on four different media, as well as on specific media for Mycobacterium spp., when indicated. For the usual respiratory pathogens, the bacterial count was considered to be significant when quantitative culture yielded >103 CFU/mL of specimen. In addition, tests for atypical pneumonia agents were performed at the request of the physician in charge, on BALF (i.e., PCR, immunofluorescence, specific staining, and culture on appropriate media) and by serology.
Viral tests
The first sample of BALF was used for viral tests, as it is the most likely to contain significant numbers of epithelial cells. The sample was mixed (volume 1:1) with viral transport medium, i.e., minimum essential medium enriched with sorbitol (70 %), bovine serum albumin, streptomycin, vancomycin, colimycin, and amphotericin B. BALF was first analyzed by immunofluorescence (IF) with a panel of monoclonal antibodies against respiratory syncytial virus (RSV), A and B influenza viruses, 1–3 parainfluenza viruses, and adenovirus, as well as herpes simplex viruses (HSV)-1 and -2, in immunocompromised patients. In parallel, BALF was inoculated onto MRC-5 and LLC-MK2 monolayer cells, and also onto MDCK cells during the influenza season. In immunocompromised patients, an immunocytochemistry (ICC) for cytomegalovirus (CMV) was performed. The detection of viral genomes was carried out according to the clinical context and a request from the physician in charge. HSV-1, HSV-2, CMV, varicella zoster virus (VZV), Epstein–Barr virus (EBV), and human herpesvirus (HHV)-6 were detected using a commercial method (Herpes Consensus®, Argène, France). In addition, CMV, HSV-1, HSV-2, EBV, HHV-6, and influenza A and B were detected with in-house PCR, adapted from previously described methods [13]. Neither PCR testing on nasopharyngeal swab/aspirates nor viral serology tests were routinely performed during the study period in our institution.
Statistical analyses
Statistical analyses were performed using SAS Software 9.0 (SAS Institute Inc., Cary, NC, USA). The results are presented as the mean ± standard deviation, with the range in parentheses. We compared the characteristics of patients with virus-positive versus virus-negative BALF using Student’s t-test (n ≥ 30) or the Mann–Whitney test (n < 30) for quantitative variables, and Fisher’s exact test for categorical variables. Multiple comparisons were performed using analysis of variance with a Bonferroni post hoc correction. Multivariate analysis was performed using logistic regression models, after adjustment for the duration of stay before BALF. Variables with a p-value < 0.2 in univariate analysis were entered into the multivariate analysis. Values of p < 0.05 were regarded as significant.
Results
Patients and procedures
In 2008, 636 BALF samples were sent to the microbiology department in our institution. Of these, viral tests were ordered in 232 BALF, from 212 patients (Fig. 1). The mean age of the patients was 54.4 ± 15.6 years (range, 19.2–89.8) and the male-to-female ratio was 1.7 (132/80). Symptoms on admission included dyspnea (71.4 %), cough (58.5 %), fever (53.9 %, with a mean body temperature of 38.9 ± 0.6 °C (range, 38.1–41.7), purulent sputum (18.3 %), and hemoptysis (4.3 %). Chest radiographic findings included consolidations (61.2 %), nodules (15.1 %), and excavation (3.9 %). Chest computed tomography (CT) scanning, performed in 157 patients, indicated ground-glass attenuations (57.9 %), micronodules (29.3 %), and bronchiectasis (10.8 %). For 108 procedures (50.9 %), BAL was performed in an ICU, including 85 procedures performed under mechanical ventilation. The mean volume of sterile saline serum instilled during the BAL procedure was 90 ± 30 mL (range, 30–200).
Diagnostic yield of viral tests on bronchoalveolar lavage (BAL). Co-morbidities include chronic heart failure, chronic obstructive pulmonary disease (COPD), hepatic, renal, or neurological disease, cancer, diabetes mellitus, alcohol abuse (daily consumption >3 units/day for men or >2 units/day for women), smoking >10 pack-years, and drug addiction. Patients were classified as immunocompromised if they were infected with human immunodeficiency virus (HIV), were on systemic corticosteroids ≥10 mg/day for at least 3 weeks, or were on any immunosuppressive therapy
Microbiology
The use of viral transport medium was adequate for 86 % of samples. Of the 232 BAL investigated for viruses, 70 (30.1 %) identified at least one virus, for a total of 85 viruses (mean, 1.2 viruses per virus-positive BALF). Viral species identified, and the yields of the four techniques used, are detailed in Table 1. Of note, 94.1 % of viruses were members of the Herpesviridae family. The most frequent virus associations were HSV-1 + HHV-6 (n = 5) and CMV + EBV (n = 4). One HIV-infected patient with interstitial pneumonia had three herpes viruses identified in the BAL: CMV (ICC), HSV-1 (viral culture), and EBV (PCR). PCR, performed in 149 BALFs, was positive in 47 cases (31.5 %). Of these, PCR was the only test positive for virus in 25 cases, confirmed the diagnosis documented by other techniques in 14 cases (HSV-1, n = 8; CMV, n = 5; EBV + CMV, n = 1) and identified a virus different to that documented by other techniques in eight cases. Had PCR not been used, the diagnostic yield would have been 20 %. Hence, PCR improved the diagnostic yield of viral tests on BALF by 50 %.
Testing for bacteria and mycobacteria was performed in 231 BALFs (99.6 %) and was positive in 103 BALFs (46.4 %). Testing for fungi was performed in 223 BALFs (96.1 %) and was positive in 97 cases (43.5 %), including eight cases of Pneumocystis jirovecii pneumonia (3.6 %). Bacteria and/or fungi were isolated in 47 of the 70 virus-positive BALFs (67 %). In this group, the main non-viral microorganisms isolated from BALFs were Candida sp. (n = 25), Aspergillus sp. (n = 7), Streptococcus sp. (n = 7), Staphylococcus sp. (n = 7), Pseudomonas aeruginosa (n = 5), Moraxella catarrhalis (n = 3), Mycobacterium tuberculosis (n = 3), and Haemophilus influenzae (n = 3). In addition, three patients with virus-positive BALFs were diagnosed with Pneumocystis jirovecii by IF testing.
Factors associated with positive viral tests in BAL
Comparisons between virus-positive and virus-negative BALF are detailed in Table 2 (univariate analysis). The variables significantly associated with positive viral tests on univariate analysis were immunosuppression (i.e., HIV infection, corticosteroids >10 mg/day for ≥3 weeks, and/or other immunosuppressive therapy), ground-glass attenuations on chest CT scans, late-onset ventilator-associated pneumonia (VAP), and durations of (i) hospital stay, (ii) ICU stay, and (iii) mechanical ventilation before BAL was performed (p < 0.01 for each comparison). On multivariate analysis, after adjustment for the duration of stay before BAL, only immunosuppression [odds ratio (OR) 6.4, 95 % confidence interval (CI) [2.8–14.3], p < 0.0001) and ground-glass attenuations (OR 3.7, 95 % CI [1.8–7.7], p = 0.0004) remained significantly associated with virus-positive BAL.
Retrospective analysis of medical charts allowed us to classify indications for the viral analysis of BALF into eight categories (Table 3) and to estimate the diagnostic yield of viral tests in each subgroup. Striking differences were observed: for example, the proportion of virus-positive BALF was 43.3 % in immunocompromised patients, as compared to 12.2 % in immunocompetent patients (p < 0.0001). None of the 15 BAL performed for the initial assessment of diffuse infiltrative lung disease was virus-positive.
Associations between viral analysis of bronchoalveolar lavage (BAL) and outcomes
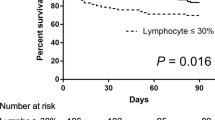
Of the 212 patients who underwent viral testing in BAL, 30 died (in-hospital mortality, 14.2 %). BALF was more frequently virus-positive in patients who died as compared to survivors (50 % [15/30] vs. 25.8 % [47/182], p = 0.0096). In patients with virus-positive BALF, bacterial or fungal co-infection was more frequent in patients who died than in survivors (93.3 % [14/15] vs. 59.6 % [28/47], p = 0.0238). Of the 70 patients with virus-positive BALF, 27 (38.6 %) received an antiviral agent: aciclovir (n = 15), ganciclovir (n = 5), valganciclovir (n = 4), or foscarnet (n = 3). ICU patients with virus-positive BALF were more likely to be treated than non-ICU-patients with virus-positive BALF (70 % vs. 30 %, p = 0.015). There was no significant association between antiviral treatment and outcome in patients with virus-positive BAL.
Discussion
This observational study evaluated the diagnostic yield of viral tests on BALF when requested by the physician in charge. Of the 232 consecutive BALF tested for viruses, 70 (30 %) were positive. Previous studies have estimated the diagnostic yield of viral tests from BALF in different settings, and the proportion of virus-positive BALF ranged from 14 to 49.5 %, depending on the population studied (e.g., immunocompromised, ICU patients) and the viral techniques used (e.g., PCR, ICC, IF, culture) [2–5, 14, 15]. A broad range of respiratory diseases have been associated with viral infections. Hence, testing for viruses in BALF may be considered in patients with a wide spectrum of clinical and radiological abnormalities [16, 17]. However, our study suggests that viral tests are unlikely to return positive except in two, non-exclusive, situations: (i) immunocompromised patients; (ii) bilateral ground-glass attenuations on CT scan.
In this study, the vast majority of viruses detected belong to the Herpesviridae family, mainly HSV-1 (38.6 % of all virus-positive BALF) and CMV (33 %), as previously described by Garbino et al. in unselected hospitalized patients [14]. Respiratory viruses were found in only five cases out of the 232 BALF investigated: RSV (n = 3), rhinovirus (n = 1), and adenovirus (n = 1), representing only a small proportion (7.1 %) of virus-positive BALF. This low prevalence of respiratory viruses may be related to the sub-optimal selection of cases where these tests were performed in our institution during the study period. In addition, the absence of any influenza diagnostic in BALF may be explained by the low intensity of seasonal flu in France during the study year (2008) and by the preferential use of PCR tests on nasopharyngeal aspirates when the diagnosis of influenza is suspected. Multiple studies performed in the ICU have identified herpes viruses, mainly HSV and CMV, in patients with VAP. HSV-1 has been found in 16–62 % of patients under mechanical ventilation for >5 days (median, 7 days) [18–20]. Luyt et al. documented an HSV-related cyto-pathogenic effect, with intra-nuclear inclusions in 20 % of VAP in patients ventilated for a median duration of 14 days [20]. CMV replication was observed in the plasma of one-third of CMV-seropositive patients with VAP after 4–12 days of mechanical ventilation, and lung involvement was documented by CMV-related cyto-pathogenic effect in 5–30 % of patients [21–23]. All these studies found that HSV and CMV in BALF are associated with increased morbidity and/or mortality. However, the causal relationship between HSV or CMV in BALF and patient outcomes cannot be ascertained from these observational studies.
Among the rapid tests currently available, PCR is one of the most valuable, the results being available within hours with high sensitivity, especially in immunocompromised patients [2]. Multiplex PCR tests, with their ability to detect several viruses in one set, may be particularly interesting in the diagnostic workup of acute respiratory diseases suspected to be of viral origin [24–26]. In our study, the diagnostic yield of viral tests would have dropped from 30 to 20 %, had PCR tests not been performed. This advocates for the systematic use of PCR techniques for viral tests in BALF, in accordance with previous studies [27, 28], in the situations where viruses may reasonably be suspected (i.e., acute lower tract respiratory disease in immunocompromised patients and/or patients with unexplained bilateral ground-glass attenuations on CT scan). On the other hand, the initial assessment of immunocompetent patients with interstitial lung disease should not include any viral test on BALF, as previously reported in 40 patients with interstitial fibrosis [29]. During the exacerbation of idiopathic fibrosis, viral tests on BALF may be of higher value, although recent papers have questioned their clinical significance [29, 30].
This study has limitations related to its retrospective, monocentric, and observational design, as investigations on BALF were not protocolized, and the request for viral testing was left to the discretion of the physician in charge. Firstly, the retrospective review of medical charts identified a significant proportion of patients who were unlikely to suffer from viral infections, and for whom viral tests should not have been performed. In contrast, among patients not included in this study as no viral test was requested, there probably were patients who would have benefited from viral tests on BALF. However, the comparison of the 212 patients who had BALFs tested for viruses in 2008 and a random selection of 40 patients who had BALF samples not tested for viruses during the same year found that the only significant differences were immunodepression (10 % vs. 83 %, p < 0.0001) and ground-glass attenuations on CT scan (23 % vs. 74 %, p < 0.0001). This suggests that clinicians are aware of the situations most likely to be associated with the presence of viruses, and that they appropriately select the patients in whom viral tests are more likely to return positive. Lastly, PCR was performed in only 149 of 232 BALF investigated for viruses. More systematic use of PCR tests in these patients may have increased the proportion of viruses identified. Secondly, the identification of virus in BALF during the diagnostic workup of a respiratory disease does not systematically imply that the virus is responsible for the disease and that the patient will improve with appropriate antiviral treatment. Despite these limitations, this observational study allowed us to identify categories of respiratory diseases where viral tests are very unlikely to return positive (e.g., initial assessment of immunocompetent patients with interstitial pneumonia or pulmonary micronodules). As a consequence, indications for viral tests in BALF were dramatically reduced for these patients in our institution. In conclusion, testing for viruses on BALF should be mostly restricted to acute lower tract respiratory disease in immunocompromised patients and/or patients with unexplained ground-glass attenuations on CT scan, especially when PCR tests are used.
References
Mayaud C, Cadranel J (2000) A persistent challenge: the diagnosis of respiratory disease in the non-AIDS immunocompromised host. Thorax 55(6):511–517
Camps Serra M, Cervera C, Pumarola T, Moreno A, Perelló R, Torres A, Jiménez de Anta MT, Marcos MA (2008) Virological diagnosis in community-acquired pneumonia in immunocompromised patients. Eur Respir J 31(3):618–624
de Roux A, Marcos MA, Garcia E, Mensa J, Ewig S, Lode H, Torres A (2004) Viral community-acquired pneumonia in nonimmunocompromised adults. Chest 125(4):1343–1351
Garbino J, Gerbase MW, Wunderli W, Deffernez C, Thomas Y, Rochat T, Ninet B, Schrenzel J, Yerly S, Perrin L, Soccal PM, Nicod L, Kaiser L (2004) Lower respiratory viral illnesses: improved diagnosis by molecular methods and clinical impact. Am J Respir Crit Care Med 170(11):1197–1203
Garbino J, Soccal PM, Aubert JD, Rochat T, Meylan P, Thomas Y, Tapparel C, Bridevaux PO, Kaiser L (2009) Respiratory viruses in bronchoalveolar lavage: a hospital-based cohort study in adults. Thorax 64(5):399–404
Bafadhel M, McKenna S, Terry S, Mistry V, Reid C, Haldar P, McCormick M, Haldar K, Kebadze T, Duvoix A, Lindblad K, Patel H, Rugman P, Dodson P, Jenkins M, Saunders M, Newbold P, Green RH, Venge P, Lomas DA, Barer MR, Johnston SL, Pavord ID, Brightling CE (2011) Acute exacerbations of COPD: identification of biological clusters and their biomarkers. Am J Respir Crit Care Med 184(6):662–671
Busse WW, Lemanske RF Jr, Gern JE (2010) Role of viral respiratory infections in asthma and asthma exacerbations. Lancet 376(9743):826–834
De Serres G, Lampron N, La Forge J, Rouleau I, Bourbeau J, Weiss K, Barret B, Boivin G (2009) Importance of viral and bacterial infections in chronic obstructive pulmonary disease exacerbations. J Clin Virol 46(2):129–133
Rosenthal LA, Avila PC, Heymann PW, Martin RJ, Miller EK, Papadopoulos NG, Peebles RS Jr, Gern JE; Infections and Asthma Committee, Environmental and Occupational Respiratory Diseases Interest Section, American Academy of Allergy, Asthma & Immunology (2010) Viral respiratory tract infections and asthma: the course ahead. J Allergy Clin Immunol 125(6):1212–1217
Stuck AE, Minder CE, Frey FJ (1989) Risk of infectious complications in patients taking glucocorticosteroids. Rev Infect Dis 11(6):954–963
[No authors listed] (1989) Technical recommendations and guidelines for bronchoalveolar lavage (BAL). Report of the European Society of Pneumology Task Group. Eur Respir J 2(6):561–585
Febvre M, Trosini-Desert V, Atassi K, Hermant C, Colchen A, Raspaud C, Vergnon JM; Endoscopy Working Group of the French Society of Pulmonary Medicine (2007) Diagnostic flexible bronchoscopy. Recommendations of the Endoscopy Working Group of the French Society of Pulmonary Medicine. Rev Mal Respir 24(10):1363–1392
Minjolle S, Michelet C, Jusselin I, Joannes M, Cartier F, Colimon R (1999) Amplification of the six major human herpesviruses from cerebrospinal fluid by a single PCR. J Clin Microbiol 37(4):950–953
Garbino J, Gerbase MW, Wunderli W, Kolarova L, Nicod LP, Rochat T, Kaiser L (2004) Respiratory viruses and severe lower respiratory tract complications in hospitalized patients. Chest 125(3):1033–1039
Ruuskanen O, Lahti E, Jennings LC, Murdoch DR (2011) Viral pneumonia. Lancet 377(9773):1264–1275
Franquet T, Rodriguez S, Martino R, Giménez A, Salinas T, Hidalgo A (2006) Thin-section CT findings in hematopoietic stem cell transplantation recipients with respiratory virus pneumonia. AJR Am J Roentgenol 187(4):1085–1090
Wong CK, Lai V, Wong YC (2012) Comparison of initial high resolution computed tomography features in viral pneumonia between metapneumovirus infection and severe acute respiratory syndrome. Eur J Radiol 81(5):1083–1087
Bruynseels P, Jorens PG, Demey HE, Goossens H, Pattyn SR, Elseviers MM, Weyler J, Bossaert LL, Mentens Y, Ieven M (2003) Herpes simplex virus in the respiratory tract of critical care patients: a prospective study. Lancet 362(9395):1536–1541
De Vos N, Van Hoovels L, Vankeerberghen A, Van Vaerenbergh K, Boel A, Demeyer I, Creemers L, De Beenhouwer H (2009) Monitoring of herpes simplex virus in the lower respiratory tract of critically ill patients using real-time PCR: a prospective study. Clin Microbiol Infect 15(4):358–363
Luyt CE, Combes A, Deback C, Aubriot-Lorton MH, Nieszkowska A, Trouillet JL, Capron F, Agut H, Gibert C, Chastre J (2007) Herpes simplex virus lung infection in patients undergoing prolonged mechanical ventilation. Am J Respir Crit Care Med 175(9):935–942
Limaye AP, Kirby KA, Rubenfeld GD, Leisenring WM, Bulger EM, Neff MJ, Gibran NS, Huang ML, Santo Hayes TK, Corey L, Boeckh M (2008) Cytomegalovirus reactivation in critically ill immunocompetent patients. JAMA 300(4):413–422
Osawa R, Singh N (2009) Cytomegalovirus infection in critically ill patients: a systematic review. Crit Care 13(3):R68
Chiche L, Forel JM, Roch A, Guervilly C, Pauly V, Allardet-Servent J, Gainnier M, Zandotti C, Papazian L (2009) Active cytomegalovirus infection is common in mechanically ventilated medical intensive care unit patients. Crit Care Med 37(6):1850–1857
Coiras MT, Aguilar JC, García ML, Casas I, Pérez-Breña P (2004) Simultaneous detection of fourteen respiratory viruses in clinical specimens by two multiplex reverse transcription nested-PCR assays. J Med Virol 72(3):484–495
Gröndahl B, Puppe W, Hoppe A, Kühne I, Weigl JA, Schmitt HJ (1999) Rapid identification of nine microorganisms causing acute respiratory tract infections by single-tube multiplex reverse transcription-PCR: feasibility study. J Clin Microbiol 37(1):1–7
Puppe W, Weigl JA, Aron G, Gröndahl B, Schmitt HJ, Niesters HG, Groen J (2004) Evaluation of a multiplex reverse transcriptase PCR ELISA for the detection of nine respiratory tract pathogens. J Clin Virol 30(2):165–174
Jain P, Sandur S, Meli Y, Arroliga AC, Stoller JK, Mehta AC (2004) Role of flexible bronchoscopy in immunocompromised patients with lung infiltrates. Chest 125(2):712–722
Costa C, Delsedime L, Solidoro P, Curtoni A, Bergallo M, Libertucci D, Baldi S, Rinaldi M, Cavallo R (2010) Herpesviruses detection by quantitative real-time polymerase chain reaction in bronchoalveolar lavage and transbronchial biopsy in lung transplant: viral infections and histopathological correlation. Transplant Proc 42(4):1270–1274
Wootton SC, Kim DS, Kondoh Y, Chen E, Lee JS, Song JW, Huh JW, Taniguchi H, Chiu C, Boushey H, Lancaster LH, Wolters PJ, DeRisi J, Ganem D, Collard HR (2011) Viral infection in acute exacerbation of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 183(12):1698–1702
Collard HR, Moore BB, Flaherty KR, Brown KK, Kaner RJ, King TE Jr, Lasky JA, Loyd JE, Noth I, Olman MA, Raghu G, Roman J, Ryu JH, Zisman DA, Hunninghake GW, Colby TV, Egan JJ, Hansell DM, Johkoh T, Kaminski N, Kim DS, Kondoh Y, Lynch DA, Müller-Quernheim J, Myers JL, Nicholson AG, Selman M, Toews GB, Wells AU, Martinez FJ; Idiopathic Pulmonary Fibrosis Clinical Research Network Investigators (2007) Acute exacerbations of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 176(7):636–643
Acknowledgments
The authors thank Jean-Marc Malecot for his statistical advice.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jouneau, S., Poineuf, JS., Minjolle, S. et al. Which patients should be tested for viruses on bronchoalveolar lavage fluid?. Eur J Clin Microbiol Infect Dis 32, 671–677 (2013). https://doi.org/10.1007/s10096-012-1791-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-012-1791-7