Abstract
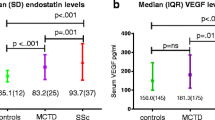
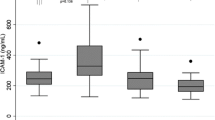
CD146, a transmembrane glycoprotein member of the immunoglobulin superfamily, acts as an adhesion molecule that helps maintain the cell monolayer. Human endothelial cells expressing CD146 are involved in angiogenesis and inflammation. Recently, we developed a sandwich ELISA for detecting soluble CD146 (sCD146) in human serum specimens. The aim of this study is to determine serum levels of sCD146 in patients with systemic sclerosis (SSc) and to examine the relationship between sCD146 levels and clinical manifestations. We quantified serum sCD146 levels in 47 serum samples from patients fulfilling criteria for SSc, 23 serum samples from patients fulfilling criteria for rheumatoid arthritis (RA), and 25 healthy controls. We also investigated the relationship between sCD146 levels and various clinical characteristics with SSc patients. Levels of sCD146 were significantly higher in the 47 patients with SSc than in the 25 healthy controls and 23 patients with RA (12.50 vs. 6.91 vs. 9.95 ng/ml; p < 0.001). Serum sCD146 levels in SSc patients with pulmonary arterial hypertension (PAH) were lower than in SSc patients without PAH (10.12 vs.13.17 ng/ml; p < 0.01). The serum levels of sCD146 were elevated in patients with SSc. However, decreased sCD146 levels were observed in SSc patients with PAH. Further studies are necessary to elucidate the sources and the mechanisms.



Similar content being viewed by others
References
Almeida I, Faria R, Vita P, Vasconcelos C (2011) Systemic sclerosis refractory disease: from the skin to the heart. Autoimmun Rev 10(11):693–701
Dagur PK, Mccoy JP Jr (2015) Endothelial-binding, proinflammatory T cells identified by MCAM (CD146) expression: characterization and role in human autoimmune diseases. Autoimmun Rev 14(5):415–422
Bardin N, Anfosso F, Massé JM, Cramer E, Sabatier F, Le Bivic A, Sampol J, Dignat-George F (2001) Identification of CD146 as a component of the endothelial junction involved in the control of cell-cell cohesion. Blood 98:3677–3684
Jiang T, Zhuang J, Duan H, Luo Y, Zeng Q, Fan K, Yan H, Lu D, Ye Z, Hao J, Feng J, Yang D, Yan X (2012) CD146 is a coreceptor for VEGFR-2 in tumor angiogenesis. Blood 120(11):2330–2339
Hadjinicolaou AV, Wu L, Fang B, Watson PA, Hall FC, Busch R (2013) Relationship of CD146 expression to activation of circulating T cells: exploratory studies in healthy donors and patients with connective tissue diseases. Clin Exp Immunol 174:73–88
Kamiyama T, Watanabe H, Iijima M, Miyazaki A, Iwamoto S (2012) Coexpression of CCR6 and CD146 (MCAM) is a marker of effector memory T-helper 17 cells. J Dermtol 39(10):838–842
Larochelle C, Cayrol R, Kebir H, Alvarez JI, Lécuyer MA, Ifergan I, Viel É, Bourbonnière L, Beauseigle D, Terouz S et al (2012) Melanoma cell adhesion molecule identifies encephalitogenic T lymphocytes and promotes their recruitment to the central nervous system. Brain 135(Pt10):2906–2924
Neidhart M, Wehrli R, Brühlmann P, Michel BA, Gay RE, Gay S (1999) Synovial fluid CD146 (MUC18), a marker for synovial membrane angiogenesis in rheumatoid arthritis. Arthritis Rheum 42(4):622–630
Bardin N, Reumaux D, Geboes K, Colombel JF, Blot-Chabaud M, Sampol J, Duthilleul P, Dignat-George F (2006) Increased expression of CD146, a new marker of the endothelial junction in active inflammatory bowel disease. Inflamm Bowel Dis 12(1):16–21
Bardin N, Blot-Chabaud M, Despoix N, Kebir A, Harhouri K, Arsanto JP, Espinosa L, Perrin P, Robert S, Vely F, Sabatier F, Le Bivic A, Kaplanski G, Sampol J, Dignat-George F (2009) CD146 and its soluble form regulate monocyte transendothelial migration. Arterioscler ThrombVasc Biol 29(15):746–753
Harhouri K, Kebir A, Guillet B, Foucault-Bertaud A, Voytenko S, Piercecchi-Marti MD, Berenguer C, Lamy E, Vely F, Pisano P et al (2010) Soluble CD146 displays angiogenic properties and promotes neovascularization in experimental hind-limb ischemia. Blood 115(18):3843–3851
Gayat E, Caillard A, Laribi S, Mueller C, Sadoune M, Seronde MF, Maisel A, Bartunek J, Vanderheyden M, Desutter J et al (2015) Soluble CD146, a new endothelial biomarker of acutely decompensated heart failure. Int J Cardiol 199:241–247
van den Hoogen F, Khanna D, Fransen J, Johnson SR, Baron M, Tyndall A, Matucci-Cerinic M, Naden RP, Medsger TA Jr, Carreira PE et al (2013) 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum 65(11):2737–2747
Hoeper MM, Bogaard HJ, Condliffe R, Frantz R, Khanna D, Kurzyna M, Langleben D, Manes A, Satoh T, Torres F et al (2014) Definitions and diagnosis of pulmonary hypertension. J Am Coll Cardiol 62(25 Suppl):D42–D50
van der Helm-van Mil AH, Huizinga TW (2012) The 2010 ACR/EULAR criteria for rheumatoid arthritis: do they affect the classification or diagnosis of rheumatoid arthritis? Ann Rheum Dis 71(10):1596–1598
Jimenez SA (2013) Role of endothelial to mesenchymal transition in the pathogenesis of the vascular alterations in systemic sclerosis. ISRN Rheumatol. doi:10.1155/2013/835948
Andersen GN, Caidahl K, Kazzam E, Petersson AS, Waldenström A, Mincheva-Nilsson L, Rantapää-Dahlqvist S (2000) Correlation between increased nitric oxide production and markers of endothelial activation in systemic sclerosis: findings with the soluble adhesion molecules E-selectin, intercellular adhesion molecule 1, and vascular cell adhesion molecule 1. Arthritis Rheum 43(5):1085–1093
Denton CP, Bickerstaff MC, Shiwen X, Carulli MT, Haskard DO, Dubois RM, Black CM (1995) Serial circulating adhesion molecule levels reflect disease severity in systemic sclerosis. Br J Rheumatol 34(11):1048–1054
Kuryliszyn-Moskal A, Klimiuk PA, Sierakowski S (2005) Soluble adhesion molecules (sVCAM-1, s E-selectin), vascular endothelial growth factor (VEGF) and endothelin-1 in patients with systemic sclerosis: relationship to organ systemic involvement. Clin Rheumatol 24(2):111–116
Celebi Sözener Z, Karabıyıkoğlu G, Düzgün N (2010) Evaluation of the functional parameters in scleroderma cases with pulmonary involvement. Tuberk Toraks 58(3):235–241
Humbert M, Morrell NW, Archer SL, Stenmark KR, MacLean MR, Lang IM, Christman BW, Weir EK, Eickelberg O, Voelkel NF, Rabinovitch M (2004) Cellular and molecular pathobiology of pulmonary arterial hypertension. J Am Coll Cardiol 43(12 Suppl S):13S–24S
Hassoun PM, Mouthon L, Barberà JA, Eddahibi S, Flores SC, Grimminger F, Jones PL, Maitland ML, Michelakis ED, Morrell NW et al (2009) Inflammation, growth factors, and pulmonary vascular remodeling. J Am Coll Cardiol 54(Suppl 1):S10–S19
Voelkel NF, Gomez-Arroyo J (2014) The role of vascular endothelial growth factor in pulmonary arterial hypertension. The angiogenesis paradox. Am J Respir Cell Mol Biol 51(4):474–484
Derrett-Smith EC, Dooley A, Gilbane AJ, Trinder SL, Khan K, Baliga R, Holmes AM, Hobbs AJ, Abraham D, Denton CP (2013) Endothelial injury in a transforming growth factor β-dependent mouse model of scleroderma induces pulmonary arterial hypertension. Arthritis Rheum 65(11):2928–2939
Taraseviciene-Stewart L, Kasahara Y, Alger L, Hirth P, Mc Mahon G, Waltenberger J, Voelkel NF, Tuder RM (2001) Inhibition of the VEGF receptor 2 combined with chronic hypoxia causes cell death-dependent pulmonary endothelial cell proliferation and severe pulmonary hypertension. FASEB J 15(2):427–438
Neuber C, Pufe J, Pietzsch J (2015) Influence of irradiation on release of endothelial microparticles (EMP) in vitro. Clin Hemorheol Microcirc 61(2):291–299
Lanuti P, Santilli F, Marchisio M, Pierdomenico L, Vitacolonna E, Santavenere E, Iacone A, Davì G, Romano M, Miscia S (2012) A novel flow cytometric approach to distinguish circulating endothelial cells from endothelial microparticles: relevance for the evaluation of endothelial dysfunction. J Immunol Methods 380(1–2):16–22
Acknowledgments
This research was supported in part by a Grant-in-Aid for Scientific Research “B” (grant no. 25293133) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Rights and permissions
About this article
Cite this article
Ito, T., Tamura, N., Okuda, S. et al. Elevated serum levels of soluble CD146 in patients with systemic sclerosis. Clin Rheumatol 36, 119–124 (2017). https://doi.org/10.1007/s10067-016-3434-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-016-3434-3