Abstract
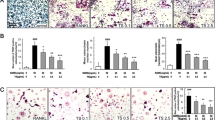
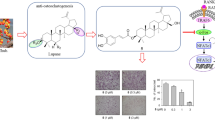
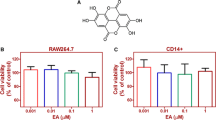
Silibinin is a polyphenolic flavonoid compound isolated from milk thistle (Silybum marianum), with known hepatoprotective, anticarcinogenic, and antioxidant effects. Herein, we show that silibinin inhibits receptor activator of NF-κB ligand (RANKL)-induced osteoclastogenesis from RAW264.7 cells as well as from bone marrow-derived monocyte/macrophage cells in a dose-dependent manner. Silibinin has no effect on the expression of RANKL or the soluble RANKL decoy receptor osteoprotegerin (OPG) in osteoblasts. However, we demonstrate that silibinin can block the activation of NF-κB, c-Jun N-terminal kinase (JNK), p38 mitogen-activated protein (MAP) kinase, and extracellular signal-regulated kinase (ERK) in osteoclast precursors in response to RANKL. Furthermore, silibinin attenuates the induction of nuclear factor of activated T cells (NFAT) c1 and osteoclast-associated receptor (OSCAR) expression during RANKL-induced osteoclastogenesis. We demonstrate that silibinin can inhibit TNF-α-induced osteoclastogenesis as well as the expression of NFATc1 and OSCAR. Taken together, our results indicate that silibinin has the potential to inhibit osteoclast formation by attenuating the downstream signaling cascades associated with RANKL and TNF-α.
Similar content being viewed by others
References
Boyle, W.J., Simonet, W.S., and Lacey, D.L. (2003). Osteoclast differentiation and activation. Nature 423, 337–342.
Hahn, G., Lehmann, H.D., Kurten, M., Uebel, H., and Vogel, G. (1968). [On the pharmacology and toxicology of silymarin, an antihepatotoxic active principle from Silybum marianum (L.) Gaertn]. Arzneimittel-Forschung 18, 698–704.
Hsu, H., Lacey, D.L., Dunstan, C.R., Solovyev, I., Colombero, A., Timms, E., Tan, H.L., Elliott, G., Kelley, M.J., Sarosi, I. et al. (1999). Tumor necrosis factor receptor family member RANK mediates osteoclast differentiation and activation induced by osteoprotegerin ligand. Proc. Natl. Acad. Sci. USA 96, 3540–3545.
Kim, N., Takami, M., Rho, J., Josien, R., and Choi, Y. (2002). A novel member of the leukocyte receptor complex regulates osteoclast differentiation. J. Exp. Med. 195, 201–209.
Kim, K., Kim, J.H., Lee, J., Jin, H.M., Lee, S.H., Fisher, D.E., Kook, H., Kim, K.K., Choi, Y., and Kim, N. (2005a). Nuclear factor of activated T cells c1 induces osteoclast-associated receptor gene expression during tumor necrosis factor-related activation-induced cytokine-mediated osteoclastogenesis. J. Biol. Chem. 280, 35209–35216.
Kim, N., Kadono, Y., Takami, M., Lee, J., Lee, S.H., Okada, F., Kim, J.H., Kobayashi, T., Odgren, P.R., Nakano, H., et al. (2005b). Osteoclast differentiation independent of the TRANCE-RANK-TRAF6 axis. J. Exp. Med. 202, 589–595.
Kim, K., Kim, J.H., Lee, J., Jin, H.M., Kook, H., Kim, K.K., Lee, S.Y., and Kim, N. (2007). MafB negatively regulates RANKL-mediated osteoclast differentiation. Blood 109, 3253–3259.
Kim, K., Lee, S.H., Ha Kim, J., Choi, Y., and Kim, N. (2008). NFATc1 induces osteoclast fusion via up-regulation of Atp6v0d2 and the dendritic cell-specific transmembrane protein (DC-STAMP). Mol. Endocrinol. (Baltimore, Md.) 22, 176–185.
Kobayashi, K., Takahashi, N., Jimi, E., Udagawa, N., Takami, M., Kotake, S., Nakagawa, N., Kinosaki, M., Yamaguchi, K., Shima, N., et al. (2000). Tumor necrosis factor alpha stimulates osteoclast differentiation by a mechanism independent of the ODF/RANKL-RANK interaction. J. Exp. Med. 191, 275–286.
Koga, T., Inui, M., Inoue, K., Kim, S., Suematsu, A., Kobayashi, E., Iwata, T., Ohnishi, H., Matozaki, T., Kodama, T., et al. (2004). Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428, 758–763.
Kwak, H.B., Sun, H.M., Ha, H., Lee, J.H., Kim, H.N., and Lee, Z.H. (2008). AG490, a Jak2-specific inhibitor, induces osteoclast survival by activating the Akt and ERK signaling pathways. Mol. Cells 26, 436–442.
Lacey, D.L., Timms, E., Tan, H.L., Kelley, M.J., Dunstan, C.R., Burgess, T., Elliott, R., Colombero, A., Elliott, G., Scully, S., et al. (1998). Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93, 165–176.
Lee, Z.H., and Kim, H.H. (2003). Signal transduction by receptor activator of nuclear factor kappa B in osteoclasts. Biochem. Biophys. Res. Comm. 305, 211–214.
Lee, S.E., Chung, W.J., Kwak, H.B., Chung, C.H., Kwack, K.B., Lee, Z.H., and Kim, H.H. (2001). Tumor necrosis factor-alpha supports the survival of osteoclasts through the activation of Akt and ERK. J. Biol. Chem. 276, 49343–49349.
Lee, J., Kim, K., Kim, J.H., Jin, H.M., Choi, H.K., Lee, S.H., Kook, H., Kim, K.K., Yokota, Y., Lee, S.Y., et al. (2006). Id helix-loop-helix proteins negatively regulate TRANCE-mediated osteoclast differentiation. Blood 107, 2686–2693.
Li, L.H., Wu, L.J., Tashiro, S.I., Onodera, S., Uchiumi, F., and Ikejima, T. (2006). The roles of Akt and MAPK family members in silymarin’s protection against UV-induced A375-S2 cell apoptosis. Int. Immunopharmacol. 6, 190–197.
Manna, S.K., Mukhopadhyay, A., Van, N.T., and Aggarwal, B.B. (1999). Silymarin suppresses TNF-induced activation of NF-kappa B, c-Jun N-terminal kinase, and apoptosis. J. Immunol. 163, 6800–6809.
Ramasamy, K., and Agarwal, R. (2008). Multitargeted therapy of cancer by silymarin. Cancer Lett. 269, 352–362.
Rho, J., Takami, M., and Choi, Y. (2004). Osteoimmunology: interactions of the immune and skeletal systems. Mol. Cells 17, 1–9.
Singh, R.P., and Agarwal, R. (2002). Flavonoid antioxidant silymarin and skin cancer. Antioxid. Redox Signal. 4, 655–663.
Singh, R.P., and Agarwal, R. (2005). Mechanisms and preclinical efficacy of silibinin in preventing skin cancer. Eur. J. Cancer 41, 1969–1979.
Singh, R.P., Dhanalakshmi, S., Tyagi, A.K., Chan, D.C., Agarwal, C., and Agarwal, R. (2002). Dietary feeding of silibinin inhibits advance human prostate carcinoma growth in athymic nude mice and increases plasma insulin-like growth factor-binding protein-3 levels. Cancer Res. 62, 3063–3069.
Singh, R.P., Dhanalakshmi, S., Agarwal, C., and Agarwal, R. (2005). Silibinin strongly inhibits growth and survival of human endothelial cells via cell cycle arrest and downregulation of survivin, Akt and NF-kappaB: implications for angioprevention and antiangiogenic therapy. Oncogene 24, 1188–1202.
Suda, T., Jimi, E., Nakamura, I., and Takahashi, N. (1997). Role of 1 alpha,25-dihydroxyvitamin D3 in osteoclast differentiation and function. Methods Enzymol. 282, 223–235.
Suda, T., Takahashi, N., Udagawa, N., Jimi, E., Gillespie, M.T., and Martin, T.J. (1999). Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 20, 345–357.
Takayanagi, H., Kim, S., Koga, T., Nishina, H., Isshiki, M., Yoshida, H., Saiura, A., Isobe, M., Yokochi, T., Inoue, J., et al. (2002). Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell 3, 889–901.
Walsh, M.C., Kim, N., Kadono, Y., Rho, J., Lee, S.Y., Lorenzo, J., and Choi, Y. (2006). Osteoimmunology: interplay between the immune system and bone metabolism. Ann. Rev. Immunol. 24, 33–63.
Yasuda, H., Shima, N., Nakagawa, N., Yamaguchi, K., Kinosaki, M., Mochizuki, S., Tomoyasu, A., Yano, K., Goto, M., Murakami, A., et al. (1998). Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl. Acad. Sci. USA 95, 3597–3602.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kim, J.H., Kim, K., Jin, H.M. et al. Silibinin inhibits osteoclast differentiation mediated by TNF family members. Mol Cells 28, 201–207 (2009). https://doi.org/10.1007/s10059-009-0123-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-009-0123-y