Abstract
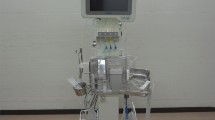
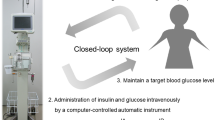
The aim of the present study was to evaluate the usefulness of a closed-loop system (STG-55; Nikkiso, Tokyo, Japan), a type of artificial endocrine pancreas for the continuous monitoring and control of intraoperative blood glucose, for preventing postoperative acute kidney injury (AKI) in patients undergoing hepatectomy. Thirty-eight patients were enrolled in this study. Glucose concentrations were controlled with either a manual injection of insulin based on a commonly used sliding scale (manual insulin group, n = 19) or the programmed infusion of insulin determined by the control algorithm of the artificial endocrine pancreas (programmed insulin group, n = 19). After the induction of anesthesia, a 20-G intravenous catheter was inserted into the peripheral forearm vein of patients in the programmed insulin group and connected to an artificial endocrine pancreas (STG-55). The target range for glucose concentrations was set to 100–150 mg/dL. The mean serum creatinine concentrations of preoperative, postoperative 24 and 48 h were 0.72, 0.78, and 0.79 mg/dL in the programmed insulin group, and 0.81, 0.95, and 1.03 mg/dL in the manual insulin group, respectively. Elevations in serum creatinine concentrations postoperative 48 h were significantly suppressed in the programmed insulin group. The STG-55 closed-loop system was effective for maintaining strict blood glucose control during hepatectomy with minimal variability in blood glucose concentrations and for suppressing elevations in serum creatinine concentrations. Strict blood glucose control by an artificial endocrine pancreas during hepatectomy may prevent postoperative AKI.




Similar content being viewed by others
References
Okabayashi T, Nishimori I, Maeda H, Yamashita K, Yatabe T, Hanazaki K. Effect of intensive insulin therapy using a closed-loop glycemic control system in hepatic resection patients: a prospective randomized clinical trial. Diabetes Care. 2009;32:1425–7.
Maeda H, Okabayashi T, Nishimori I, Yamashita K, Sugimoto T, Hanazaki K. Hyperglecemia during hepatic resection: continuous monitoring of blood glucose concentration. Am J Surg. 2010;199:8–13.
Yatabe T, Kitagawa H, Kawano T, Munekage M, Okabayashi T, Yamashita K, Hanazaki K, Yokoyama M. Continuous monitoring of glucose levels in the hepatic vein and systemic circulation during the Pringle maneuver in beagles. J Artif Organs. 2011;14:232–7.
Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012;120:c179–84.
Coca SG, Yusuf B, Shlipak MG, Garg AX, Parikh CR. Long-term risk of mortality and other adverse outcome after acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis. 2009;53:961–73.
Tsai MS, Lin CL, Chang SN, Lee PH, Kao CH. Diabetes mellitus and increased postoperative risk of acute renal failure after hepatectomy for hepatocellular carcinoma: a nationwide population-based study. Ann Surg Oncol. 2014;21:3810–6.
Song JW, Shim JK, Yoo KJ, Oh SY, Kwak YL. Impact of intraoperative hyperglycaemia on renal dysfunction after off-pump coronary artery bypass. Interact CardioVasc Thorac Surg. 2013;17:473–8.
Van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R. Intensive insulin therapy in critically ill patients. N Eng J Med. 2001;345:1359–67.
Van den Berghe G, Wouters PJ, Bouillon R, Weekers F, Verwaest C, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P. Outcome benefit of intensive insulin therapy in the critically ill: insulin dose versus glycemic control. Crit Care Med. 2003;31:359–66.
Egi M, Bellomo R, Stachowski E, French CJ, Hart G. Variability of blood glucose concentration and short-term mortality in critically ill patients. Anesthesiology. 2006;105:244–52.
Hanazaki K, Nosé Y, Brunicardi FC. Artificial endocrine pancreas. J Am Coll Surg. 2001;193:310–22.
Yamashita K, Okabayashi T, Yokoyama T, Yatabe T, Maeda H, Manabe M, Hanazaki K. The accuracy of a continuous blood glucose monitor during surgery. Anesth Analg. 2008;106:160–3.
Clarke WL, Cox D, Gonder-Frederick LA, Carter W, Pohl SL. Evaluating clinical accuracy of systems for self-monitoring of blood glucose. Diabetes Care. 1987;10:622–8.
Tomozawa A, Ishikawa S, Shiota N, Cholvisudhi P, Makita K. Perioperative risk factors for acute kidney injury after liver resection surgery: an historical cohort study. Can J Anaesth. 2015;62:753–61.
Okabayashi T, Nishimori I, Yamashita K, Sugimoto T, Yatabe T, Maeda H, Kobayashi M, Hanazaki K. Risk factors and predictors for surgical site infection after hepatic resection. J Hosp Infect. 2009;73:47–53.
Hirose R, Xu F, Dang K, Liu T, Behrends M, Brakeman PR, Kronish JW, Niemann CU. Transient hyperglycemia affects the extent of ischemia-reperfusion-induced renal injury in rats. Anesthesiology. 2008;108:402–14.
Vanhorebeek I, Gunst J, Ellger B, Boussemaere M, Lerut E, Debaveye Y, Rabbani N, Thornalley PJ, Schetz M, Van den Berghe G. Hyperglycemic kidney damage in an animal model of prolonged critical illness. Kidney Int. 2009;76:512–20.
Hall AM, Unwin RJ. The not so ‘mighty chondrion’: emergence of renal diseases due to mitochondrial dysfunction. Nephron Physiol. 2007;105:p1–10.
Esposito K, Nappo F, Marfella R, Giugliano G, Giugliano F, Ciotola M, Quagliaro C, Ceriello A, Giugliano D. Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans: role of oxidative stress. Circulation. 2002;106:2067–72.
Ellger B, Debaveye Y, Vanhorebeek I, Langouche L, Giulietti A, Van Etten E, Herijgers P, Mathieu C, Van den Berghe G. Survival benefits of intensive insulin therapy in critical illness: impact of maintaining normoglycemia versus glycemia-independent actions of insulin. Diabetes. 2006;55:1096–105.
Basile DP. The endothelial cell in ischemic acute kidney injury: implications for acute and chronic function. Kidney Int. 2007;72:151–6.
Tuuminen R, Jouppila A, Salvail D, Laurent C-E, Benoit M-C, Syrjälä S, Helin H, Lemström K, Lassila R. Dual antiplatelet and anticoagulant APAC prevents experimental ischemia-reperfusion-induced acute kidney injury. Clin Exp Nephrol. 2016. doi:10.1007/s10157-016-1308-2.
Finney SJ, Zekveld C, Elia A, Evans TW. Glucose control and mortality in critically ill patients. JAMA. 2003;290:2041–7.
Krinsley JS. Effect of an intensive glucose management protocol on the mortality of critically ill adult patients. Mayo Clin Proc. 2004;79:992–1000.
Lipshutz AKM, Gropper MA. Perioperative glycemic control: an evidence-based review. Anesthesiology. 2009;110:408–21.
The NICE-SUGAR study Investigators. Intensive versus conventional glucose control in critically ill patients. N Eng J Med. 2009;360:1283–97.
Wiener RS, Wiener DC, Larson RJ. Benefits and risks of tight glucose control in critically ill adults: a meta-analysis. JAMA. 2008;300:933–44.
Arabi YM, Dabbagh OC, Tamim HM, Al-Shimemeri AA, Memish ZA, Haddad SH, Syed SJ, Giridhar HR, Rishu AH, Al-Daker MO, Kahoul SH, Britts RJ, Sakkijha MH. Intensive versus conventional insulin therapy: a randomized controlled trial in medical and surgical critically ill patients. Crit Care Med. 2008;36:3190–7.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Financial support
This study was supported by intramural departmental funds.
Rights and permissions
About this article
Cite this article
Mita, N., Kawahito, S., Soga, T. et al. Strict blood glucose control by an artificial endocrine pancreas during hepatectomy may prevent postoperative acute kidney injury. J Artif Organs 20, 76–83 (2017). https://doi.org/10.1007/s10047-016-0925-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-016-0925-6