Abstract
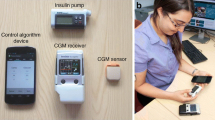
The incidence of diabetes is increasing at an unprecedented pace and has become a serious health concern worldwide during the last two decades. Despite this, adequate glycemic control using an artificial pancreas has not been established, although the 21st century has seen rapid developments in this area. Herein, we review current topics in glycemic control for both the wearable artificial pancreas for type 1 and type 2 diabetic patients and the bedside artificial pancreas for surgical diabetic patients. In type 1 diabetic patients, nocturnal hypoglycemia associated with insulin therapy remains a serious problem that could be addressed by the recent development of a wearable artificial pancreas. This smart phone-like device, comprising a real-time, continuous glucose monitoring system and insulin pump system, could potentially significantly reduce nocturnal hypoglycemia compared with conventional glycemic control. Of particular interest in this space are the recent inventions of a low-glucose suspend feature in the portable systems that automatically stops insulin delivery 2 h following a glucose sensor value <70 mg/dL and a bio-hormonal pump system consisting of insulin and glucagon pumps. Perioperative tight glycemic control using a bedside artificial pancreas with the closed-loop system has also proved safe and effective for not only avoiding hypoglycemia, but also for reducing blood glucose level variability resulting in good surgical outcomes. We hope that a more sophisticated artificial pancreas with closed-loop system will now be taken up for routine use worldwide, providing enormous relief for patients suffering from uncontrolled hyperglycemia, hypoglycemia, and/or variability in blood glucose concentrations.








Similar content being viewed by others
References
Vashist SK. Continuous glucose monitoring systems: a review. Diagnostics. 2013;3:385–412.
Hanazaki K, Nosé Y, Brunicardi FC. Artificial endocrine pancreas: a review. J Am Coll Surg. 2001;193:310–22.
Phillip M, Battelio T, Atlas E, et al. Nocturnal glucose control with an artificial pancreas at a diabetes camp. N Engl J Med. 2013;368:824–33.
Bergenstal RM, Klonoff DC, Garg SK, et al. Threshold-based insulin-pump interruption for reduction of hypoglycemia. N Engl J Med. 2013;369:224–32.
Hovorka R, Elleri D, Thabit H, et al. Overnight closed-loop insulin delivery in young people with type 1 diabetes: a free-living, randomized clinical trial. Diabetes Care. 2014;37:1204–11.
Russell SJ, El-Khatib FH, Sinha M, et al. Outpatient glycemic control with a bionic pancreas in type 1 diabetes. N Engl J Med. 2014;371:313–25.
Thabit H, Tauschmann M, Allen JM, et al. Home use of an artificial beta cell in type 1 diabetes. N Engl J Med. 2015;373:2129–40.
van den Berghe G, Wouters P, Weekers F, et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345:1359–67.
Wiener RS, Wiener DC, Larson RJ. Benefits and risks of tight glucose control in critically ill adults. JAMA. 2008;300:933–44.
NICE-SUGAR Study Investigators, Finfer S, Chittock DR, Su SY, et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360:1283–97.
Hanazaki K, Maeda H, Okabayashi T. Tight perioperative glycemic control using an artificial endocrine pancreas. Surg Today. 2010;40:1–7.
Yatabe T, Yamazaki R, Kitagawa H, et al. The evaluation of the ability of closed-loop glycemic control device to maintain the blood glucose concentration in intensive unit patients. Crit Care Med. 2011;39:575–8.
Hanazaki K, Kitagawa H, Yatabe T, et al. Perioperative intensive insulin therapy using an artificial endocrine pancreas with closed-loop glycemic control system: the effect of no hypoglycemia. Am J Surg. 2014;207:935–41.
Guillod L, Comte-Perret S, Monbaron D, Gaillard RC, Ruiz J. Nocturnal hypoglycemias in type 1 diabetic patients: what we can leran with continuous glucose monitoring? Diabetes Metab. 2007;33:360–5.
Maia FFR, Araujo LR. Effect of continuous glucose monitoring system (CGMS) to detect postprandial hyperglycemia and unrecognized hypoglycemia in type 1 diabetic patients. Diabetes Res Clin Pract. 2007;75:30–4.
Kaufman FR, Gibson LC, Halvorson M, Carpenter S, Fisher LK, Pitukcheewanont P. A pilot study of the continuous glucose monitoring system. Diabetes Care. 2001;24:2030–4.
Fatourechi MM, Kudva YC, Murad MH, et al. Hypoglycemia with intensive insulin therapy: a systematic review and meta analyses of randomized trials of CSII versus MDI. J Clin Endocrinol Metab. 2009;94:729–40.
Jeitler K, Horvath K, Berghold A, et al. Continuous subcutaneous insulin infusion versus multiple daily insulin injections in patients with diabetes mellitus: systemic review and meta-analysis. Diabetologia. 2008;51:941–51.
Monami M, Lamanna C, Marchionni N, et al. CSII versus MDI in type 1 diabetes: a meta-analysis. Acta Diabetol. 2010;47:77–81.
Fredheim S, Johansen A, Thorsen SU, et al. Nationwide reduction in the frequency of severe hypoglycemia by half. Acta Diabetol. 2014;52:591–9.
Steineck I, Cederholm J, Eliasson B, et al. Insulin pump therapy, multiple daily injections, and cardiovascular mortality in 18168 people with type 1 diabetes: observational study. BMJ. 2015;350:h3234.
Garg S, Brazg RL, Bailey TS, et al. Reduction in duration of hypoglycemia by automatic suspension of insulin delivery: the in-clinic ASPIRE study. Diabetes Techol Ther. 2012;14:205–9.
Haidar A, Legault L, Mateau-Pelletier L, et al. Outpatient overnight glucose control with dual-hormone artificial pancreas, single-hormone artificial pancreas, or conventional insulin pump therapy in children and adolescents with type 1 diabetes: an open-label, randomized controlled trial. Lancet Diabetes Endocrinol. 2015;3:595–604.
Haidar A, Legault L, Messier V, Mitre TM, Leroux C, Rabasa-Lhoret R. Comparison of dual-hormone artificial pancreas, single-hormone artificial pancreas, and conventional insulin pump therapy for glycaemic control in patients with type 1 diabetes: an open-label randomized controlled crossover trial. Diabetes Endocrinol. 2015;3:17–26.
Leelarathna L, Dellweg S, Mader JK, et al. Day and night home closed-loop insulin delivery in adults with type 1 diabetes: three-center randomized crossover study. Diabetes Care. 2014;37:1931–7.
Nimri R, Muller I, Atlas E, et al. MD-Logic overnight control 6 weeks of home use in patients with type 1 diabetes: randomized crossover trial. Diabetes Care. 2014;37:3025–32.
Thabit H, Lubina-Solomon A, Stadler M, et al. Home use of closed-loop insulin delivery for overnight glucose control in adults with type 1 diabetes: a 4-week, multicentre, randomized crossover study. Lancet Diabetes Endocrinol. 2014;2:701–9.
Hermanides J, Engström AE, Wentholt IM, et al. Sensor-augmented insulin pump therapy to treat hyperglycemia at the coronary care unit: a randomized clinical pilot trial. Diabetes Technol Ther. 2010;12:537–42.
Rosenlund S, Hansen TW, Rossing P, Andersen S. Effect of sensor-augmented pump treatment versus multiple daily injections on albuminuria: a 1-year randomized study. J Clin Endocrinol Metab. 2015;100:4181–8.
Tsukamoto Y, Okabayashi T, Hanazaki K. Progressive artificial endocrine pancreas: the era of novel perioperative blood glucose control for surgery. Surg Today. 2011;41:1344–51.
Tsukamoto Y, Kinoshita Y, Kitagawa H, et al. Evaluation of a novel artificial pancreas: closed loop glycemic control system with continuous blood glucose monitoring. Artif Organ. 2013;37:E67–73.
Yamashita K, Okabayashi T, Yokoyama T, et al. The accuracy of continuous blood glucose monitor during surgery. Anesth Analg. 2008;106:160–3.
Yamashita K, Okabayashi T, Yokoyama T, et al. Accuracy and reliability of continuous blood glucose monitor in post-surgical patients. Acta Anaesthesiol Scand. 2009;53:66–71.
Hanazaki K, Maeda H, Okabayashi T. Relationship between perioperative glycemic control and postoperative infections. World J Gastroenterol. 2009;15:4122–5.
Hanazaki K. Tight glycemic control using an artificial endocrine pancreas may play an important role in preventing infection after pancreatic resection. World J Gastroenterol. 2012;18:3787–9.
Finney SJ, Zekveld C, Elia A, et al. Glucose control and mortality in critically ill patients. JAMA. 2003;290:2041–7.
Rady MY, Johnson DJ, Patel BM, et al. Influence of individual characteristics on outcome of glycemic control in intensive care unit patients with or without diabetes mellitus. Mayo Clin Proc. 2005;80:1558–67.
Gabbanelli V, Pantanetti S, Donati A, et al. Correlation between hyperglycemia and mortality in a medical and surgical intensive care unit. Minerva Anestesiol. 2005;71:717–25.
Ata A, Lee J, Bestle SL, et al. Postoperative hyperglycemia and surgical site infection in general surgery patients. Arch Surg. 2010;145:858–64.
Eshuis WJ, Hermanides J, van Dalen JW, et al. Early postoperative hyperglycemia is associated with postoperative complications after pancreatoduodenectomy. Ann Surg. 2011;253:739–44.
van den Berghe G. Insulin therapy in the intensive care unit should be targeted to maintain blood glucose between 4.4 and 6.1 mmol/l. Diabetologia. 2008;51:911–5.
Kono T, Hanazaki K, Yazawa K, et al. Pancreatic polypeptide administration reduces insulin requirements of artificial pancreas in pancreatectomized dogs. Artif Organs. 2005;29:83–7.
Workgroup on Hypoglycemia. American diabetes association. defining and reporting hypoglycemia in diabetes: a report from the american diabetes association workgroup on hypoglycemia. Diabetes Care. 2005;28:1245–9.
Hanazaki K, Yatabe T, Kobayashi M, et al. Perioperative glycemic control using an artificial endocrine pancreas in patients undergoing total pancreatectomy: tight glycemic control may be justified in order to avoid brittle diabetes. Biomed Mat Eng. 2013;23:109–16.
Mibu K, Yatabe T, Hanazaki K. Blood glucose control using an artificial pancreas reduces the workload of ICU nurses. J Artif Organs. 2012;15:71–6.
Egi M, Bellomo R, Stachowski E, French CJ, Hart G. Variability of blood glucose concentration and short-term mortality in critically ill patients. Anesthesiology. 2006;105:244–52.
Krinsley JS. Glycemic variability: a strong independent predictor of mortality in critically ill patients. Crit Care Med. 2008;36:3008–13.
Hermanides J, Vriesendrop TM, Bosman RJ, Zandstra DF, Hoekstra JB, Devries JH. Glucose variability is associated with intensive care unit mortality. Crit Care Med. 2010;38:838–42.
Mackenzie IMJ, Whitehouse T, Nightingale PG. The metrics of glycaemic control in critical care. Intensive Care Med. 2011;37:435–43.
Munekage M, Yatabe T, Kitagawa H, et al. An artificial pancreas provided a novel model of blood glucose level variability in beagles. J Artif Organs. 2015;18(4):387–90.
DeFronzo RA, Tobin JD, Andres R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Phys. 1979;237:E214–23.
Kwon S, Thompson R, Dellinger P, et al. Importance of perioperative glycemic control in general surgery: a report from the surgical care and outcomes assessment program. Ann Surg. 2013;257:8–14.
Frisch A, Chandra P, Smiley D, et al. Prevalence and clinical outcome of hyperglycemia in the perioperative period in noncardiac surgery. Diabetes Care. 2010;33:1783–8.
Kotagal M, Symons RG, Hirsch IB, et al. Perioperative hyperglycemia and risk of adverse events among patients with and without diabetes. Ann Surg. 2015;261:97–103.
Munekage M, Yatabe T, Sakaguchi M, et al. Comparison of subcutaneous and intravenous continuous glucose monitoring accuracy in an operating room and an intensive care unit. J Artif Organs. 2015. doi:10.1007/s10047-015-0877-2.
Malchesky PS. Artificial organs and vanishing boundaries. Artif Organs. 2001;25:75–8.
Malchesky PS. Artificial organs 2014: a year in review. Artif Organs. 2015;39:260–87.
Welman T, Michel S, Segaren N, Shanmugarajah K. Bioengineering for organ transplantation: progress and challenges. Bioengineered. 2015;6:257–61.
Chakkera HA, Kudva YC, Chang YH et al. (2015) Glucose homeostasis after simultaneous pancreas and kidney transplantation: a comparison of subjects with C-peptide positive non-type 1 diabetes mellitus and type 1 diabetes mellitus. Clin Transplant. Oct 20 [Epub ahead of print].
Staels W, De Groef S, Heremans Y, et al. Accessory cells for β-cell transplantation. Diabetes Obes Metab. 2016;18:115–24.
Okita K, Mizuguchi T, Shigenori O, et al. Pancreatic regeneration: basic research and gene regulation. Surg Today. 2016;46:633–40.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hanazaki, K., Munekage, M., Kitagawa, H. et al. Current topics in glycemic control by wearable artificial pancreas or bedside artificial pancreas with closed-loop system. J Artif Organs 19, 209–218 (2016). https://doi.org/10.1007/s10047-016-0904-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-016-0904-y