Abstract
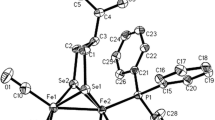
A series of diphosphonate-bridged dipalladacycles [(phpy)Pd(EtO)2P(O)]2, [(bhq)Pd(EtO)2P(O)]2, [(phpz)Pd(EtO)2P(O)]2 (phpy=2-phenylpyridine, bhq=benzo[h]quinoline, phpz=1-phenylpyrazole), which are known to be involved in catalytic C–H phosphonation reactions, was prepared and characterized by NMR spectroscopy and cyclic voltammetry in acetonitrile solutions and in carbon paste electrode. Diphosphonate dipalladacycles are oxidized irreversibly at more positive potentials as distinguished from related acetate palladacycles. Electrochemical preparative oxidations carried out under mild conditions without any specially added oxidants quantitatively afforded corresponding arylphosphonates. For complete conversion of dipalladacycles into arylphosphonates, four electrons per each palladium atom are required, that probably indicates a mechanism involving Pd(IV)/Pd(II) redox couple.






Similar content being viewed by others
References
Henry PM (1980) Palladium catalyzed oxidation of hydrocarbons. D. Reidel, Boston
Negishi E (2002) Handbook of organopalladium chemistry for organic synthesis. Wiley, Hoboken
Hartwig JF (2010) Organotransition metal chemistry: from bonding to catalysis. University Science Books, Sausalito
Miyaura N, Suzuki A (1995) Chem Rev 95:2457–2483
Stahl SS (2004) Angew Chem Int Ed 43:3400–3420
Stoltz BM (2004) Chem Lett 33:362–367
Gligorich KM, Sigman MS (2009) Chem Commun: 3854–3867
Dick AR, Hull KL, Sanford MS (2004) J Am Chem Soc 126:2300–2301
Canty A (2009) Dalton Trans: 10409–10417
Muniz K (2009) Angew Chem Int Ed 48:9412–9423
Lyons TW, Sanford MS (2010) Chem Rev 110:1147–1169
Sehnal P, Taylor RJK, Fairlamb IJS (2010) Chem Rev 110:824–889
Xu L-M, Li B-J, Yang Z, Shi Z-J (2010) Chem Soc Rev 39:712–733
Byers PK, Canty AJ, Skelton BW, White AH (1986) Chem Commun:1722–1724
Canty AJ (1992) Acc Chem Res 25:83–90
Alsters PL, Engel PF, Hogerheide MP, Copijn M, Spek AL, van Koten G (1993) Organometallics 12:1831–1844
van Asselt R, Rijnberg E, Elsevier CJ (1994) Organometallics 13:706–720
van Belzen R, Hoffmann H, Elsevier CJ (1997) Angew Chem Int Ed 36:1743–1745
Lagunas MC, Gossage RA, Spek AL, van Koten G (1998) Organometallics 17:731–741
Yamamoto Y, Ohno T, Itoh K (2002) Angew Chem Int Ed 41:3662–3665
Yamamoto Y, Kuwabara S, Matsuo S, Ohno T, Nishiyama H, Itoh K (2004) Organometallics 23:3898–3906
Campora J, Palma P, del Rio D, Carmona E, Graiff C, Tiripicchio A (2003) Organometallics 22:3345–3347
Campora J, Palma P, del Rio D, Lopez JA, Valerga P (2004) Chem Commun: 1490–1491
Powers DC, Ritter T (2011) Top Organomet Chem (2011) 35:129–156
Mirica LM, Khusnutdinova JR (2013) Coord Chem Rev 257:299–314
Hickman AJ, Sanford MS (2012) Nature 484:177–185
Kargin YM, Budnikova YH, Martynov BI, Turygin VV, Tomilov AP (2001) J Electroanal Chem 507:157–169
Budnikova YG, Yakhvarov DG, Kargin YM (1997) Mendeleev Commun: 67-68
Klein A, Budnikova YH, Sinyashin OG (2007) J Organomet Chem 692:3156–3166
Budnikova Y, Kargin Y, Nedelec JY (1999) J Organomet Chem 575:63–66
Jutand A (2008) Chem Rev 108:2300–2347
Bercaw JE, Durrell AC, Gray HB, Green JC, Hazari N, Labinger JA, Winkler JR (2010) Inorg Chem 49:1801–1810
Dudkina YB, Mikhaylov DY, Gryaznova TV, Tufatullin AI, Kataeva ON, Vicic DA, Budnikova YH (2013) Organometallics 32:4785–4792
Dudkina YB, Mikhaylov DY, Gryaznova TV, Sinyashin OG, Vicic DA, Budnikova YH (2012) Eur J Org Chem: 2114–2117
Budnikova YH, Dudkina YB, Khrizanforov MN, Gryaznova TV (2014) J Organomet Chem 751:301–305
Chuang GJ, Wang W, Lee E, Ritter T (2011) J Am Chem Soc 133:1760–1762
Berry JF, Cotton FA, Ibragimov SA, Murillo CA, Wang X (2005) Inorg Chem 44:6129–6137
Powers DC, Geibel MAL, Klein JEMN, Ritter T (2009) J Am Chem Soc 131:17050–17051
Li C, Yano T, Ishida N, Murakami M (2013) Angew Chem Int Ed 52:9801–9804
Grayaznova TV, Dudkina YB, Islamov DR, Kataeva ON, Sinyashin OG, Vicic DA, Budnikova YН (2015) J Organomet Chem 785:68–71
Powers DC, Ritter T (2009) Nat Chem 1:302–309
Serrano JL, García L, Pérez J, Pérez E, Galiana JM, García J, Martínez M, Sánchez G, da Silva I (2011) Dalton Trans 40:156–168
Campbell MG, Powers DC, Raynaud J, Graham MJ, Xie P, Lee E, Ritter T (2011) Nat Chem 3:949–953
Kohler MC, Stockland RA Jr, Rath NP (2006) Organometallics 25:5746–5756
Stockland RA Jr, D.Wilson B, Goodman CC, Giese BJ, Shrimp FL II (2007) J Chem Educ 84:694–696
Acknowledgments
The work is supported by the Russian Science Foundation (grant no. 14-23-00016).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gryaznova, T., Dudkina, Y., Khrizanforov, M. et al. Electrochemical properties of diphosphonate-bridged palladacycles and their reactivity in arene phosphonation. J Solid State Electrochem 19, 2665–2672 (2015). https://doi.org/10.1007/s10008-015-2875-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-015-2875-y