Abstract
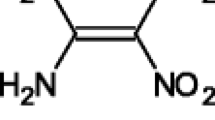
Cyclic voltammetric and EPR/UV-vis-NIR spectroelectrochemical studies were performed to examine the cathodic reduction of 2,6-dichlorophenolindophenolate (DCIP) in proton-donating aqueous and methanol solutions, as well as in aprotic dimethylsulfoxide (DMSO), and to characterize the paramagnetic species generated upon the DCIP reduction. In situ EPR and UV-vis-NIR spectroelectrochemistry confirmed the formation of the radical anion DCIP•– in DMSO and methanol. The same radical anion was found also in the reaction system consisting of KO2 mixed under argon with DCIP in DMSO or methanol, evidencing the electron transfer from superoxide radical anion to DCIP. The expected radical anion DCIP•– was not detected in the photoexcited suspensions DCIP/TiO2/DMSO under argon, which indicates fast consecutive reactions of photogenerated DCIP•– in the vicinity of TiO2 surface. The reduction of blue-color DCIP to the final colorless product DCIPH2 can be realized in multiple reaction pathways determined mainly by the proton-donating capacity of the solvent. Following the calculated total DFT energies, the oxygen on the indophenol moiety represents the first proton acceptor site for DCIP, DCIP•–, as well as for DCIP2– species.











Similar content being viewed by others
References
Cohen B, Gibbs HD, Clark WM (1924) Public Health Rep 39:381–414
Bidoia ED, Mantagnolli RN, Lopes PRM (2010) In: Mendez-Vilas A (ed) Current research, technology and education topics in applied microbiology and microbial biotechnology, vol 2. Formatex Research Center, Badajoz, pp 1277–1288
Cabello C, Bair W, Bause A, Wondrak G (2009) Biochem Pharmacol 78:344–354
Kumar S, Acharya S (1999) Anal Biochem 268:89–93
Aiuchi T, Nakajo S, Nakaya K (2004) Biol Pharm Bull 27:736–738
Kong C, Li DW, Li Y, Partovi-Nia R, James TD, Long YT, Tian H (2012) Analyst 137:1094–1096
Florou AB, Prodromidis MI, Karayannis MI, Tzouwara-Karayanni SM (1998) Electroanalysis 10:1261–1268
Florou AB, Prodromidis MI, Karayannis MI, Tzouwara-Karayanni SM (2000) Electroanalysis 12:361–368
Mills A, McGrady M, Wang J, Hepburn J (2008) Int J Photoenergy ID 504945
Brezová V, Čeppan M, Veselý M, Lapčík L (1991) Chem Pap 45:233–246
Mills A, McGrady M (2008) J Photochem Photobiol A Chem 193:228–236
Krýsa J, Baudys M, Mills A (2015) Catal Today 240:132–137
Kafizas A, Mills A, Parkin IP (2010) Anal Chim Acta 663:69–76
Prokof'ev AN, Solodovnikov SP, Nikiforov GA, Ershov VV (1971) Bull Acad Sci USSR Div Chem Sci 20:262–265
Duling DR (1994) J Magn Reson B 104:105–110
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Vosko SH, Wilk L, Nusair M (1980) Can J Phys 58:1200–1211
Stephens PJ, Devlin FJ, Chabalowski CF, Frisch MJ (1994) J Phys Chem 98:11623–11627
Hehre WJ, Ditchfield K, Pople JA (1972) J Chem Phys 56:2257–2261
Francl MM, Pietro WJ, Hehre WJ, Binkley JS, Gordon MS, DeFrees DJ, Pople JA (1982) J Chem Phys 77:3654–3665
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J Chem Phys 72:650–654
McLean AD, Chandler GS (1980) J Chem Phys 72:5639–5648
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, Revision A,1. Gaussian Inc, Pittsburgh
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2013) Gaussian 09, Revision D.01. Gaussian Inc, Wallingford
Miertuš S, Scrocco E, Tomasi J (1981) Chem Phys 55:117–129
Barone V, Cossi M, Tomasi J (1997) J Chem Phys 107:3210–3221
Bauernschmitt R, Ahlrichs R (1996) Chem Phys Lett 256:454–464
Runge E, Gross EKU (1984) Phys Rev Lett 52:997–1000
Barone V (1995) In: Chong DP (ed) Recent advances in density functional methods. Part 1. World Scientific, Singapore
Gosser DK (1993) Cyclic voltammetry: simulation and analysis of reaction mechanisms. VCH
Laviron E (1984) J Electroanal Chem 164:213–227
Rao PS, Hayon E (1973) J Phys Chem 77:2753–2756
Fujishima A, Zhang X, Tryk D (2008) Surf Sci Rep 63:515–582
Brezová V, Tarábek P, Dvoranová D, Staško A, Biskupič S (2003) J Photochem Photobiol A Chem 155:179–198
Dvoranová D, Barbieriková Z, Brezová V (2014) Molecules 19:17279–17304
Micic O, Zhang Y, Cromack K, Trifunac A, Thurnauer M (1993) J Phys Chem 97:13284–13288
Di Paola A, Bellardita M, Palmisano L, Barbieriková Z, Brezová V (2014) J Photochem Photobiol A Chem 273:59–67
Bahnemann D, Hilgendorff M, Memming R (1997) J Phys Chem B 101:4265–4275
Barbieriková Z, Mihalíková M, Brezová V (2012) Photochem Photobiol 88:1442–1454
Brezová V, Gabčová S, Dvoranová D, Staško A (2005) J Photochem Photobiol B Biol 79:121–134
Buettner GR (1987) Free Radic Biol Med 3:259–303
Wardman P (1989) J Phys Chem Ref Data 18:1637–1755
Brezová V, Blažková A, Šurina I, Havlínová B (1997) J Photochem Photobiol A Chem 107:233–237
Acknowledgments
This work was supported by the Scientific Grant Agency of the Slovak Republic (Projects VEGA/1/0041/15, VEGA/1/0307/14, VEGA/1/0327/12). The calculations were performed at HPC center, SUT Bratislava (SIVVP project, ITMS code 26230120002, funded by the European Region Development Funds) and Computing Centre SAS, code 26210120002 (Slovak infrastructure for high-performance computing) supported by the Research & Development Operational Program funded by the ERDF.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Prof. Mikhail A. Vorotyntsev in the occasion of his 70th birthday.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 656 kb)
Rights and permissions
About this article
Cite this article
Dvoranová, D., Barbieriková, Z., Dorotíková, S. et al. Redox processes of 2,6-dichlorophenolindophenolate in different solvents. A combined electrochemical, spectroelectrochemical, photochemical, and theoretical study. J Solid State Electrochem 19, 2633–2642 (2015). https://doi.org/10.1007/s10008-015-2823-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-015-2823-x