Abstract
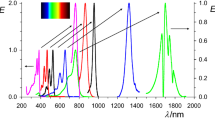
Due to unpaired electrons, both radicals and superalkali are investigated widely. In this work, two interesting complexes (Li3O-PLY and Li3-PLY) were constructed by phenalenyl radical and superalkali atoms. Why are they interesting? Firstly, for Li3O-PLY and Li3-PLY, although the charge transfer between superalkali atoms and PLY is similar, the sandwich-like charge distribution for Li3O-PLY causes a smaller dipole moment than that of Li3-PLY. Secondly, their UV–vis absorption show that the maximum wavelengths for Li3O-PLY and Li3-PLY display a bathochromic shift compared to PLY. Moreover, Li3-PLY has two new peaks at 482 and 633 nm. Significantly, the β 0 values of Li3-PLY (4943–5691 a.u.) are much larger than that of Li3O-PLY (225–347 a.u.). Further, the β HRS values of Li3O-PLY decrease slightly while β HRS of Li3-PLY increase dramatically with increasing frequency. It is our expectation that these results might provide beneficial information for theoretical and experimental studies on complexes with superalkali and PLY radicals.

Two interesting complexes (Li3O-PLY and Li3-PLY) were constructed by phenalenyl radical and superalkali atoms. We explore their structures, Wiberg bond indices, interaction energies and the static first hyperpolarizabilities (β 0). The β 0 values of Li3-PLY (4943–5691 a.u.) were much larger than those of Li3O-PLY (225–347 a.u.).






Similar content being viewed by others
References
Suzuki S, Morita Y, Fukui K, Sato K, Shiomi D, Takui T, Nakasuji K (2006) Aromaticity on the pancake-bonded dimer of neutral phenalenyl radical as studied by MS and NMR spectroscopies and NICS analysis. J Am Chem Soc 128:2530
Huang J, Kertesz M (2007) Intermolecular covalent π−π bonding interaction indicated by bond distances, energy bands, and magnetism in biphenalenyl biradicaloid molecular crystal. J Am Chem Soc 129:1634
Craciun S, Donald KJ (2009) Radical bonding: structure and stability of bis(phenalenyl) complexes of divalent metals from across the periodic table. Inorg Chem 48:5810
Small D, Rosokha SV, Kochi JK, Head-Gordon M (2005) Characterizing the dimerizations of phenalenyl radicals by ab initio calculations and spectroscopy: σ-bond formation versus resonance π-stabilization. J Phys Chem A 109:11261
Nakano M, Takebe A, Kishi R, Fukui H, Minami T, Kubota K, Takahashi H, Kubo T, Kamada K, Ohta K, Champagne B, Botek E (2008) Intermolecular interaction effects on the second hyperpolarizability of open-shell singlet diphenalenyl radical dimer. Chem Phys Lett 454:97
Chi X, Itkis ME, Patrick BO, Barclay TM, Reed RW, Oakley RT, Cordes AW, Haddon RC (1999) The first phenalenyl-based neutral radical molecular conductor. J Am Chem Soc 121:10395
Itkis M, Chi X, Cordes A, Haddon R (2002) Magneto-opto-electronic bistability in a phenalenyl-based neutral radical. Science 296:1443
Cyrański MK, Havenith RWA, Dobrowolski MA, Gray BR, Krygowski TM, Fowler PW, Jenneskens LW (2007) The phenalenyl motif: a magnetic chameleon. Chem Eur J 13:2201
Ueda A, Wasa H, Suzuki S, Okada K, Sato K, Takui T, Morita Y (2012) Chiral stable phenalenyl radical: synthesis, electronic-spin structure, and optical properties of [4]helicene-structured diazaphenalenyl. Angew Chem-Int Edit 51:6691
Sarkar A, Itkis ME, Tham FS, Haddon RC (2011) Synthesis, structure, and physical properties of a partial π-stacked phenalenyl-based neutral radical molecular conductor. Chem Eur J 17:11576
Li Z-R, Wang F-F, Wu D, Li Y, Chen W, Sun X-Y, Gu FL, Aoki Y (2006) Royal crown-shaped electride Li3-N3-Be containing two superatoms: new knowledge on aromaticity. J Comput Chem 27:986
Wang B-Q, Li Z-R, Wu D, Wang F-F (2007) Structures and static electric properties of novel alkalide anions F-Li+Li- and F−Li3 +Li3. J Phys Chem A 111:6378
Tong J, Li Y, Wu D, Wu Z-J (2012) Theoretical study on polynuclear superalkali cations with various functional groups as the central core. Inorg Chem 51:6081
Tong J, Wu Z, Li Y, Wu D (2013) Prediction and characterization of novel polynuclear superalkali cations. Dalton T 42:577
Cochran E, Meloni G (2014) Hypervalence in monoxides and dioxides of superalkali clusters. J Chem Phys 140:204–319
Sun W-M, Fan L-T, Li Y, Liu J-Y, Wu D, Li Z-R (2014) On the potential application of superalkali clusters in designing novel alkalides with large nonlinear optical properties. Inorg Chem 53:6170
Yokoyama K, Tanaka H, Kudo H (2001) Structure of hyperlithiated Li3O and evidence for electronomers. J Phys Chem A 105:4312
Đustebek J, Veličković S, Veljković F, Veljković M (2012) Production of heterogeneous superalkali clusters LinF (n = 2–6) by knudesn-cell mass spectrometry. Dig J Nano Mater Bios 7:1365
Li Y, Wu D, Li Z-R (2008) Compounds of superatom clusters: preferred structures and significant nonlinear optical properties of the BLi6-X (X = F, LiF2, BeF3, BF4) motifs. Inorg Chem 47:9773
Tong J, Li Y, Wu D, Li Z-R, Huang X-R (2011) Ab initio investigation on a new class of binuclear superalkali cations M2Li2k+1+ (F2Li3 +, O2Li5 +, N2Li7 +, and C2Li9 +). J Phys Chem A 115:2041
Kudo H, Wu CH, Ihle HR (1978) Mass-spectrometric study of the vaporization of Li2O(s) and thermochemistry of gaseous LiO, Li2O, Li3O, and Li2O2. J Nucl Mater, J 78:380
Wu CH, Kudoa H, Ihle HR (1979) Thermochemical properties of gaseous Li3O and Li2O2. J Chem Phys 70:1815
Alexandrova AN, Boldyrev AI (2003) σ-Aromaticity and σ-antiaromaticity in alkali metal and alkaline earth metal small clusters. J Phys Chem A 107:554
Karplus M, Porter RN (1970) Atoms and molecules; an introduction for students of physical chemistry. Benjamin, New York
Zhong R-L, Zhang J, Muhammad S, Hu Y-Y, Xu H-L, Su Z-M (2011) Boron/nitrogen substitution of the central carbon atoms of the biphenalenyl diradical π dimer: a novel 2e–12c bond and large NLO responses. Chem Eur J 17:11773
Tian Y-H, Huang J, Kertesz M (2010) Fluxional [sigma]-bonds of 2,5,8-tri-tert-butyl-1,3-diazaphenalenyl dimers: stepwise [3,3], [5,5] and [7,7] sigmatropic rearrangements via [small pi]-dimer intermediates. Phys Chem Chem Phys 12:5084
Tian Y-H, Kertesz M (2010) Is there a lower limit to the CC bonding distances in neutral radical π-dimers? The case of phenalenyl derivatives. J Am Chem Soc 132:10648
Sini G, Sears JS, Brédas J-L (2011) Evaluating the performance of DFT functionals in assessing the interaction energy and ground-state charge transfer of donor/acceptor complexes:tetrathiafulvalene − tetracyanoquinodimethane (TTF − TCNQ) as a model case. J Chem Theory Comput 7:602
Zhong R-L, Xu H-L, Sun S-L, Qiu Y-Q, Zhao L, Su Z-M (2013) Theoretical investigation on the 2e/12c bond and second hyperpolarizability of azaphenalenyl radical dimers: strength and effect of dimerization. J Chem Phys 139, 124314
Chen S, Sun S-L, Wu H-Q, Xu H-L, Zhao L, Su Z-M (2014) Superatoms (Li3O and BeF3) induce phenalenyl radicalπ–dimer: fascinating interlayer charge-transfer and large NLO responses. Dalton T 43:12657
Alkorta I, Elguero J (1998) Theoretical study of strong hydrogen bonds between neutral molecules: the case of amine oxides and phosphine oxides as hydrogen bond acceptors. J Phys Chem A 103:272–279
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566
Chen W, Li Z-R, Wu D, Li Y, Sun C-C, Gu FL (2005) The structure and the large nonlinear optical properties of Li@Calix[4]pyrrole. J Am Chem Soc 127:10977–10981
Xu H-L, Li Z-R, Wu D, Wang B-Q, Li Y, Gu FL, Aoki Y (2007) Structures and large NLO responses of new electrides: Li-doped fluorocarbon chain. J Am Chem Soc 129:2967
Muhammad S, Xu H, Liao Y, Kan Y, Su Z (2009) Quantum mechanical design and structure of the Li@B10H14 basket with a remarkably enhanced electro-optical response. J Am Chem Soc 131:11833
Dongdong Qi. NLO Calculator, Version 0.2. University of Science and Technology Beijing, Beijing 100083, China
Zhang L, Qi D, Zhao L, Chen C, Bian Y, Li W (2012) Density functional theory study on subtriazaporphyrin derivatives: dipolar/octupolar contribution to the second order nonlinear optical activity. J Phys Chem A 116:10249
Frisch MJ, Trucks GW, Schlegel HB et al (2009) Gaussian 09, revision A.02; Gaussian, Inc. Wallingford, CT
Oudar JL, Chemla DS (1977) Hyperpolarizabilities of the nitroanilines and their relations to the excited state dipole moment. J Chem Phys 66:2664
Oudar JL (1977) Optical nonlinearities of conjugated molecules. Stilbene derivatives and highly polar aromatic compounds. J Chem Phys 67:446
Gorelsky SI (2009) AOMix version 6.46: program for molecular orbital analysis, University of Ottawa. Available at: http://www.sg-chem.net/
Gorelsky SI, Lever ABP (2001) J Organomet Chem 635:187
Acknowledgments
The authors gratefully acknowledge the financial support from National Science Foundation of China (NSFC) (21003019, 21473026), the Science and Technology Development Planning of Jilin Province (201201062 and 20140101046JC), the Computing Center of Jilin Province provided essential support and H.-L.X. acknowledges support from the Hong Kong Scholars Program And Project funded by the China Postdoctoral Science Foundation (2014 M560227).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, S., Xu, HL., Sun, SL. et al. Superalkali atoms bonding to the phenalenyl radical: structures, intermolecular interaction and nonlinear optical properties. J Mol Model 21, 209 (2015). https://doi.org/10.1007/s00894-015-2750-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2750-x