Abstract
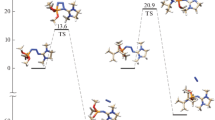
DNA phosphotriester adducts are common alkylation products of DNA phosphodiester moiety induced by N-nitrosoureas. The 2-hydroxyethyl phosphotriester was reported to hydrolyze more rapidly than other alkyl phosphotriesters both in neutral and in alkaline conditions, which can cause DNA single strand scission. In this work, DFT calculations have been employed to map out the four lowest activation free-energy profiles for neutral and alkaline hydrolysis of triethyl phosphate (TEP) and diethyl 2-hydroxyethyl phosphate (DEHEP). All the hydrolysis pathways were illuminated to be stepwise involving an acyclic or cyclic phosphorane intermediate for TEP or DEHEP, respectively. The rate-limiting step for all the hydrolysis reactions was found to be the formation of phosphorane intermediate, with the exception of DEHEP hydrolysis in alkaline conditions that the decomposition process turned out to be the rate-limiting step, owing to the extraordinary low formation barrier of cyclic phosphorane intermediate catalyzed by hydroxide. The rate-limiting barriers obtained for the four reactions are all consistent with the available experimental information concerning the corresponding hydrolysis reactions of phosphotriesters. Our calculations performed on the phosphate triesters hydrolysis predict that the lower formation barriers of cyclic phosphorane intermediates compared to its acyclic counter-part should be the dominant factor governing the hydrolysis rate enhancement of DEHEP relative to TEP both in neutral and in alkaline conditions.
















Similar content being viewed by others
References
Gnewuch CT, Sosnovsky G (1997) Chem Rev 97:829–1014
Wang PG, Xian M, Tang X, Wu X, Wen Z, Cai T, Janczuk AJ (2002) Chem Rev 102:1091–1134
Morimoto K, Tanaka A, Yamaha T (1983) Carcinogenesis 4:1455–1458
Singer B, Grunberger D (1983) Molecular biology of mutagens and carcinogens. Plenum, New York
Saffhill R (1984) Carcinogenesis 5:621–625
Beranek DT (1990) Mutat Res 231:11–30
Singer B (1976) Nature 264:333–339
Singer B, Spengler S, Bodell WJ (1981) Carcinogenesis 2:1069–1073
Swenson DH, Harbach PR, Trzos RJ (1980) Carcinogenesis 1:931–936
Singer B (1975) Progress in nucleic acid research and molecular biology. Academic, New York
Lown JW, McLaughlin LW (1979) Biochem Pharmacol 28:1631–1638
Walles S, Ehrenberg L (1968) Acta Chem Scand 22:2727–2729
Gutin PH, Hilton J, Fein VJ, Allan AE, Rottman A, Walker MD (1977) Cancer Res 37:3761–3765
Erickson LC, Bradley MO, Kohn KW (1977) Cancer Res 37:3744–3750
Hilton J, Bowie DL, Gutin PH, Zito DM, Walker MD (1977) Cancer Res 37:2262–2266
Gamper HB, Tung ASC, Straub K, Bartholomew JC, Calvin M (1977) Science 197:671–674
Swenson DH, Frei JV, Lawley PD (1979) J Natl Cancer Inst 63:1469–1473
Brown DM, Todd AR (1952) J Chem Soc p 44–51
Lawley PD, Brookes P (1963) Biochem J 89:127–138
Bannon P, Verly WG (1972) Eur J Biochem 31:103–111
Shooter KV (1976) Chem Biol Interact 13:151–163
Swenson DH, Farmer PB, Lawley PD (1976) Chem Biol Interact 15:91–100
Conrad J, Muller N, Eisenbrand G (1986) Chem Biol Interact 60:57–65
Zeller WJ, Lijinsky W, Eisenbrand G (1985) J Cancer Res Clin Oncol 109:A46
Zeller WJ, Fruhauf S, Chen G, Eisenbrand G, Lijinsky W (1989) Cancer Res 49:3267–3270
Lopez X, Dejaegere A, Karplus M (2001) J Am Chem Soc 123:11755–11763
Kirby AJ, Lima MF, da Silva D, Nome F (2004) J Am Chem Soc 126:1350–1351
Kirby AJ, Dutta-Roy N, da Silva D, Goodman JM, Lima MF, Roussev CD, Nome F (2005) J Am Chem Soc 127:7033–7040
Kirby AJ, Lima MF, da Silva D, Roussev CD, Nome F (2006) J Am Chem Soc 128:16944–16952
Klahn M, Rosta E, Warshel A (2006) J Am Chem Soc 128:15310–15323
Berente I, Beke T, Naray-Szabo G (2007) Theor Chem Acc 118:129–134
Rosta E, Kamerlin SCL, Warshel A (2008) Biochemistry 47:3725–3735
Yang Y, Yu HB, York D, Elstner M, Cui Q (2008) J Chem Theor Comput 4:2067–2084
Kirby AJ, Tondo DW, Medeiros M, Souza BS, Priebe JP, Lima MF, Nome F (2009) J Am Chem Soc 131:2023–2028
Kamerlin SCL, Williams NH, Warshel A (2008) J Org Chem 73:6960–6969
Florian J, Warshel A (1998) J Phys Chem B 102:719–734
Aqvist J, Kolmodin K, Florian J, Warshel A (1999) Chem Biol 6:R71–R80
Menegon G, Loos M, Chaimovich H (2002) J Phys Chem A 106:9078–9084
Chang NY, Lim C (1997) J Phys Chem A 101:8706–8713
Chang NY, Lim C (1998) J Am Chem Soc 120:2156–2167
Iche-Tarrat N, Barthelat JC, Rinaldi D, Vigroux A (2005) J Phys Chem B 109:22570–22580
Iche-Tarrat N (2010) J Mol Struc THEOCHEM 941:56–60
Brown DM, Todd AR (1952) J Chem Soc p 52–58
Zhou DM, Taira K (1998) Chem Rev 98:991–1026
Lim C, Tole P (1992) J Am Chem Soc 114:7245–7252
Lim C, Tole P (1992) J Phys Chem 96:5217–5219
Tole P, Lim CM (1993) J Phys Chem 97:6212–6219
Tole P, Lim C (1994) J Am Chem Soc 116:3922–3931
Liu Y, Gregersen BA, Lopez X, York DM (2005) J Phys Chem B 109:19987–20003
Lopez CS, Faza ON, de Lera AR, York DM (2005) Chem Eur J 11:2081–2093
Liu Y, Gregersen BA, Hengge A, York DM (2006) Biochemistry 45:10043–10053
Lopez X, Dejaegere A, Leclerc F, York DM, Karplus M (2006) J Phys Chem B 110:11525–11539
Lopez X, Schaefer M, Dejaegere A, Karplus M (2002) J Am Chem Soc 124:5010–5018
Range K, McGrath MJ, Lopez X, York DM (2004) J Am Chem Soc 126:1654–1665
Westheimer FH (1968) Acc Chem Res 1:70–78
Brown DM, Usher DA (1963) Proc Chem Soc p 309–310
Brown DM, Usher DA (1965) J Am Chem Soc p 6547–6558
Brown DM (1974) Basic principles in nucleic acid chemistry. Academic, New York
Kuimelis RG, McLaughlin LW (1998) Chem Rev 98:1027–1044
Tomasi J, Persico M (1994) Chem Rev 94:2027–2094
Cossi M, Barone V, Cammi R, Tomasi J (1996) Chem Phys Lett 255:327–335
Mineva T, Russo N, Sicilia E (1998) J Comput Chem 19:290–299
Becke AD (1993) J Chem Phys 98:5648–5652
Lee CT, Yang WT, Parr RG (1988) Phys Rev B 37:785–789
Cossi M, Scalmani G, Rega N, Barone V (2002) J Chem Phys 117:43–54
Cossi M, Rega N, Scalmani G, Barone V (2003) J Comput Chem 24:669–681
Banas P, Jurecka P, Walter NG, Sponer J, Otyepka M (2009) Methods 49:202–216
Rios-Font R, Rodriguez-Santiago L, Bertran J, Sodupe M (2007) J Phys Chem B 111:6021–6077
Mayaan E, Range K, York DM (2004) J Biol Inorg Chem 9:807–817
Liu Y, Lopez X, York DM (2005) Chem Commun 31:3909–3911
Tidor B, Karplus M (1994) J Mol Biol 238:405–414
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussion 09, Revision B.01. Gaussian Inc, Wallingford
Aksnes G, Bergesen K (1966) Acta Chem Scand 20:2508–2514
Thatcher GRJ, Kluger R (1989) Adv Phys Org Chem 25:99–265
Kluger R, Covitz F, Dennis E, Williams LD, Westheimer FH (1969) J Am Chem Soc 91:6066–6072
Kluger R, Taylor SD (1990) J Am Chem Soc 112:6669–6671
Kluger R, Taylor SD (1991) J Am Chem Soc 113:5714–5719
Shiiba T, Komiyama M (1992) Tetrahedron Lett 33:5571–5574
Kuimelis RG, McLaughlin LW (1995) Nucleic Acids Res 23:4753–4760
Kuimelis RG, McLaughlin LW (1995) J Am Chem Soc 117:11019–11020
Kuimelis RG, McLaughlin LW (1996) Biochemistry 35:5308–5317
Thomson JB, Patel BK, Jimenez V, Eckart K, Eckstein F (1996) J Org Chem 61:6273–6281
Kuimelis RG, McLaughlin LW (1997) Bioorgan Med Chem 5:1051–1061
Acknowledgments
This work was supported by the grants from the National Natural Science Foundation of China (No. 20907002), the Beijing Nova Program (No. 2009B08) and the Training Program Foundation for the Beijing Municipal Natural Science Foundation (No. 8113031).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 417 kb)
Rights and permissions
About this article
Cite this article
Liu, T., Zhao, L. & Zhong, R. DFT investigations of phosphotriesters hydrolysis in aqueous solution: a model for DNA single strand scission induced by N-nitrosoureas. J Mol Model 19, 647–659 (2013). https://doi.org/10.1007/s00894-012-1592-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1592-z