Abstract
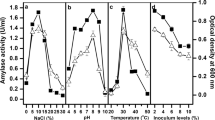
An ionic liquid-tolerant bacterium, Bacillus amyloliquefaciens CMW1, was isolated from a Japanese fermented soybean paste. Strain CMW1 grew in the presence of 10 % (v/v) 1-butyl-3-methylimidazolium chloride ([BMIM]Cl), a commonly used ionic liquid. Additionally, strain CMW1 grew adequately in the presence of the hydrophilic ionic liquids 10 % (v/v) 1-ethyl-3-methylimidazolium trifluoromethanesulfonate ([EMIM]CF3SO3) or 2.5 % (v/v) 1-butyl-3-methylimidazolium trifluoromethanesulfonate ([BMIM]CF3SO3). Strain CMW1 produced an extracellular protease (BapIL) in the culture medium. BapIL was stable in the presence of 80 % (v/v) ionic liquids, [EMIM]CF3SO3, [BMIM]Cl, [BMIM]CF3SO3, 1-butyl-3-methylimidazolium tetrafluoroborate, 1-butyl-3-methylimidazolium hexafluorophosphate, and 1-butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide, and functioned in 10 % (v/v) these ionic liquids. BapIL was stable at pH 4.0–12.6 or in 4004 mM NaCl solution, and exhibited activity in the presence of 50 % (v/v) hydrophilic or hydrophobic organic solvents. BapIL was completely inhibited by 1 mM PMSF and partially by 5 mM EDTA. BapIL belongs to the true subtilisins according to analysis of the deduced amino acid sequence. We showed that BapIL from the ionic liquid-tolerant B. amyloliquefaciens CMW1 exhibited tolerance to ionic liquid and halo, alkaline, and organic solvents.





Similar content being viewed by others
References
Attri P, Venkatesu P, Kumar A (2011) Activity and stability of α-chymotrypsin in biocompatible ionic liquids: enzyme refolding by triethyl ammonium acetate. Phys Chem Chem Phys 13:2788–2796
Bendtsen JD, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340:783–795
Brar SK, Verma M, Tyagi R, Surampalli R, Barnabé S, Valéro J (2007) Bacillus thuringiensis proteases: production and role in growth, sporulation and synergism. Process Biochem 42:773–790
Castro GR (1999) Enzymatic activities of proteases dissolved in organic solvents. Enzyme Microb Technol 25:689–694
Cornmell RJ, Winder CL, Tiddy GJ, Goodacre R, Stephens G (2008) Accumulation of ionic liquids in Escherichia coli cells. Green Chem 10:836–841
Dabirmanesh B, Daneshjou S, Sepahi AA, Ranjbar B, Khavari-Nejad RA, Gill P, Heydari A, Khajeh K (2011) Effect of ionic liquids on the structure, stability and activity of two related α-amylases. Int J Biol Macromol 48:93–97
Docherty KM, Dixon JK, Kulpa CF Jr (2007) Biodegradability of imidazolium and pyridinium ionic liquids by an activated sludge microbial community. Biodegradation 18:481–493
Gupta R, Beg Q, Khan S, Chauhan B (2002) An overview on fermentation, downstream processing and properties of microbial alkaline proteases. Appl Microbiol Biotechnol 60:381–395
Kaar JL, Jesionowski AM, Berberich JA, Moulton R, Russell AJ (2003) Impact of ionic liquid physical properties on lipase activity and stability. J Am Chem Soc 125:4125–4131
Khudyakov JI, D’haeseleer P, Borglin SE, DeAngelis KM, Woo H, Lindquist EA, Hazen TC, Simmons BA, Thelen MP (2012) Global transcriptome response to ionic liquid by a tropical rain forest soil bacterium, Enterobacter lignolyticus. Proc Natl Acad Sci 109:E2173–E2182
Kurata A, Uchimura K, Shimamura S, Kobayashi T, Horikoshi K (2007) Nucleotide and deduced amino acid sequences of a subtilisin-like serine protease from a deep-sea bacterium, Alkalimonas collagenimarina AC40T. Appl Microbiol Biotechnol 77:311–319
Kurata A, Kitamura Y, Irie S, Takemoto S, Akai Y, Hirota Y, Fujita T, Iwai K, Furusawa M, Kishimoto N (2010a) Enzymatic synthesis of caffeic acid phenethyl ester analogues in ionic liquid. J Biotechnol 148:133–138
Kurata A, Uchimura K, Kobayashi T, Horikoshi K (2010b) Collagenolytic subtilisin-like protease from the deep-sea bacterium Alkalimonas collagenimarina AC40T. Appl Microbiol Biotechnol 86:589–598
Kurata A, Takemoto S, Fujita T, Iwai K, Furusawa M, Kishimoto N (2011) Synthesis of 3-cyclohexylpropyl caffeate from 5-caffeoylquinic acid with consecutive enzymatic conversions in ionic liquid. J Mol Catal B Enzym 69:161–167
Kurata A, Hirose Y, Misawa N, Hurunaka K, Kishimoto N (2014) Draft genome sequence of the ionic liquid-tolerant bacterium Bacillus amyloliquefaciens CMW1. Genome Announc 2(5). doi:10.1128/genomeA.01051-14
Lee S-M, Chang W-J, Choi A-R, Koo Y-M (2005) Influence of ionic liquids on the growth of Escherichia coli. Korean J Chem Eng 22:687–690
Liang C, Xue Y, Fioroni M, Rodríguez-Ropero F, Zhou C, Schwaneberg U, Ma Y (2011) Cloning and characterization of a thermostable and halo-tolerant endoglucanase from Thermoanaerobacter tengcongensis MB4. Appl Microbiol Biotechnol 89:315–326
Nakashima K, Maruyama T, Kamiya N, Goto M (2005) Comb-shaped poly (ethylene glycol)-modified subtilisin Carlsberg is soluble and highly active in ionic liquids. Chem Commun 13(34):4297–4299
Naushad M, ALOthman ZA, Khan AB, Ali M (2012) Effect of ionic liquid on activity, stability, and structure of enzymes: a review. Int J Biol Macromol 51:555–560
Neidhart DJ, Petsko GA (1988) The refined crystal structure of subtilisin Carlsberg at 2.5 Å resolution. Protein Eng 2:271–276
Nielsen H, Engelbrecht J, Brunak S, von Heijne G (1997) Identification of prokaryotic and eukaryotic signal peptides and prediction of their cleavage sites. Protein Eng 10:1–6
Noritomi H, Suzuki K, Kikuta M, Kato S (2009) Catalytic activity of α-chymotrypsin in enzymatic peptide synthesis in ionic liquids. Biochem Eng J 47:27–30
Noritomi H, Minamisawa K, Kamiya R, Kato S (2011) Thermal stability of proteins in the presence of aprotic ionic liquids. J Biomed Sci Eng 4:94
Ogino H, Ishikawa H (2001) Enzymes which are stable in the presence of organic solvents. J Biosci Bioeng 91:109–116
Park S, Kazlauskas RJ (2003) Biocatalysis in ionic liquids–advantages beyond green technology. Curr Opin Biotechnol 14:432–437
Ranke J, Mölter K, Stock F, Bottin-Weber U, Poczobutt J, Hoffmann J, Ondruschka B, Filser J, Jastorff B (2004) Biological effects of imidazolium ionic liquids with varying chain lengths in acute Vibrio fischeri and WST-1 cell viability assays. Ecotoxicol Environ Saf 58:396–404
Siezen RJ, Leunissen JA (1997) Subtilases: the superfamily of subtilisin-like serine proteases. Prot Sci 6:501–523
Singer SW, Reddy AP, Gladden JM, Guo H, Hazen TC, Simmons BA, VanderGheynst JS (2011) Enrichment, isolation and characterization of fungi tolerant to 1-ethyl-3-methylimidazolium acetate. J Appl Microbiol 110:1023–1031
Singh M, Singh RS, Banerjee UC (2009) Stereoselective synthesis of (R)-1-chloro-3 (3, 4-difluorophenoxy)-2-propanol using lipases from Pseudomonas aeruginosa in ionic liquid-containing system. J Mol Catal B Enzym 56:294–299
Toyokawa Y, Takahara H, Reungsang A, Fukuta M, Hachimine Y, Tachibana S, Yasuda M (2010) Purification and characterization of a halotolerant serine proteinase from thermotolerant Bacillus licheniformis RKK-04 isolated from Thai fish sauce. Appl Microbiol Biotechnol 86:1867–1875
Trivedi N, Gupta V, Reddy C, Jha B (2013) Detection of ionic liquid stable cellulase produced by the marine bacterium Pseudoalteromonas sp. isolated from brown alga Sargassum polycystum C. Agardh. Bioresour Technol 132:313–319
Tsujibo H, Miyamoto K, Tanaka K, Kaidzu Y, Imada C, Okami Y, Inamori Y (1996) Cloning and sequence analysis of a protease-encoding gene from the marine bacterium Alteromonas sp. strain O-7. Biosci Biotechnol Biochem 60:1284–1288
Vasantha N, Thompson L, Rhodes C, Banner C, Nagle J, Filpula D (1984) Genes for alkaline protease and neutral protease from Bacillus amyloliquefaciens contain a large open reading frame between the regions coding for signal sequence and mature protein. J Bacteriol 159:811–819
Zhang T, Datta S, Eichler J, Ivanova N, Axen SD, Kerfeld CA, Chen F, Kyrpides N, Hugenholtz P, Cheng J-F (2011) Identification of a haloalkaliphilic and thermostable cellulase with improved ionic liquid tolerance. Green Chem 13:2083–2090
Zhao H, Jackson L, Song Z, Olubajo O (2006) Using ionic liquid [EMIM][CH3COO] as an enzyme-‘friendly’co-solvent for resolution of amino acids. Tetrahedron Asymmetry 17:2491–2498
Acknowledgments
This work was supported by Grant-in-Aid for Young Scientists (B) (No. 26870731) from JSPS (to A.K.) and a Grant for Scientific Research from Faculty of Agriculture, Kindai University (to A.K.).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by L. Huang.
A. Kurata, H. Senoo, Y. Ikeda, H. Kaida, C. Matsuhara and N. Kishimoto contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
792_2016_832_MOESM1_ESM.docx
Supplemental Fig. 1. Effect of NaCl or pH on CMW1 growth. CMW1 was cultivated using the medium (pH 7.3) containing 1 % yeast extract, 0.25 % tryptone, 0.5 % KCl, 1 % MgSO4 7H2O and 0-16 % (w/v) NaCl (A). Using the medium containing 1 % yeast extract, 0.25 % tryptone, 0.5 % KCl, and 1 % MgSO4 7H2O at the pH values with 1.9-10.9, CMW1 was cultivated for 3 h (B). ●, without addition of NaCl; ○, 5.0 %(w/v); ■,10 %(w/v); □,16 %(w/v). Supplemental Fig. 2. Effects of various ionic liquids on CMW1 growth. CMW1 was cultivated in the presence or absence of (A) [EMIM]CF3SO3, (B) [BMIM]Cl, (C) [BMIM]CF3SO3, (D) [BMIM]BF4, (E) [BMIM]PF6, or (F) [BMIM]NTf2 as described in Materials and Methods. ●, without addition of ionic liquid; ■, 2.5 %(v/v); □, 5.0 %(v/v); ○, 10.0 %(v/v); △, 16.0 %(v/v) (DOCX 167 kb)
Rights and permissions
About this article
Cite this article
Kurata, A., Senoo, H., Ikeda, Y. et al. Properties of an ionic liquid-tolerant Bacillus amyloliquefaciens CMW1 and its extracellular protease. Extremophiles 20, 415–424 (2016). https://doi.org/10.1007/s00792-016-0832-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-016-0832-z