Abstract
Objectives
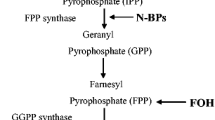
Bisphosphonates-related osteonecrosis of the jaw (BRONJ) is a common problem in patients undergoing long-term administration of highly potent nitrogen-containing bisphosphonates (N-BPs). This pathology occurs via bone and soft tissue mechanism. Zoledronic acid (ZA) is the most potent intravenous N-BP used to prevent bone loss in patients with bone dysfunction. The objective of this in vitro study was to evaluate the role of different ZA concentrations on the cells from human oral cavity, as well as the potential of plasma rich in growth factors (PRGF) to overcome the negative effects of this BP.
Material and methods
Primary human gingival fibroblasts and primary human alveolar osteoblasts were used. Cell proliferation was evaluated by means of a fluorescence-based method. A colorimetric assay to detect DNA fragmentation undergoing apoptosis was used to determine cell death, and the expression of both NF-κB and pNF-κB were quantified by Western blot analysis.
Results
ZA had a cytotoxic effect on both human gingival fibroblasts and human alveolar osteoblasts. This BP inhibits cell proliferation, stimulates apoptosis, and induces inflammation. However, the addition of PRGF suppresses all these negative effects of the ZA.
Conclusions
PRGF shows a cytoprotective role against the negative effects of ZA on primary oral cells.
Clinical relevance
At present, there is no definitive treatment for bisphosphonates-related osteonecrosis of the jaw (BRONJ), being mainly palliatives. Our results revealed that PRGF has a cytoprotective role in cells exposed to zoledronic acid, thus providing a reliable adjunctive therapy for the treatment of BRONJ pathology.





Similar content being viewed by others
References
Walter C, Klein MO, Pabst A, Al-Nawas B, Duschner H, Ziebart T (2010) Influence of bisphosphonates on endothelial cells, fibroblasts, and osteogenic cells. Clin Oral Investig 14(1):35–41. doi:10.1007/s00784-009-0266-4
Acil Y, Moller B, Niehoff P, Rachko K, Gassling V, Wiltfang J, Simon MJ (2012) The cytotoxic effects of three different bisphosphonates in-vitro on human gingival fibroblasts, osteoblasts and osteogenic sarcoma cells. J Craniomaxillofac Surg Off Publ Eur Assoc Craniomaxillofac Surg 40(8):e229–e235. doi:10.1016/j.jcms.2011.10.024
Koch FP, Yekta SS, Merkel C, Ziebart T, Smeets R (2010) The impact of bisphosphonates on the osteoblast proliferation and Collagen gene expression in vitro. Head Face Med 6:12. doi:10.1186/1746-160X-6-12
Xu XL, Gou WL, Wang AY, Wang Y, Guo QY, Lu Q, Lu SB, Peng J (2013) Basic research and clinical applications of bisphosphonates in bone disease: what have we learned over the last 40 years? J Transl Med 11:303. doi:10.1186/1479-5876-11-303
Cartsos VM, Zhu S, Zavras AI (2008) Bisphosphonate use and the risk of adverse jaw outcomes: a medical claims study of 714,217 people. J Am Dent Assoc 139(1):23–30
Tanaka Y, Nagai Y, Dohdoh M, Oizumi T, Ohki A, Kuroishi T, Sugawara S, Endo Y (2013) In vitro cytotoxicity of zoledronate (nitrogen-containing bisphosphonate: NBP) and/or etidronate (non-NBP) in tumour cells and periodontal cells. Arch Oral Biol 58(6):628–637. doi:10.1016/j.archoralbio.2012.11.010
Hinchy NV, Jayaprakash V, Rossitto RA, Anders PL, Korff KC, Canallatos P, Sullivan MA (2013) Osteonecrosis of the jaw—prevention and treatment strategies for oral health professionals. Oral Oncol 49(9):878–886. doi:10.1016/j.oraloncology.2013.06.008
Marx RE (2003) Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg Off J Am Assoc Oral Maxillofac Surg 61(9):1115–1117
Agis H, Blei J, Watzek G, Gruber R (2010) Is zoledronate toxic to human periodontal fibroblasts? J Dent Res 89(1):40–45. doi:10.1177/0022034509354298
Ravosa MJ, Ning J, Liu Y, Stack MS (2011) Bisphosphonate effects on the behaviour of oral epithelial cells and oral fibroblasts. Arch Oral Biol 56(5):491–498. doi:10.1016/j.archoralbio.2010.11.003
Anitua E, Begona L, Orive G (2013) Treatment of hemimandibular paresthesia in a patient with bisphosphonate-related osteonecrosis of the jaw (BRONJ) by combining surgical resection and PRGF-Endoret. Br J Oral Maxillofac Surg 51(8):e272–e274. doi:10.1016/j.bjoms.2012.08.018
Mozzati M, Gallesio G, Arata V, Pol R, Scoletta M (2012) Platelet-rich therapies in the treatment of intravenous bisphosphonate-related osteonecrosis of the jaw: a report of 32 cases. Oral Oncol 48(5):469–474. doi:10.1016/j.oraloncology.2011.12.004
Scoletta M, Arata V, Arduino PG, Lerda E, Chiecchio A, Gallesio G, Scully C, Mozzati M (2013) Tooth extractions in intravenous bisphosphonate-treated patients: a refined protocol. J Oral Maxillofac Surg Off J Am Assoc Oral Maxillofac Surg 71(6):994–999. doi:10.1016/j.joms.2013.01.006
Raja S, Byakod G, Pudakalkatti P (2009) Growth factors in periodontal regeneration. Int J Dent Hyg 7(2):82–89. doi:10.1111/j.1601-5037.2009.00380.x
Chen FM, An Y, Zhang R, Zhang M (2011) New insights into and novel applications of release technology for periodontal reconstructive therapies. J Control Release Off J Control Release Soc 149(2):92–110. doi:10.1016/j.jconrel.2010.10.021
Anitua E, Orive G (2012) Endogenous regenerative technology using plasma- and platelet-derived growth factors. J Control Release Off J Control Release Soc 157(3):317–320. doi:10.1016/j.jconrel.2011.11.011
Albanese A, Licata ME, Polizzi B, Campisi G (2013) Platelet-rich plasma (PRP) in dental and oral surgery: from the wound healing to bone regeneration. Immun Ageing 10(1):23. doi:10.1186/1742-4933-10-23
Anitua E, Sanchez M, Prado R, Orive G (2011) Plasma rich in growth factors: the pioneering autologous technology for tissue regeneration. J Biomed Mater Res A 97(4):536. doi:10.1002/jbm.a.33053
Anitua E, Sanchez M, Orive G, Andia I (2007) The potential impact of the preparation rich in growth factors (PRGF) in different medical fields. Biomaterials 28(31):4551–4560. doi:10.1016/j.biomaterials.2007.06.037
Anitua E, Troya M, Orive G (2012) Plasma rich in growth factors promote gingival tissue regeneration by stimulating fibroblast proliferation and migration and by blocking transforming growth factor-beta1-induced myodifferentiation. J Periodontol 83(8):1028–1037. doi:10.1902/jop.2011.110505
Anitua E, Tejero R, Zalduendo MM, Orive G (2013) Plasma rich in growth factors promotes bone tissue regeneration by stimulating proliferation, migration, and autocrine secretion in primary human osteoblasts. J Periodontol 84(8):1180–1190. doi:10.1902/jop.2012.120292
Colella AD, Chegenii N, Tea MN, Gibbins IL, Williams KA, Chataway TK (2012) Comparison of Stain-Free gels with traditional immunoblot loading control methodology. Anal Biochem 430(2):108–110. doi:10.1016/j.ab.2012.08.015
Scheper M, Chaisuparat R, Cullen K, Meiller T (2010) A novel soft-tissue in vitro model for bisphosphonate-associated osteonecrosis. Fibrogenesis Tissue Repair 3:6. doi:10.1186/1755-1536-3-6
Orriss IR, Key ML, Colston KW, Arnett TR (2009) Inhibition of osteoblast function in vitro by aminobisphosphonates. J Cell Biochem 106(1):109–118. doi:10.1002/jcb.21983
Tipton DA, Seshul BA, Dabbous M (2011) Effect of bisphosphonates on human gingival fibroblast production of mediators of osteoclastogenesis: RANKL, osteoprotegerin and interleukin-6. J Periodontal Res 46(1):39–47. doi:10.1111/j.1600-0765.2010.01306.x
Anitua E, Alkhraisat MH, Orive G (2012) Perspectives and challenges in regenerative medicine using plasma rich in growth factors. J Control Release Off J Control Release Soc 157(1):29–38. doi:10.1016/j.jconrel.2011.07.004
Anitua E, Pascual C, Perez-Gonzalez R, Antequera D, Padilla S, Orive G, Carro E (2013) Intranasal delivery of plasma and platelet growth factors using PRGF-Endoret system enhances neurogenesis in a mouse model of Alzheimer's disease. PLoS One 8(9):e73118. doi:10.1371/journal.pone.0073118
Anitua E, Pascual C, Antequera D, Bolos M, Padilla S, Orive G, Carro E (2014) Plasma rich in growth factors (PRGF-Endoret) reduces neuropathologic hallmarks and improves cognitive functions in an Alzheimer's disease mouse model. Neurobiol Aging 35(7):1582–1595. doi:10.1016/j.neurobiolaging.2014.01.009
Okada S, Kiyama T, Sato E, Tanaka Y, Oizumi T, Kuroishi T, Takahashi T, Sasaki K, Sugawara S, Endo Y (2013) Inhibition of phosphate transporters ameliorates the inflammatory and necrotic side effects of the nitrogen-containing bisphosphonate zoledronate in mice. Tohoku J Exp Med 231(2):145–158
Lesclous P, Abi Najm S, Carrel JP, Baroukh B, Lombardi T, Willi JP, Rizzoli R, Saffar JL, Samson J (2009) Bisphosphonate-associated osteonecrosis of the jaw: a key role of inflammation? Bone 45(5):843–852. doi:10.1016/j.bone.2009.07.011
Alessandri AL, Sousa LP, Lucas CD, Rossi AG, Pinho V, Teixeira MM (2013) Resolution of inflammation: mechanisms and opportunity for drug development. Pharmacol Ther 139(2):189–212. doi:10.1016/j.pharmthera.2013.04.006
Nathan C, Ding A (2010) Nonresolving inflammation. Cell 140(6):871–882. doi:10.1016/j.cell.2010.02.029
Yamamoto Y, Gaynor RB (2004) IkappaB kinases: key regulators of the NF-kappaB pathway. Trends Biochem Sci 29(2):72–79. doi:10.1016/j.tibs.2003.12.003
Zhang J, Middleton KK, Fu FH, Im HJ, Wang JH (2013) HGF mediates the anti-inflammatory effects of PRP on injured tendons. PLoS One 8(6):e67303. doi:10.1371/journal.pone.0067303
Bendinelli P, Matteucci E, Dogliotti G, Corsi MM, Banfi G, Maroni P, Desiderio MA (2010) Molecular basis of anti-inflammatory action of platelet-rich plasma on human chondrocytes: mechanisms of NF-kappaB inhibition via HGF. J Cell Physiol 225(3):757–766. doi:10.1002/jcp.22274
Montaseri A, Busch F, Mobasheri A, Buhrmann C, Aldinger C, Rad JS, Shakibaei M (2011) IGF-1 and PDGF-bb suppress IL-1beta-induced cartilage degradation through down-regulation of NF-kappaB signaling: involvement of Src/PI-3K/AKT pathway. PLoS One 6(12):e28663. doi:10.1371/journal.pone.0028663
Pabst AM, Kruger M, Ziebart T, Jacobs C, Walter C (2015) Isoprenoid geranylgeraniol: the influence on cell characteristics of endothelial progenitor cells after bisphosphonate therapy in vitro. Clin Oral Investig. doi:10.1007/s00784-014-1394-z
Martins MA, Martins MD, Lascala CA, Curi MM, Migliorati CA, Tenis CA, Marques MM (2012) Association of laser phototherapy with PRP improves healing of bisphosphonate-related osteonecrosis of the jaws in cancer patients: a preliminary study. Oral Oncol 48(1):79–84. doi:10.1016/j.oraloncology.2011.08.010
Rasmusson L, Abtahi J (2014) Bisphosphonate associated osteonecrosis of the jaw: an update on pathophysiology, risk factors, and treatment. Int J Dent 2014:471035. doi:10.1155/2014/471035
Mozzati M, Arata V, Gallesio G (2012) Tooth extraction in patients on zoledronic acid therapy. Oral Oncol 48(9):817–821. doi:10.1016/j.oraloncology.2012.03.009
Mozzati M (2013) A report on a 7-year follow up of the surgical management with PRGF®-ENDORET® of oncologic patients affected by intravenous bisphosphonate related osteonecrosis of the jaw. Sur Curr Res 01(S12). doi:10.4172/2161-1076.s12-011
Compliance with ethical standards
ᅟ
Funding
This study was funded by Pharmascreen Saiotek project (Basque Government).
Conflict of interest
The authors declare the following competing financial interest(s): EA is the Scientific Director of, GO, MZ and MT are scientists at BTI Biotechnology Institute, a dental implant company that investigates in the fields of oral implantology and PRGF-Endoret technology.
Ethical approval
The study was performed following the principles of the Declaration of Helsinki, as revised in 2008 and after approval from the Foundation Eduardo Anitua Institutional Review Board.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anitua, E., Zalduendo, M., Troya, M. et al. PRGF exerts a cytoprotective role in zoledronic acid-treated oral cells. Clin Oral Invest 20, 513–521 (2016). https://doi.org/10.1007/s00784-015-1528-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-015-1528-y