Abstract
Objective
Cdk6 is a key regulator during the G1/S cell cycle transition. Aberrant expression of cdk6 protein has been observed in many cancer types. However, little is known about the expression of cdk6 in head and neck squamous cell carcinoma (HNSCC) and its clinical significance. This study evaluated the expression of cdk6 in HNSCC and analyzed the relationship between cdk6 expression and clinicopathological parameters of HNSCC.
Materials and methods
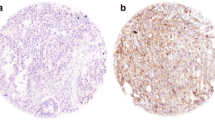
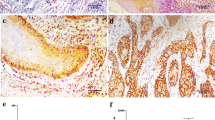
Expression of cdk6 was immunohistochemically investigated in 98 HNSCCs. Nuclear and cytoplasmic positive cells were counted separately. Data were presented as the percentage of positive cells. The correlation between the percentage of positive cells and clinicopathological factors was determined.
Results
Nuclear and cytoplasmic staining for cdk6 were detected in 91 cases and 97 cases, respectively. A significant correlation was found only between the percentage of nuclear positive cells and T classification (p value = 0.0410). Tumors with high nuclear cdk6-positive cells showed a linear trend toward advanced tumor status (p value = 0.0064).
Conclusions
Cdk6 was highly expressed in HNSCC. Tumors with high nuclear cdk6 expression tended to have advanced tumor status. These results suggest that cdk6 plays a vital role in HNSCC and is involved in tumor progression of this cancer.
Clinical relevance
An increased nuclear cdk6 expression is an unfavorable factor for HNSCC. Cdk6 may serve as a therapeutic target in this cancer.



Similar content being viewed by others
References
An HX, Beckmann MW, Reifenberger G, Bender HG, Niederacher D (1999) Gene amplification and overexpression of CDK4 in sporadic breast carcinomas is associated with high tumor cell proliferation. Am J Pathol 154:113–118
Dobashi Y, Goto A, Fukayama M, Abe A, Ooi A (2004) Overexpression of cdk4/cyclin D1, a possible mediator of apoptosis and an indicator of prognosis in human primary lung carcinoma. Int J Cancer 110:532–541
Feng L, Xie Y, Zhang H, Wu Y (2012) miR-107 targets cyclin-dependent kinase 6 expression, induces cell cycle G1 arrest and inhibits invasion in gastric cancer cells. Med Oncol 29:856–863
Mendrzyk F, Radlwimmer B, Joos S et al (2005) Genomic and protein expression profiling identifies CDK6 as novel independent prognostic marker in medulloblastoma. J Clin Oncol 23:8853–8862
Poomsawat S, Buajeeb W, Khovidhunkit SO, Punyasingh J (2010) Alteration in the expression of cdk4 and cdk6 proteins in oral cancer and premalignant lesions. J Oral Pathol Med 39:793–799
Whiteway SL, Harris PS, Venkataraman S et al (2013) Inhibition of cyclin-dependent kinase 6 suppresses cell proliferation and enhances radiation sensitivity in medulloblastoma cells. J Neurooncol 111:113–121
Sherr CJ (1994) G1 phase progression: cycling on cue. Cell 79:551–555
Morgan DO (1995) Principles of CDK regulation. Nature 374:131–134
Baba Y, Watanabe M, Murata A et al (2014) LINE-1 hypomethylation, DNA copy number alterations, and CDK6 amplification in esophageal squamous cell carcinoma. Clin Cancer Res 20:1114–1124
Chen SM, Chen HC, Chen SJ et al (2013) MicroRNA-495 inhibits proliferation of glioblastoma multiforme cells by downregulating cyclin-dependent kinase 6. World J Surg Oncol 11:87
Chilosi M, Doglioni C, Yan Z et al (1998) Differential expression of cyclin-dependent kinase 6 in cortical thymocytes and T-cell lymphoblastic lymphoma/leukemia. Am J Pathol 152:209–217
Wang G, Zheng L, Yu Z et al (2012) Increased cyclin-dependent kinase 6 expression in bladder cancer. Oncol Lett 4:43–46
Landis MW, Pawlyk BS, Li T, Sicinski P, Hinds PW (2006) Cyclin D1-dependent kinase activity in murine development and mammary tumorigenesis. Cancer Cell 9:13–22
Shao Y, Qu Y, Dang S, Yao B, Ji M (2013) MiR-145 inhibits oral squamous cell carcinoma (OSCC) cell growth by targeting c-Myc and Cdk6. Cancer Cell Int 13:51
Wang X, Wu J, Lin Y et al (2014) MicroRNA-320c inhibits tumorous behaviors of bladder cancer by targeting Cyclin-dependent kinase 6. J Exp Clin Cancer Res 33:69
Kollmann K, Heller G, Schneckenleithner C et al (2013) A kinase-independent function of CDK6 links the cell cycle to tumor angiogenesis. Cancer Cell 24:167–181
Patel V, Jakus J, Harris CM, Ensley JF, Robbins KC, Yeudall WA (1997) Altered expression and activity of G1/S cyclins and cyclin-dependent kinases characterize squamous cell carcinomas of the head and neck. Int J Cancer 73:551–555
Piboonniyom SO, Timmermann S, Hinds P, Munger K (2002) Aberrations in the MTS1 tumor suppressor locus in oral squamous cell carcinoma lines preferentially affect the INK4A gene and result in increased cdk6 activity. Oral Oncol 38:179–186
Tsai JW, Li CF, Kao YC et al (2012) Recurrent amplification at 7q21.2 Targets CDK6 gene in primary myxofibrosarcomas and identifies CDK6 overexpression as an independent adverse prognosticator. Ann Surg Oncol 19:2716–2725
Lu X, Fang Y, Wang Z et al (2013) Downregulation of gas5 increases pancreatic cancer cell proliferation by regulating CDK6. Cell Tissue Res 354:891–896
Zhang Z, Huang L, Yu Z et al (2014) Let-7a functions as a tumor suppressor in Ewing’s sarcoma cell lines partly by targeting cyclin-dependent kinase 6. DNA Cell Biol 33:136–147
Saito K, Kawakami K, Matsumoto I, Oda M, Watanabe G, Minamoto T (2010) Long interspersed nuclear element 1 hypomethylation is a marker of poor prognosis in stage IA non-small cell lung cancer. Clin Cancer Res 16:2418–2426
Shigaki H, Baba Y, Watanabe M et al (2013) LINE-1 hypomethylation in gastric cancer, detected by bisulfite pyrosequencing, is associated with poor prognosis. Gastric Cancer 16:480–487
van Hoesel AQ, van de Velde CJ, Kuppen PJ et al (2012) Hypomethylation of LINE-1 in primary tumor has poor prognosis in young breast cancer patients: a retrospective cohort study. Breast Cancer Res Treat 134:1103–1114
Barnes L, Eveson JW, Reichart P, Sidransky D (2005) Word Health Organization classification of tumours of head and neck. IARC, Lyon
Liu F, Korc M (2012) Cdk4/6 inhibition induces epithelial-mesenchymal transition and enhances invasiveness in pancreatic cancer cells. Mol Cancer Ther 11:2138–2148
Pierson J, Hostager B, Fan R, Vibhakar R (2008) Regulation of cyclin dependent kinase 6 by microRNA 124 in medulloblastoma. J Neurooncol 90:1–7
Mahony D, Parry DA, Lees E (1998) Active cdk6 complexes are predominantly nuclear and represent only a minority of the cdk6 in T cells. Oncogene 16:603–611
Nagasawa M, Melamed I, Kupfer A, Gelfand EW, Lucas JJ (1997) Rapid nuclear translocation and increased activity of cyclin-dependent kinase 6 after T cell activation. J Immunol 158:5146–5154
Matushansky I, Radparvar F, Skoultchi AI (2000) Reprogramming leukemic cells to terminal differentiation by inhibiting specific cyclin-dependent kinases in G1. Proc Natl Acad Sci U S A 97:14317–14322
Ericson KK, Krull D, Slomiany P, Grossel MJ (2003) Expression of cyclin-dependent kinase 6, but not cyclin-dependent kinase 4, alters morphology of cultured mouse astrocytes. Mol Cancer Res 1:654–664
Ogasawara T, Kawaguchi H, Jinno S et al (2004) Bone morphogenetic protein 2-induced osteoblast differentiation requires Smad-mediated down-regulation of Cdk6. Mol Cell Biol 24:6560–6568
Leonard JP, LaCasce AS, Smith MR et al (2012) Selective CDK4/6 inhibition with tumor responses by PD0332991 in patients with mantle cell lymphoma. Blood 119:4597–4607
Michaud K, Solomon DA, Oermann E et al (2010) Pharmacologic inhibition of cyclin-dependent kinases 4 and 6 arrests the growth of glioblastoma multiforme intracranial xenografts. Cancer Res 70:3228–3238
Schwartz GK, LoRusso PM, Dickson MA et al (2011) Phase I study of PD 0332991, a cyclin-dependent kinase inhibitor, administered in 3-week cycles (Schedule 2/1). Br J Cancer 104:1862–1868
Sutherland RL, Musgrove EA (2009) CDK inhibitors as potential breast cancer therapeutics: new evidence for enhanced efficacy in ER+ disease. Breast Cancer Res 11:112
Blagden S, de Bono J (2005) Drugging cell cycle kinases in cancer therapy. Curr Drug Targets 6:325–335
Konecny GE, Winterhoff B, Kolarova T et al (2011) Expression of p16 and retinoblastoma determines response to CDK4/6 inhibition in ovarian cancer. Clin Cancer Res 17:1591–1602
Buajeeb W, Poomsawat S, Punyasingh J, Sanguansin S (2009) Expression of p16 in oral cancer and premalignant lesions. J Oral Pathol Med 38:104–108
Kim HS, Chung WB, Hong SH et al (2000) Inactivation of p16INK4a in primary tumors and cell lines of head and neck squamous cell carcinoma. Mol Cells 10:557–565
Mendelsohn AH, Lai CK, Shintaku IP et al (2010) Histopathologic findings of HPV and p16 positive HNSCC. Laryngoscope 120:1788–1794
Muirhead DM, Hoffman HT, Robinson RA (2006) Correlation of clinicopathological features with immunohistochemical expression of cell cycle regulatory proteins p16 and retinoblastoma: distinct association with keratinisation and differentiation in oral cavity squamous cell carcinoma. J Clin Pathol 59:711–715
Acknowledgments
This study was supported by Faculty of Dentistry, Mahidol University Research Grant (2013). The authors would like to thank Mrs. Unruen Meesakul for her technical assistance and staff of the Army Institute of Pathology, Pramongkutklao Hospital and the Department of Oral and Maxillofacial Pathology for their assistance in this study.
Conflict of interest
The authors declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poomsawat, S., Sanguansin, S., Punyasingh, J. et al. Expression of cdk6 in head and neck squamous cell carcinoma. Clin Oral Invest 20, 57–63 (2016). https://doi.org/10.1007/s00784-015-1482-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-015-1482-8