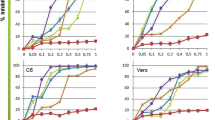
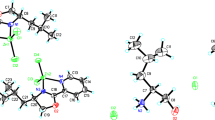
Abstract
While N-heterocyclic carbenes (NHC) are ubiquitous ligands in catalysis for organic or industrial syntheses, their potential to form transition metal complexes for medicinal applications has still to be exploited. Within this frame, we synthesized new homo- and heterobimetallic complexes based on the Au(I)–NHC scaffold. The compounds were synthesized via a microwave-assisted method developed in our laboratories using Au(I)–NHC complexes carrying a pentafluorophenol ester moiety and another Au(I) phosphane complex or a bipyridine ligand bearing a pendant amine function. Thus, we developed two different methods to prepare homo- and heterobimetallic complexes (Au(I)/Au(I) or Au(I)/Cu(II), Au(I)/Ru(II), respectively). All the compounds were fully characterized by several spectroscopic techniques including far infrared, and were tested for their antiproliferative effects in a series of human cancer cells. They showed moderate anticancer properties. Their toxic effects were also studied ex vivo using the precision-cut tissue slices (PCTS) technique and initial results concerning their reactivity with the seleno-enzyme thioredoxin reductase were obtained.







Similar content being viewed by others
Notes
No signal corresponding to disproportionation or exchange phenomena was observed on HRMS spectrum.
References
Muggia F, Farrell N (2005) Crit Rev Oncol Hematol 53:1–2
Barnes KR, Lippard SJ (2004) Met Ions Biol Syst 42:143–177
Hartinger CG, Dyson PJ (2009) Chem Soc Rev 38:391–401
Vessieres A, Top S, Beck W, Hillard E, Jaouen G (2006) Dalton Trans:529–541. doi:10.1039/B509984F
Komeda S, Casini A (2012) Curr Top Med Chem 12:219–235
Peacock AFA, Sadler PJ (2008) Chem Asian J 3:1890–1899
Claffey J, Hogan M, Müller-Bunz H, Pampillón C, Tacke M (2008) Chem Med Chem 3:729–731
Gasser G, Ott I, Metzler-Nolte N (2011) J Med Chem 54:3–25
Tiekink ER (2008) Inflammopharmacology 16:138–142
Nobili S, Mini E, Landini I, Gabbiani C, Casini A, Messori L (2010) Med Res Rev 30:550–580
Ott I (2009) Coord Chem Rev 253:1670–1681
Bertrand B, Casini A (2014) Dalton Trans 43:4209–4219
Maribel N (2009) For a review on anti-parasitic effect of gold compounds. Coord Chem Rev 253:1619–1626
Fiskus W, Saba N, Shen M, Ghias M, Liu J, Gupta SD, Chauhan L, Rao R, Gunewardena S, Schorno K, Austin CP, Maddocks K, Byrd J, Melnick A, Huang P, Wiestner A, Bhalla KN (2014) Cancer Res 74:2520–2532
See for examples of clinical trials in progress in 2014–2015. https://clinicaltrials.gov/ct2/show/NCT01747798, https://clinicaltrials.gov/ct2/show/NCT01419691, http://www.cancer.gov/clinicaltrials/search/view?cdrid=743608&version=HealthProfessional#StudyIdInfo_CD0000743608. Accessed 20 July 2015
Kim IS, Jin JY, Lee IH, Park SJ (2004) Brit J Pharmacol 142:749–755
Mirabelli CK, Johnson RK, Hill DT, Faucette LF, Girard GR, Kuo GY, Sung SM, Crooke ST (1986) J Med Chem 29:218–223
Scheffler H, You Y, Ott I (2010) Polyhedron 29:66–69
Deponte M, Urig S, Arscott LD, Fritz-Wolf K, Réau R, Herold-Mende C, Konkarevic S, Meyer M, Daviout-Charvet E, Ballou DP, Willianms CH, Becker K (2005) J Biol Chem 280:20628–20637
Viry E, Battaglia E, Deborde V, Müller T, Réau R, Davioud-Charvet E, Bagrel D (2008) Chem Med Chem 3:1667–1670
Ott I, Qian X, Xu Y, Vlecken DHW, Marques IJ, Kubutat D, Will J, Sheldrick WS, Jesse P, Prokop A, Bagowski CP (2009) J Med Chem 52:763–770
Bagowski CP, You Y, Scheffler H, Vlecken DH, Schmitz DJ, Ott I (2009) Dalton Trans:10799–10805
Ortego L, Cardoso F, Fillat MF, Laguna A, Meireles M, Villacampa MD, Gimeno MC (2014) J Inorg Biochem 130:32–37
Baker MV, Barnard PJ, Berners-Price SJ, Brayshaw SK, Hickley JL, Skelton BW, White AH (2006) Dalton Trans:3708–3715
Barnard PJ, Baker MV, Berners-Price SJ, Day AD (2004) J Inorg Biochem 98:1642–1647
Hickey JL, Ruhayel RA, Barnard PJ, Baker MV, Berners-Price SJ, Filipovska A (2008) J Am Chem Soc 130:12570–12571
Rubbiani R, Can S, Kitanovic I, Alborzinia H, Stefanopoulou M, Kokoschka M, Mönchgesang S, Sheldrick WS, Wölfl S, Ott I (2011) J Med Chem 54:8646–8657
Berners-Price SJ, Filipovska A (2011) Metallomics 3:863–873
Bindoli A, Rigobello MP, Scutari G, Gabbiani C, Casini A, Messori L (2009) Coord Chem Rev 253:1692–1707
Liu WK, Gust R (2013) Chem Soc Rev 42:755–773
Casini A, Gabbiani C, Sorrentino F, Rigobello MP, Bindoli A, Geldbach TJ, Marrone A, Re N, Hartinger CG, Dyson PJ, Messori L (2008) J Med Chem 51:6773–6781
Arnér ES (2009) J Biochim Biophys Acta 1790:495–526
Holmgren A, Lu J (2010) J Biochem Biophys Res Commun 396:120–124
Rhee SG, Kang SW, Chang TS, Jeong W, Kim K (2001) IUBMB Life 52:35–41
Huber RE, Criddle RS (1967) Arch Biochem Biophys 122:164–173
Citta A, Folda A, Bindoli A, Pigeon P, Top S, Vessières A, Salmain M, Jaouen G, Rigobello MP (2014) J Med Chem 57:8849–8859
Luthman M, Holmgren A (1982) Biochemistry 21:6628–6633
Rigobello MP, Callegaro MT, Barzon E, Benetti M, Bindoli A (1998) Free Radic Biol Med 24:370–376
Urig S, Becker K (2006) Cancer Biol 16:452–465
Liu Y, Li Y, Yu S, Zhao G (2012) Curr Drug Targets 13:1432–1444
Schuh E, Pflüger C, Citta A, Folda A, Rigobello MP, Bindoli A, Casini A, Mohr F (2012) J Med Chem 55:5518–5528
Citta A, Schuh E, Mohr F, Folda A, Massimino M, Bindoli A, Casini A, Rigobello MP (2013) Metallomics 5:1006–1015
Brigelius-Flohé R, Maiorino M (2013) Biochim Biophys Acta 1830:3289–3303
Lease N, Vasilevski V, Carreira M, de Almeida A, Sanaú M, Hirva P, Casini A, Contel M (2013) J. Med. Chem. 56:5806–5818
Pelletier F, Comte V, Massard A, Wenzel M, Toulot S, Richard P, Picquet M, Le Gendre P, Zava O, Edafe F, Casini A, Dyson PJ (2010) J Med Chem 53:6923–6933
Wenzel M, Bertrand B, Eymin M-J, Comte V, Harvey JA, Richard P, Groessl M, Zava O, Amrouche H, Harvey PD, Le Gendre P, Picquet M, Casini A (2011) Inorg Chem 50:9472–9480
Wenzel M, Bigaeva E, Richard P, Le Gendre P, Picquet M, Casini A, Bodio E (2014) J Inorg Biochem 141:10–16
Boseli L, Carraz M, Mazères S, Paloque L, Gonzalès G, Benoit-Vical F, Valentin A, Hemmert C, Gornitza H (2015) Organometallics 34:1046–1053
Lemke J, Metzler-Nolte N (2008) Eur J Inorg Chem 21:3359–3366
Bertrand B, Bodio E, Richard P, Picquet M, Le Gendre P, Casini A (2015) J Organomet Chem 775:124–129
de Graaf IAM, Olinga P, de Jager MH, Merema MT, de Kanter R, van de Kerkhof EG, Groothuis GMM (2010) Nat Protoc 5:1540–1551
Vickers AEM, Rose K, Fisher R, Saulnier M, Sahota P, Bentley P (2004) Toxicol Pathol 32:577–590
Bertrand B, Stefan L, Pirrotta M, Monchaud D, Bodio E, Richard P, Le Gendre P, Warmerdam E, de Jager MH, Groothuis GM, Picquet M, Casini A (2014) Inorg Chem 53:2296–2303
Daum S, Chekhun VF, Todor IN, Lukianova NY, Shvets YV, Sellner L, Putzker K, Lewis J, Zenz T, de Graaf IA, Groothuis GM, Casini A, Zozulia O, Hampel F, Mokhir A (2015) J Med Chem 58:2015–2024
Vergara E, Cerrada E, Clavel C, Casini A, Laguna M (2011) Dalton Trans 40:10927–10935
Bertrand B, de Almeida A, van der Burgt EPM, Picquet M, Citta A, Folda A, Rigobello MP, Le Gendre P, Bodio E, Casini A (2014) Eur J Inorg Chem 27:4532–4536
Gunatilleke SS, Barrios AM (2006) J Med Chem 49:3933–3937
Allen EA, Wilkinson W (1972) Spectrochim Acta Part A 28:2257–2262
Santini C, Pellei M, Gandin V, Porchia M, Tisato F, Marzano C (2013) Chem Rev 114:815–862
Srishailama A, Kumar YP, Reddya PV, Nambigari N, Vuruputuri U, Singhc SS, Satyanarayanaa S (2014) J Photochem Photobiol B Biol 132:111–123
Yi X, Zhao J, Sun J, Guo S, Zhang H (2013) Dalton Trans 42:2062–2074
Habtemariam A, Melchart M, Fernández R, Parsons S, Oswald IDH, Parkin A, Fabbiani FPA, Davidson JE, Dawson A, Aird RE, Jodrell DI, Sadler PJ (2006) J Med Chem 49:6858–6868
Clark RJH, Williams CS (1965) Inorg Chem 4:350–357
Zhanga Y, Zhaoa J, Tanga G, Zhub L (2005) Spectrochim Acta Part A 61:697–706
Oehninger L, Stefanopoulou M, Alborzinia H, Schur J, Ludewig S, Namikawa K, Muñoz-Castro A, Köster RW, Baumann K, Wölfl S, Sheldrick WS, Ott I (2013) Dalton Trans 42:1657–1666
Lu L, Zhu M (2014) Antioxid Redox Signal 20:2210–2224
Jungwirth U, Kowol CR, Keppler BK, Hartinger CG, Berger W, Heffeter P (2011) Antioxid Redox Signal 15:1085–1127
Duncan C, White AR (2012) Metallomics 4:127–138
van der Steen S, de Hoog P, van der Schilden K, Gamez P, Pitié M, Kiss R, Reedijk J (2010) Chem Commun 46:3568–3570
Uson R, Laguna A, Laguna M, Briggs DA, Murray HH, Fackler JJP (1989) Inorg Synth 26:85–91
Dupré N, Brazel C, Fensterbank L, Malacria M, Thorimbert S, Hasenknopf B, Lacôte E (2012) Chem Eur J 18:12962–12965
Busche C, Comba P, Mayboroda A, Wadepohl H (2010) Eur J Inorg Chem 2010:1295–1302
Rigobello MP, Bindoli A (2010) Methods Enzymol 474:109–122
Acknowledgments
The “Centre National de la Recherche Scientifique” (ICMUB, UMR CNRS 6302) is gratefully thanked for financial support. Support was provided by the CNRS, the “Université de Bourgogne” and the “Conseil Régional de Bourgogne” through the 3MIM integrated project (“Marquage de Molécules par les Métaux pour l’Imagerie Médicale”) and PARI SSTIC n°6. Dr. Fanny Picquet, Marie-José Penouilh, and Marcel Soustelle are warmly acknowledged for technical support. E. B. is thankful for COST Action TD1004 financial support. A. C. thanks the University of Groningen for funding (Rosalind Franklin Fellowship). Authors are indebted to EU COST Actions CM1105 and CM1106 for stimulating discussion. M. P. R. acknowledges PRIN 20107Z8XBW granted by the Ministero dell’Istruzione, Università e Ricerca (MIUR) (Italy), CPDA130272 granted by the University of Padova (Italy), and Consorzio Interuniversitario di Ricerca in Chimica dei Metalli nei Sistemi Biologici (CIRCSMB).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bertrand, B., Citta, A., Franken, I.L. et al. Gold(I) NHC-based homo- and heterobimetallic complexes: synthesis, characterization and evaluation as potential anticancer agents. J Biol Inorg Chem 20, 1005–1020 (2015). https://doi.org/10.1007/s00775-015-1283-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-015-1283-1