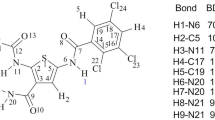
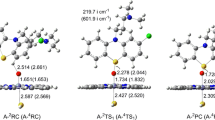
Abstract
4-Alkyl-substituted 1,4-dihydropyridines (DHP) exhibit inhibitory activity toward certain cytochrome P450 enzymes (P450) during their biotransformation by these enzymes, which is called mechanism-based inactivation. Though much experimental evidence had proved the essentiality of alkyl radical for P450 inactivation, the underlying mechanism of such radical formation remains elusive. In the present study, density functional calculations were employed to investigate the dealkylation mechanism of 4-alkyl-substituted DHPs mediated by P450. Interestingly, our results indicate that the initial N–H activation proceeds via a proton-coupled electron transfer process, not via the long presumed hydrogen atom transfer mechanism or the stepwise electron transfer/proton transfer one, to form the amino radical and Cpd II complex. Subsequently, homolytic C–C bond cleavage at the 4-position occurs to afford the product complex involving an alkyl radical, an aromatic pyridine derivative. This C–C cleavage step is rate determining for the whole metabolic reaction, with an energy barrier of 7.9/7.9 kcal/mol on the quartet/doublet state, to which aromatization contributes as an essential intrinsic driving force. The 4-substituent groups induce differences in activation energy barriers and in the transition state structures of hydrogen abstraction process. The substrate reactivity correlates well with the stability of the generated alkyl radical as well as the C–C bond dissociation energy. Understanding the metabolic mechanism of DHP analogs is indeed essential for the related design of safer and more efficient drugs. Furthermore, our findings also enrich the mechanistic picture of amine oxidation catalyzed by P450.





Similar content being viewed by others
References
Swarnalatha G, Prasanthi G, Sirisha N, Madhusudhana Chetty C (2011) Int J ChemTech Res 3:75–89
Baranda AB, Alonso RM, Jiménez RM, Weinmann W (2006) Forensic Sci Int 156:23–34
Kumar A, Maurya RA, Sharma S (2009) Bioorg Med Chem Lett 19:4432–4436
Böcker RH, Guengerich FP (1986) J Med Chem 29:1596–1603
Kawase M, Shah A, Gaveriya H, Motohashi N, Sakagami H, Varga A, Molnar J (2002) Bioorg Med Chem 10:1051–1055
Triggle DJ, Langs DA, Janis RA (1989) Med Res Rev 9:123–180
Poondra RR, Nallamelli RV, Meda CLT, Srinivas BNV, Grover A, Muttabathula J, Voleti SR, Sridhar B, Pal M, Parsa KVL (2013) Bioorg Med Chem Lett 23:1104–1109
Liu Q, Li J, Shen XX, Xing RG, Yang J, Liu Z, Zhou B (2009) Tetrahedron Lett 50:1026–1028
Itoh T, Nagata K, Miyazaki M, Ishikawa H, Kurihara A, Ohsawa A (2004) Tetrahedron 60:6649–6655
Ortiz de Montellano PR, Beilan HS, Kunze KL (1981) J Biol Chem 256:6708–6713
Ortiz de Montellano PR, Beilan HS, Kunze KL (1981) Proc Natl Acad Sci USA 78:1490–1494
Augusto O, Beilan HS, Ortiz de Montellano PR (1982) J Biol Chem 257:1288–1295
Taxak N, Desai PV, Patel B, Mohutsky M, Klimkowski VJ, Gombar V, Bharatam PV (2012) J Comput Chem 33:1740–1747
Hirao H, Chuanprasit P, Cheong YY, Wang XQ (2013) Chemistry 19:7361–7369
Ortiz de Montellano PR, De Voss JJ (2002) Nat Prod Rep 19:477–493
Kobus SM, Wong SGW, Marks GS (2001) Can J Physiol Pharm 79:814–821
Lavigne JA, Nakatsu K, Marks GS (2002) Drug Metab Dispos 30:788–794
Wong SGW, Marks GS (1999) J Pharmacol Toxicol 42:107–113
Gamble JT, Nakatsu KJ, Marks GS (2003) Drug Metab Dispos 31:202–205
Lee JS, Jacobsen NE, Ortiz de Montellano PR (1988) Biochemistry 27:7703–7710
Abdel-monem MM (1975) J Med Chem 18:427–430
Miwa GT, Garland WA, Hodshon BJ, Lu AYH, Northrop DB (1980) J Biol Chem 255:6049–6054
Miwa GT, Walsh JS, Kedderis GL, Hollenberg PF (1983) J Biol Chem 258:14445–14449
Hollenberg PF, Miwa GT, Walsh JS, Dwyer LA, Rickert DE, Kedderis GL (1985) Drug Metab Dispos 13:272–275
Guengerich FP, Yun CH, Macdonald TL (1996) J Biol Chem 271:27321–27329
Shono T, Toda T, Oshino N (1982) J Am Chem Soc 104:2639–2641
Galliani G, Rindone B, Dagnino G, Salmona M (1984) Eur J Drug Metab Pharmokinet 9:289–293
Burka LT, Guengerich FP, Willard RJ, Macdonald TL (1985) J Am Chem Soc 107:2549–2551
Galliani G, Nali M, Rindone B, Tollari S, Rocchetti M, Salmona M (1986) Xenobiotica 16:511–517
Macdonald TL, Gutheim WG, Martin RB, Guengerich FP (1989) Biochemistry 28:2071–2077
Parker VD, Tilset M (1991) J Am Chem Soc 113:8778–8781
Baciocchi E, Lanzalunga O, Lapi A, Manduchi L (1998) J Am Chem Soc 120:5783–5787
Goto Y, Watanabe Y, Fukuzumi S, Jones JP, Dinnocenzo JP (1998) J Am Chem Soc 120:10762–10763
Baciocchi E, Gerini MF, Lanzalunga O, Lapi A, Mancinelli S, Mencarelli P (2000) Chem Commun 393–394
Baciocchi E, Gerini MF, Lanzalunga O, Lapi A, Lo Piparo MG, Mancinelli S (2001) Eur J Org Chem 2:2305–2310
Baciocchi E, Bietti M, Gerini MF, Lanzalunga O (2005) J Org Chem 70:5144–5149
Hanzlik RP, Kishore V, Tullman R (1979) J Med Chem 22:759–761
Hanzlik RP, Tullman R (1982) J Am Chem Soc 104:2048–2050
Macdonald TL, Zirvi K, Burka LT, Peyman P, Guengerich FP (1982) J Am Chem Soc 104:2050–2052
Bondon A, Macdonald TL, Harris TM, Guengerich FP (1989) J Biol Chem 264:1988–1997
Chen H, de Groot MJ, Vermeulen NPE, Hanzlik RP (1997) J Org Chem 62:8227–8230
Dinnocenzo JP, Karki SB, Jones JP (1993) J Am Chem Soc 115:7111–7116
Karki SB, Dinnocenzo JP (1995) Xenobiotica 25:711–724
Karki SB, Dinnocenzo JP, Jones JP, Korzekwa KR (1995) J Am Chem Soc 117:3657–3657
Manchester JI, Dinnocenzo JP, Higgins LA, Jones JP (1997) J Am Chem Soc 119:5069–5070
Jurva U, Bissel P, Isin EM, Igarashi K, Kuttab S, Castagnoli N Jr (2005) J Am Chem Soc 127:12368–12377
Bhakta MN, Wimalasena K (2002) J Am Chem Soc 124:1844–1845
Bhakta MN, Hollenberg PF, Wimalasena K (2005) Chem Commun 265–267
Bhakta MN, Hollenberg PF, Wimalasena K (2005) J Am Chem Soc 127:1376–1377
Bhakta MN, Wimalasena K (2005) Eur J Org Chem 4801–4805
Shaffer CL, Morton MD, Hanzlik RP (2001) J Am Chem Soc 123:8502–8508
Shaffer CL, Morton MD, Hanzlik RP (2001) J Am Chem Soc 123:349–350
Shaffer CL, Harriman S, Koen YM, Hanzlik RP (2002) J Am Chem Soc 124:8268–8274
Cerny MA, Hanzlik RP (2005) Arch Biochem Biophys 436:265–275
Cerny MA, Hanzlik RP (2006) J Am Chem Soc 128:3346–3354
Li CS, Wu W, Kumar D, Shaik S (2006) J Am Chem Soc 128:394–395
Wang Y, Kumar D, Yang CL, Han KL, Shaik S (2007) J Phys Chem B 111:7700–7710
Wang Y, Li DM, Han KL, Shaik S (2010) J Phys Chem B 114:2964–2970
Usharani D, Lacy DC, Borovik AS, Shaik S (2013) J Am Chem Soc 135:17090–17104
Nehru K, Seo MS, Kim J, Nam W (2007) Inorg Chem 46:293–298
Shaik S, Kumar D, de Visser SP, Altun A, Thiel W (2005) Chem Rev 105:2279–2328
Wang Y, Wang HM, Wang YH, Yang CL, Yang L, Han KL (2006) J Phys Chem B 110:6154–6159
Wang Y, Yang CL, Wang HM, Han KL, Shaik S (2007) ChemBioChem 8:277–281
Schyman P, Usharani D, Wang Y, Shaik S (2010) J Phys Chem B 114:7078–7089
Li DM, Wang Y, Han KL (2012) Coord Chem Rev 256:1137–1150
Li XX, Zheng QC, Wang Y, Zhang HX (2014) Dalton Trans 43:14833–14840
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2013) Gaussian 09, revision D, 01st edn. Gaussian, Wallingford
Lee CT, Yang WT, Parr RG (1988) Phys Rev B 37:785–789
Becke AD (1992) J Chem Phys 97:9173–9177
Becke AD (1992) J Chem Phys 96:2155–2160
Becke AD (1993) J Chem Phys 98:5648–5652
Hay PJ, Wadt WR (1985) J Chem Phys 82:299–310
Filatov M, Harris N, Shaik S (1999) Angew Chem Int Ed 38:3510–3512
Harris N, Cohen S, Filatov M, Ogliaro F, Shaik S (2000) Angew Chem Int Ed 39:2003–2007
Ogliaro F, Cohen S, Filatov M, Harris N, Shaik S (2000) Angew Chem Int Ed 39:3851–3855
Ogliaro F, Harris N, Cohen S, Filatov M, de Visser SP, Shaik S (2000) J Am Chem Soc 122:8977–8989
De Visser SP, Ogliaro F, Harris N, Shaik S (2001) J Am Chem Soc 123:3037–3047
De Visser SP, Ogliaro F, Sharma PK, Shaik S (2002) Angew Chem Int Ed 41:1947–1951
Rydberg P, Ryde U, Olsen L (2008) J Chem Theory Comput 4:1369–1377
Kumar D, de Visser SP, Sharma PK, Cohen S, Shaik S (2004) J Am Chem Soc 126:1907–1920
Shaik S, de Visser SP, Kumar D (2004) J Biol Inorg Chem 9:661–668
Hirao H, Kumar D, Thiel W, Shaik S (2005) J Am Chem Soc 127:13007–13018
Kumar D, de Visser SP, Sharma PK, Derat E, Shaik S (2005) J Biol Inorg Chem 10:181–189
Hirao H, Kumar D, Shaik S (2006) J Inorg Biochem 100:2054–2068
Shaik S, Hirao H, Kumar D (2007) Nat Prod Rep 24:533–552
Janardanan D, Usharani D, Chen H, Shaik S (2011) J Phys Chem Lett 2:2610–2617
Usharani D, Janardanan D, Li CS, Shaik S (2013) Acc Chem Res 46:471–482
Hirao H, Thellamurege NM, Chuanprasit P, Xu K (2013) Int J Mol Sci 105:2279–2328
Lai WZ, Li CS, Chen H, Shaik S (2012) Angew Chem Int Ed 51:5556–5578
Mayer JM, Hrovat DA, Thomas JL, Borden WT (2002) J Am Chem Soc 124:11142–11147
Shaik S, Filatov M, Schroder D, Schwarz H (1998) Chemistry 4:193–199
Groves JT, McClusky GA (1976) J Am Chem Soc 98:859–861
Ji L, Schüürmann G (2013) Angew Chem Int Ed 52:744–748
Rydberg P, Olsen L (2011) J Chem Theory Comput 7:3399–3404
Kumar D, Latifi R, Kumar S, Rybak-Akimova EV, Sainna MA, de Visser SP (2013) Inorg Chem 52:7968–7979
Csonka GI, Vydrov OA, Scuseria GE, Ruzsinszky A, Perdew JP (2007) J Chem Phys 126:1–8
Ji L, Schüürmann G (2012) J Phys Chem B 116:903–912
Zheng F, Zhan CG (2011) Future Med Chem 3:9–13
Acknowledgments
This work is supported by Natural Science Foundation of China (Grant no. 21003116, 21173211 and 21233008) and by OSSO State Key Laboratory.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, XX., Zhang, X., Zheng, QC. et al. Bio-activation of 4-alkyl analogs of 1,4-dihydropyridine mediated by cytochrome P450 enzymes. J Biol Inorg Chem 20, 665–673 (2015). https://doi.org/10.1007/s00775-015-1252-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-015-1252-8