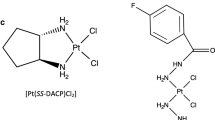
Abstract
In this study, the DNA sequence specificity of four DNA-targeted 9-aminoacridine carboxamide Pt complexes was compared with cisplatin, using two specially constructed plasmid templates. One plasmid contained 5′-CG and 5′-GA insert sequences while the other plasmid contained a G-rich transferrin receptor gene promoter insert sequence. The damage profiles of each compound on the different DNA templates were quantified via a polymerase stop assay with fluorescently labelled primers and capillary electrophoresis. With the plasmid that contained 5′-CG and 5′-GA dinucleotides, the four 9-aminoacridine carboxamide Pt complexes produced distinctly different damage profiles as compared with cisplatin. These 9-aminoacridine complexes had greatly increased levels of DNA damage at CG and GA dinucleotides as compared with cisplatin. It was shown that the presence of a CG or GA dinucleotide was sufficient to reveal the altered DNA sequence selectivity of the 9-aminoacridine carboxamide Pt analogues. The DNA sequence specificity of the Pt complexes was also found to be similarly altered utilising the transferrin receptor DNA sequence.




Similar content being viewed by others
Abbreviations
- CE-LIF:
-
Capillary electrophoresis with laser-induced-fluorescence detection
- Cisplatin:
-
cis-Diamminedichloridoplatinum(II)
- 9AmAcPtCl2 :
-
Dichlorido(N-2-[(2-aminoethyl)amino]-ethyl)-9-aminoacridine-4-carboxamide)platinum(II)
- 7-Methoxy-9AmAcPtCl2 :
-
Dichlorido(N-2-[(2-aminoethyl)amino]-ethyl)-7-methoxy-9-aminoacridine-4-carboxamide)platinum(II)
- 7-Fluoro-9AmAcPtCl2 :
-
Dichlorido(N-2-[(2-aminoethyl)amino]-ethyl)-7-fluoro-9-aminoacridine-4-carboxamide)platinum(II)
- 9-Ethanolamine-AcPtCl2 :
-
Dichlorido(N-2-[(2-aminoethyl)amino]-ethyl)-9-ethanolamine-acridine-4-carboxamide)platinum(II)
- DMF:
-
Dimethylformamide
- NER:
-
Nucleotide excision repair
- TFRC:
-
Transferrin receptor
References
Kelland L (2007) Nat Rev Cancer 7:573–584
Loehrer PJ, Einhorn LH (1984) Ann Intern Med 100:704–713
Jamieson ER, Lippard SJ (1999) Chem Rev 99:2467–2498
Barry MA, Behnke CA, Eastman A (1990) Biochem Pharmacol 40:2353–2362
Eastman A (1990) Cancer Cells 2:275–280
Fichtinger-Schepman AMJ, van der Veer JL, den Hartog JHJ, Lohman PHM, Reedijk J (1985) Biochemistry 24:707–713
Yang DZ, van Boom S, Reedijk J, van Boom JH, Wang AHJ (1995) Biochemistry 34:12912–12920
Gelasco A, Lippard SJ (1998) Biochemistry 37:9230–9239
Corda Y, Anin MF, Leng M, Job D (1992) Biochemistry 31:1904–1908
Murray V, Whittaker J, McFayden WD (1998) Chem-Biol Interact 110:27–37
Murray V, Motyka H, England PR, Wickham G, Lee HH, Denny WA, McFadyen WD (1992) J Biol Chem 267:18805–18809
Murray V, Motyka H, England PR, Wickham G, Lee HH, Denny WA, McFadyen WD (1992) Biochemistry 31:11812–11817
Murray V, Whittaker JK, Temple MD, McFadyen WD (1997) Biochem Biophys Acta 1354:261–271
Murray V, Whittaker JK, McFadyen WD (1998) Chem Biol Interact 110:27–37
Paul M, Murray V (2011) J Biol Inorg Chem 16:735–743
Galea AM, Murray V (2008) Can Informat 6:315–355
Allardyce CS, Dyson PJ, Coffey J, Johnson N (2002) Rapid Commun Mass Spectrom 16:933–935
Khalaila I, Allardyce CS, Verma CS, Dyson PJ (2005) ChemBioChem 6:1788–1795
Piccart MJ, Lamb H, Vermorken JB (2001) Ann Oncol 12:1195–1203
Siddik ZH (2003) Oncogene 22:7265–7279
Smith DJ, Jaggi M, Zhang W, Galich A, Du C, Sterrett SP, Smith LM, Balaji KC (2006) Urology 67:1341–1347
Galluzzi L, Senovilla L, Vitale I, Michels J, Martins I, Kepp O, Castedo M, Kroemer G (2012) Oncogene 31:1869–1883
Chaney SG, Sancar A (1996) J Natl Cancer Inst 88:1346–1360
Furuta T, Ueda T, Aune G, Sarasin A, Kraemer KH, Pommier Y (2002) Cancer Res 62:4899–4902
Weiss RB, Christian MC (1993) Drugs 46:360–377
Wheate NJ, Walker S, Craig GE, Oun R (2010) Dalt Transact 39:8113–8127
Murray V (2000) Prog Nucl Acid Res Mol Biol 63:367–415
Whittaker JK, McFadyen WD, Wickham G, Wakelin LPG, Murray V (1998) Nucl Acids Res 26:3933–3939
Whittaker JK, McFadyen WD, Baguley BC, Murray V (2001) Anti-Cancer Drug Des 16:81–89
Temple MD, McFadyen WD, Holmes RJ, Denny WA, Murray V (2000) Biochemistry 39:5593–5599
Temple MD, Recabarren P, McFadyen WD, Holmes RJ, Denny WA, Murray V (2002) Biochim Biophys Acta 1574:223–230
Murray V, Chen JK, Galea AM (2014) Anti-Cancer Agents Med Chem 14:695–705
Denny WA (1989) Anti-Cancer Drug Des 4:241–263
Carland M, Grannas MJ, Cairns MJ, Roknic VJ, Denny WA, McFadyen WD, Murray V (2010) J Inorg Biochem 104:815–819
Lee HH, Palmer BD, Baguley BC, Chin M, McFadyen WD, Wickham G, Thorsbourne-Palmer D, Wakelin LPG, Denny WA (1992) J Med Chem 35:2983–2987
Holmes RJ, McKeage MJ, Murray V, Denny WA, McFadyen WD (2001) J Inorg Biochem 85:209–217
Murray V (1989) Nucleic Acids Res 17:8889
Murray V, Motyka H, England PR, Wickham G, Lee HH, Denny WA, McFadyen WD (1992) J Biol Chem 267:18805–18809
Murray V, Nguyen TV, Chen JK (2012) Chem Biol Drug Des 80:1–8
Murray V, Kandasamy N (2012) Anti-Cancer Agents Med Chem 12:177–181
Nguyen HTQ, Galea AM, Murray V (2013) Bioorg Med Chem Lett 23:1041–1045
White RJ, Phillips DR (1989) Biochemistry 28:4277–4283
Mattes WB, Hartley JA, Kohn KW (1986) Nucleic Acids Res 14:2971–2987
Nambu A, Kumamoto Y (1995) Jpn J Urol 86:1221–1230
Adams A, Guss JM, Collyer CA, Denny WA, Wakelin LPG (1999) Biochemistry 38:9221–9233
Brabec V (2002) Prog Nucl Acid Res Mol Biol 71:1–68
Budiman ME, Alexander RW, Bierbach U (2004) Biochemistry 43:8560–8567
Guddneppanavar R, Choudhury JR, Kheradi AR, Steen BD, Saluta G, Kucera GL, Day CS, Bierbach U (2007) J Med Chem 50:2259–2263
Chvalova K, Brabec V, Kasparkova J (2007) Nucleic Acids Res 35:1812–1821
Malina J, Natile G, Brabec V (2013) Chem Eur J 19:11984–11991
Acknowledgments
Support of this work by the University of New South Wales, Science Faculty Research Grant Scheme, is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
775_2014_1144_MOESM1_ESM.pdf
Supplementary Figure 1: Electropherograms showing DNA adduct formation caused by cisplatin and the four analogues with the T7.TFRC.G10 plasmid. The relative fluorescence intensity is shown on the y-axis while the DNA fragment size (in nucleotides) is on the x-axis. The peaks indicate the intensity of DNA adduct binding of the Pt compound at the indicated DNA sequence sites on the DNA template. The telomere T7 region is shown T1-T7, left to right. The TFRC insert region includes 5′-GG, 5′-CGGGGG and 5′-GGGGG sequence elements. A) The DMF negative control; B) Treatment with 1.0 μM cisplatin; C) 0.1 μM 9AmAcPtCl2; D) 3.0 μM 7-methoxy-9AmAcPtCl2; E) 0.1 μM 7-fluoro-9AmAcPtCl2 and F) 0.1 μM 9-ethanolamine-9AcPtCl2. (PDF 1272 kb)
Rights and permissions
About this article
Cite this article
Kava, H.W., Galea, A.M., Md. Jamil, F. et al. Characterising the atypical 5′-CG DNA sequence specificity of 9-aminoacridine carboxamide Pt complexes. J Biol Inorg Chem 19, 997–1007 (2014). https://doi.org/10.1007/s00775-014-1144-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-014-1144-3