Abstract
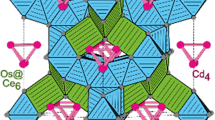
This paper reports on hydrothermal synthesis and crystal structure refinement of dicadmium arsenate hydroxide, Cd2(AsO4)(OH), obtained at 220 °C and autogenous pressure. Its crystal structure is monoclinic, space group P21/a, with a = 13.097(3), b = 14.089(3), c = 10.566(2) Å, β = 108.38(3)°, V = 1850.2(6) Å3 (Z = 16). It is isotypic with the members of the triploidite group of minerals and synthetic compounds, and thus shows a close topological relationship with the triplite group. The complex framework contains edge- and corner-sharing CdO4(OH) and CdO4(OH)2 polyhedra, linked via corner-sharing to AsO4 tetrahedra (average As—O distances range between 1.682 and 1.688 Å). Four five-coordinated Cd sites are at the centers of distorted trigonal bipyramids (average Cd—O distances are between 2.225 and 2.251 Å), whereas the remaining four Cd sites have a distorted octahedral coordination environment (average Cd—O distances are between 2.297 and 2.320 Å). The positions of all the hydrogen atoms were located in a difference-Fourier map and refined with an isotropic displacement parameter. The hydrogen-bonds are weak to very weak. The unusual five-coordination of Cd is briefly discussed in relation to comparable minerals and compounds. Among triploidite-type compounds, Cd2(AsO4)(OH) is the member with the largest unit cell reported so far, and the second known arsenate member.



Similar content being viewed by others
References
Aime S, Digilio G, Gobetto R, Bigi A, Ripamonti A, Roveri N, Gazzano M (1996) Relationship between solid state NMR parameters and X-ray structural data in tricadmium phosphates. Inorg Chem 35:149–154
Antenucci D, Fontan F, Fransolet AM (1989) X-ray powder diffraction data for wolfeite: (Fe0.59Mn0.40Mg0.01)2PO4(OH). Powder Diffract 4:34–35
Belokoneva EL, Sirota MI, Simonov MA, Belov NV (1975) Crystal structure of cadmium germanate, Cd2Ge3O7(OH)2, with new type of band radical (Ge3O7(OH)2). Kristallografiya 20:42–45 (in Russian)
Ben Yahia H, Gaudin E, Feral-Martin C, Darriet J (2010) Structural study of the NaCdVO4-Cd3V2O8 and CdO-V2O5 sections of the ternary system Na2O-CdO-V2O5. J Solid State Chem 183:776–783
Boudjada A, Durif A, Guitel JC (1980) Structure of an acid orthoarsenate of cadmium: CdH10(AsO4)4. Acta Crystallogr B36:133–135 (in French)
Brese NE, O’Keeffe M (1991) Bond-valence parameters for solids. Acta Crystallogr B47:192–197
Chopin C, Armbruster T, Leyx C (2003) Polytypism in wagnerite, Mg2PO4(F,OH). EGS–AGU–EUG Joint Assembly, Abstract book, p. 8323
Coda A, Giuseppetti G, Tadini C (1967) The crystal structure of wagnerite. Atti Accad Naz Lincei Rend Cl Sci Fis Mat Natur 43:212–224
Dal Negro A, Giuseppetti G, Martin Pozas JM (1974) The crystal structure of sarkinite, Mn2AsO4(OH). Tschermaks Mineral Petrogr Mitt 21:246–260
de Pedro I, Rojo JM, Insausti M, Mesa JL, Arriortua MI, Rojo T (2005) Effect of the atmospheric conditions on the thermal behaviour of the sarkinite mineral, Mn2(OH)AsO4. Z Anorg Allg Chem 631:2096–2100
Đorđević T (2008a) BaCo2(AsO4)2. Acta Crystallogr E64:i58–i59
Đorđević T (2008b) Barium zinc diarsenate. Acta Crystallogr E64:i57
Đorđević T (2010) Zn1.86Cd0.14(OH)VO4. Acta Crystallogr E66:i79
Đorđević T (2011) Ba(ZnAsO4)2·H2O, a non-centrosymmetric framework structure related to feldspar. Eur J Mineral 23:437–447
Đorđević T, Karanović LJ (2008) Synthesis, crystal structure, infrared and Raman spectra of Sr4Cu3(AsO4)2(AsO3OH)4·3H2O and Ba2Cu4(AsO4)2(AsO3OH)3. J Solid State Chem 181:2889–2898
Đorđević T, Karanović LJ (2010) A new polymorph of Ba(AsO3OH): synthesis, crystal structure and vibrational spectra. J Solid State Chem 183:2835–2844
Đorđević T, Karanović LJ, Tillmanns E (2008a) Mg13.4(OH)6(HVO4)2(H0.2VO4)6: structural and spectroscopic study. Cryst Res Technol 43:1202–1209
Đorđević T, Stojanović J, Šutović S, Karanović LJ (2008b) Sr-, Ba- and Cd-arsenates with the apatite-type structure. Acta Crystallogr C64:i82–i86
Dowty E (2001) Atoms 6.1. Shape Software, Kingsport, Tennessee 37663, USA; http://www.shapesoftware.com/
El Belghiti AA, Boukhari A, Holt EM (1991) Structures of CdSrP2O7 and CdBaP2O7. Acta Crystallogr C47:473–477
Engel G (1989) The crystal structure of Cd2AsO4F and its relation to a series of oxide silicates and oxide germanates of rare earths. J Less Comm Met 154:367–374 (in German)
Engel G, Klee WE (1970) Crystal structure of cadmium orthoarsenate Cd3(AsO4)2. Z Kristallogr 132:332–339 (in German)
Felsche F (1973) The crystal chemistry of rare-earth silicates. Struct Bond 13:99–197
Fischer RX, Tillmanns E (1988) The equivalent isotropic displacement factor. Acta Crystallogr C44:775–776
Flink G (1924) Sarkinite from Långban, a new mineral from this locality. Geol Fören Stockh Förh 46:661–670
Fontan F (1981) Iron-containing magniotriplite from Alberes massif (eastern Pyrenees, France). A new variety. Bull Minéral 104:672–676 (in French)
Frondel C (1949) Wolfeite, xanthoxenite, and whitlockite from the Palermo mine, New Hampshire. Am Mineral 34:692–705
Hatert F (2007) FeII 2(PO4)(OH), a synthetic analogue of wolfeite. Acta Crystallogr C63:i119–i121
Jin S, Chai G, Liu J, Wang W, Chen X (2009) The centrosymmetric metal metaborate KCdB3O6. Acta Crystallogr C65:i42–i44
Johnson CD, Skakle JMS, Johnston MG, Feldmann J, Macphee DE (2003) Hydrothermal synthesis, crystal structure and aqueous stability of two cadmium arsenate phases, CdNH4(HAsO4)OH and Cd5H2(AsO4)4·4H2O. J Mater Chem 13:1429–1432
Kazanskaya EV, Sandomirskii PA, Simonov MA, Belov NV (1978) Crystal structure of lithium cadmium borate (LiCdBO3). Dokl Akad Nauk SSSR 238:1340–1343 (in Russian)
Keller P, Fontan F, Velasco Roldan F, Melgarejo i Draper JC (1997) Stanĕkite, Fe3+(Mn,Fe2+,Mg)(PO4)O: a new phosphate mineral in pegmatites at Karibib (Namibia) and French Pyrénées (France). Eur J Mineral 9:475–482
Keller P, Lissner F, Schleid T (2006) The crystal structure of stanĕkite, (Fe3+,Mn2+,Fe2+,Mg)2[PO4]O, from Okatjimukuju, Karibib (Namibia), and its relationship to the polymorphs of synthetic Fe2[PO4]O. Eur J Mineral 18:113–118
Keller P, Lissner F, Schleid T (2007) The crystal structure of joosteite, (Mn2+,Mn3+,Fe3+)2(PO4)O, from the Helikon II Mine, Karibib (Namibia) and its relationship to stanekite, (Fe3+,Mn2+,Fe2+,Mg)2(PO4)O. N Jb Mineral Abh 184:225–230
Kolitsch U (2003) Mg-rich wolfeite, (FeII,Mg)2(PO4)(OH): structure refinement and Raman spectroscopic data. Acta Crystallogr E59:i125–i128
Lazić B, Armbruster T, Chopin C, Grew ES, Baronnet A, Palatinus L (2010) Superspace description of modulated wagnerite polytypes. 20th General Meeting of the IMA, Budapest, Hungary, August 21–27, 2010, Acta Mineralogica-Petrographica, Abstract Series, Vol. 6, 739 (Abs.)
Louër D, Labarre J, Auffredic JP, Louër M (1982) Structural study of cadmium hydroxysulfates. IV. Crystal structure of Cd8(OH)12(SO4)2·H2O. Acta Crystallogr B38:1079–1084 (in French)
Masau M, Stanĕk J, Černý P, Chapman R (2000) Metasomatic wolfeite and associated phosphates from the Otov I granitic pegmatite, western Bohemia. J Czech Geol Soc 45:159–173
Mertens B, Müller-Buschbaum H (1997) Cadmium in square pyramids of oxygen in the barium cadmium oxovanadate Ba2Cd3(VO4)2(V2O7). Z Anorg Allg Chem 623:1061–1066 (in German)
Moore PB, Smith JR (1968) Crystal chemistry of the basic manganese arsenates. III. Crystal structure of eveite, Mn2(OH)(AsO4). Am Mineral 53:1841–1845
Otwinowski Z, Borek D, Majewski W, Minor W (2003) Multiparametric scaling of diffraction intensities. Acta Crystallogr A59:228–234
Phanon D, Černý R (2008) Crystal structure of the B-type dierbium oxide ortho-oxosilicate Er2O[SiO4]. Z Anorg Allg Chem 634:1833–1835
Raade G, Rømming C (1986) The crystal structure of β-Mg2(PO4)(OH), a synthetic hydroxyl analogue of wagnerite. Z Kristallogr 177:1–13
Rea JR, Kostiner E (1972) Crystal structure of manganese fluorophosphate, Mn2(PO4)F. Acta Crystallogr B28:2525–2529
Rea JR, Kostiner E (1974) Cadmium fluorophosphate, Cd2(PO4)F. Acta Crystallogr B30:2901–2903
Rea JR, Kostiner E (1976) The crystal structure of copper fluorophosphate, Cu2(PO4)F. Acta Crystallogr B32:1944–1947
Ren L, Grew ES, Xiong M, Ma Z (2003) Wagnerite-Ma5bc, a new polytype of Mg2(PO4)(F,OH), from granulite-facies paragneiss, Larsemann Hills, Prydz Bay, East Antarctica. Can Mineral 41:393–411
Rojo JM, Mesa JL, Pizarro JL, Lezama L, Arriortua MI, Rojo T (1997) Structural and spectroscopic study of the (Mg,Ni)2(OH)(AsO4) arsenates. J Solid State Chem 132:107–112
Sandomirskii PA, Simonov MA, Belov NV (1975) Crystal structure of Zn-triploidite. Dokl Akad Nauk SSSR 220:89–92 (in Russian)
Sheldrick GM (2008) A short history of SHELX. Acta Crystallogr A64:112–122
Simonov MA, Egorov-Tismenko YK, Belov NV (1968) New trisilicate radical, [Si3O10] in the structure of Na4Cd2[Si3O10]. Dokl Akad Nauk SSSR 179:1329–1332 (in Russian)
Simonov MA, Egorov Tismenko YK, Belov NV (1977) Improved crystal structure of Na, Cd-triorthosilicate Na2Cd3(Si3O10). Dokl Akad Nauk SSSR 236:866–868 (in Russian)
Simonov MA, Egorov Tismenko YK, Belov NV (1978) The refinement of the crystal structure of the Na, Cd-triorthosilicate Na4Cd2(Si3O10). Dokl Akad Nauk SSSR 238:348–352 (in Russian)
Sokolova EV, Boronikhin VA, Simonov MA, Belov NV (1979) Crystal structure of a triclinic modification of lithium cadmium borate. Dokl Akad Nauk SSSR 246:1126–1129 (in Russian)
Sokolova EV, Simonov MA, Belov NV (1980) Refined crystal structure of a hexagonal modification of lithium cadmium borate (LiCdBO3). Kristallografiya 25:1285–1286 (in Russian)
Stock N, Stucky GD, Cheetham AK (2002) Synthesis and characterization of the synthetic minerals villyaellenite and sarkinite, Mn5(AsO4)2(HAsO4)2·4(H2O) and Mn2(AsO4)(OH). Z Anorg Allg Chem 628:357–362
Stojanović J, Đorđević T, Karanović LJ (2012) Structural features of two novel alluaudite-like arsenates Cd1.16Zn2.34(AsO4)1.5(HAsO4)(H2AsO4)0.5 and Cd0.74Mg2.76(AsO4)1.5(HAsO4)(H2AsO4)0.5. J Alloys Compd 520:180–189
Taasti KI, Christensen AN, Norby P, Hanson JC, Lebech B, Jakobsen HJ, Skibsted J (2002) Hydrothermal Synthesis, single-crystal structure analysis, and solid-state NMR characterization of Zn2(OH)0.14(3)F0.86(3)(PO4). J Solid State Chem 164:42–50
Tadini C (1981) Magniotriplite: its crystal structure and relation to the triplite-triploidite group. Bull Soc Fr Minéral Cristallogr 104:677–680
Waldrop L (1969) Crystal structure of triplite, (Mn,Fe)2FPO4. Z Kristallogr 130:1–14
Waldrop L (1970) The crystal structure of triploidite and its relation to the structures of other minerals of the triplite-triploidite group. Z Kristallogr 131:1–20
Wei L, Huang Q, Zhou Z, Yin X, Dai G, Liang J (1990) Phase diagram of the LiBO2-CdO system, phase transition, and structure of LiCdBO3. J Solid State Chem 89:16–22
Weil M, Đorđević T, Lengauer CL, Kolitsch U (2009) Investigations in the systems Sr–As–O–X (X = H, Cl): Preparation and crystal structure refinements of the anhydrous arsenates(V) Sr3(AsO4)2, Sr2As2O7, α- and β-SrAs2O6, and of the apatite-type phases Sr5(AsO4)3OH and Sr5(AsO4)3Cl. Solid State Sci 11:2111–2117
Yakubovich OV, Simonov MA, Matvienko EN, Belov NV (1978) The crystal structure of the synthetic finite Fe-term of the series triplite-zwieselite Fe2(PO4)F. Dokl Akad Nauk SSSR 238:576–579 (in Russian)
Yang YW, Stevenson RA, Siegel AM, Downs GW (2011) Redetermination of eveite, Mn2AsO4(OH), based on single-crystal X-ray diffraction data. Acta Crystallogr E67:i68–i68
Yu R, Wang D, Takei T, Koizumi H, Kumada N, Kinomura N (2002) Crystal structure of a novel iron-cobalt phosphate fluoride. J Soc Inorg Mater Jpn 9:99–104
Acknowledgments
Financial support of the Austrian Science Foundation (FWF) (Grant T300-N19) to the first author is gratefully acknowledged. Part of this work was done while the second author was financially supported by the FWF (Grant P17623-N10). We thank Prof. Dr. Manfred Wildner for the editorial handling.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial handling: M. Wildner
Rights and permissions
About this article
Cite this article
Ðorđević, T., Kolitsch, U. Five-coordinate Cd in the crystal structure of triploidite-type Cd2(AsO4)(OH). Miner Petrol 107, 243–251 (2013). https://doi.org/10.1007/s00710-012-0235-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00710-012-0235-5