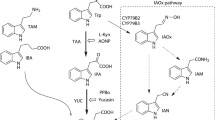
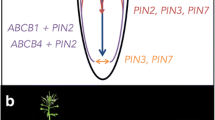
Abstract
Here we present an overview of what is known about endogenous plant compounds that act as inhibitors of hormonal transport processes in plants, about their identity and mechanism of action. We have also summarized commonly and less commonly used compounds of non-plant origin and synthetic drugs that show at least partial ‘specificity’ to transport or transporters of particular phytohormones. Our main attention is focused on the inhibitors of auxin transport. The urgent need to understand precisely the molecular mechanism of action of these inhibitors is highlighted.


Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- ABCB:
-
ATP-binding cassette subfamily B
- ABCG:
-
ATP-binding cassette subfamily G
- ARF:
-
ADP-ribosylation factor
- ARF:
-
Auxin response factor
- ARF-GAP:
-
ARF-GTPase-activating protein
- ARF/GEF:
-
ADP-ribosylation factor/GTPase guanine-nucleotide exchange factor
- AtVPS45:
-
Vacuolar protein sorting 45
- AUX1:
-
Auxin resistant 1
- BEN:
-
BFA-visualized endocytic trafficking defective
- bex :
-
BFA- visualized exocytic trafficking defective
- BFA:
-
Brefeldin A
- CCCP:
-
m-Chlorophenyl hydrazone
- CHPAA:
-
3-Chloro-4-hydroxyphenylacetic acid
- CPD:
-
Cyclopropyl propane dione
- CSI:
-
Chromosaponin I
- CTPP:
-
COOH-terminal propeptide
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- ER:
-
Endoplasmic reticulum
- FCCP:
-
Carbonyl cyanide-4-(trifluoromethoxy)phenyl hydrazone
- GNL2:
-
GNOM-LIKE 2
- HFCA:
-
9-Hydroxyfluorene-9-carboxylic acid
- IAA:
-
Indole-3-acetic acid
- MDR:
-
Multidrug resistance
- MVBs:
-
Multivesicular bodies
- NAA:
-
Naphthalene-1-acetic acid
- 2-NAA:
-
Naphthalene-2-acetic acid
- NBP:
-
NPA-binding protein
- 1-NOA:
-
1-Naphthoxyacetic acid
- 2-NOA:
-
2-Naphthoxyacetic acid
- NPA:
-
1-Naphthylphthalamic acid
- OxIAA:
-
2-Oxindole-3-acetic acid
- PBA:
-
2-(1-Pyrenoyl)-benzoic acid
- PGP:
-
Phosphoglycoprotein
- PI3K:
-
Phosphatidylinositol-3-kinase
- PI4K:
-
Phosphatidylinositol-4-kinase
- PIN:
-
Pin-formed
- PM:
-
Plasma membrane
- PP2A:
-
Phosphatase 2A
- PVC:
-
Prevacuolar compartment
- RabA1b:
-
Ras genes from rat brain A1b
- ROS:
-
Reactive oxygen species
- SCF:
-
Scarface
- SEC8:
-
Secretory 8
- TGN/EE:
-
Trans-Golgi network/early endosome
- TIBA:
-
2,3,5-Triiodobenzoic acid
- TOL:
-
Target of MYB (TOM) 1-like
- trans-CA:
-
Trans-cinnamic acid
- tt4 :
-
Transparent testa4
- TWD1:
-
Twisted-dwarf-1
- VAN:
-
Vascular network defective
- VLCFAs:
-
Very long chain fatty acids
References
Agati G, Azzarello E, Pollastri S, Tattini M (2012) Flavonoids as antioxidants in plants: location and functional significance. Plant Sci 196:67–76. doi:10.1016/j.plantsci.2012.07.014
Aldridge DC, Armstrong JJ, Speake RN, Turner WB (1967) The cytochalasins, a new class of biologically active mould metabolites. Chem Commun 26. doi: 10.1039/c19670000026
Anders N, Jürgens G (2008) Large ARF guanine nucleotide exchange factors in membrane trafficking. Cell Mol Life Sci 65:3433–3445. doi:10.1007/s00018-008-8227-7
Bailly A, Sovero V, Vincenzetti V et al (2008) Modulation of P-glycoproteins by auxin transport inhibitors is mediated by interaction with immunophilins. J Biol Chem 283:21817–21826. doi:10.1074/jbc.M709655200
Banbury DN, Oakley JD, Sessions RB, Banting G (2003) Tyrphostin A23 inhibits internalization of the transferrin receptor by perturbing the interaction between tyrosine motifs and the medium chain subunit of the AP-2 adaptor complex. J Biol Chem 278:12022–12028. doi:10.1074/jbc.M211966200
Benjamins R, Scheres B (2008) Auxin: the looping star in plant development. Annu Rev Plant Biol 59:443–465. doi:10.1146/annurev.arplant.58.032806.103805
Bennett T, Leyser O (2014) The auxin question: a philosophical overview. In: Zažímalová E, Petrášek J, Benková E (eds) Auxin and its role in plant development. Springer Vienna, Vienna, pp 3–19
Bennett MJ, Marchant A, Green HG et al (1996) Arabidopsis AUX1 gene: a permease-like regulator of root gravitropism. Science 273:948–950. doi:10.1126/science.273.5277.948
Bernasconi P, Patel BC, Reagan JD, Subramanian MV (1996) The N-1-naphthylphthalamic acid-binding protein is an integral membrane protein. Plant Physiol 111:427–432. doi:10.1104/pp. 111.2.427
Beyer EM (1972) Auxin transport: a new synthetic inhibitor. Plant Physiol 50:322–327
Beyer EM, Johnson AL, Sweetser PB (1976) A new class of synthetic auxin transport inhibitors. Plant Physiol 57:839–841
Bouchard R, Bailly A, Blakeslee JJ et al (2006) Immunophilin-like TWISTED DWARF1 modulates auxin efflux activities of Arabidopsis P-glycoproteins. J Biol Chem 281:30603–30612. doi:10.1074/jbc.M604604200
Buer CS, Muday GK (2004) The transparent testa4 mutation prevents flavonoid synthesis and alters auxin transport and the response of Arabidopsis roots to gravity and light. Plant Cell 16:1191–1205. doi:10.1105/tpc.020313
Bürkle L, Cedzich A, Döpke C et al (2003) Transport of cytokinins mediated by purine transporters of the PUP family expressed in phloem, hydathodes, and pollen of Arabidopsis. Plant J 34:13–26. doi:10.1046/j.1365-313X.2003.01700.x
Butler JH, Hu S, Brady SR et al (1998) In vitro and in vivo evidence for actin association of the naphthylphthalamic acid-binding protein from zucchini hypocotyls. Plant J 13:291–301. doi:10.1046/j.1365-313X.1998.00017.x
Cox DN (1994) NPA binding activity is peripheral to the plasma membrane and is associated with the cytoskeleton. Plant Cell 6:1941–1953. doi:10.1105/tpc.6.12.1941
Crews P, Manes LV, Boehler M (1986) Jasplakinolide, a cyclodepsipeptide from the marine sponge, Jaspis sp. Tetrahedron Lett 27:2797–2800. doi:10.1016/S0040-4039(00)84645-6
Darwin C (1880) The power of movement in plants. John Murray, London
daSilva LLP, Taylor JP, Hadlington JL et al (2005) Receptor salvage from the prevacuolar compartment is essential for efficient vacuolar protein targeting. Plant Cell 17:132–148. doi:10.1105/tpc.104.026351
Davies PJ (1995) The plant hormone concept: concentration, sensitivity and transport. In: Davies PJ (ed) Plant hormones: physiology, biochemistry and molecular biology. Springer Netherlands, Dordrecht, p 13–38
Davies PJ (ed) (2010) Plant hormones: biosynthesis, signal transduction, action. Springer Netherlands, Dordrecht
Delbarre A, Muller P, Imhoff V, Guern J (1996) Comparison of mechanisms controlling uptake and accumulation of 2,4-dichlorophenoxy acetic acid, naphthalene-1-acetic acid, and indole-3-acetic acid in suspension-cultured tobacco cells. Planta 198:532–541. doi:10.1007/BF00262639
Dhonukshe P, Aniento F, Hwang I et al (2007) Clathrin-mediated constitutive endocytosis of PIN auxin efflux carriers in Arabidopsis. Curr Biol 17:520–527. doi:10.1016/j.cub.2007.01.052
Dhonukshe P, Grigoriev I, Fischer R et al (2008) Auxin transport inhibitors impair vesicle motility and actin cytoskeleton dynamics in diverse eukaryotes. Proc Natl Acad Sci U S A 105:4489–4494. doi:10.1073/pnas.0711414105
Dhonukshe P, Huang F, Galvan-Ampudia CS et al (2010) Plasma membrane-bound AGC3 kinases phosphorylate PIN auxin carriers at TPRXS(N/S) motifs to direct apical PIN recycling. Development 137:3245–3255. doi:10.1242/dev.052456
Dixon RA (2001) Natural products and plant disease resistance. Nature 411:843–847. doi:10.1038/35081178
Dixon MW, Jacobson JA, Cady CT, Muday GK (1996) Cytoplasmic orientation of the naphthylphthalamic acid-binding protein in zucchini plasma membrane vesicles. Plant Physiol 112:421–432. doi:10.1104/pp. 112.1.421
Doyle SM, Robert S (2014) Using a reverse genetics approach to investigate small-molecule activity. In: Hicks GR, Robert S (eds) Plant chemical genomics: methods and protocols. Humana Press, New York, p 51–62
Doyle SM, Haeger A, Vain T et al (2015a) An early secretory pathway mediated by GNOM-LIKE 1 and GNOM is essential for basal polarity establishment in Arabidopsis thaliana. Proc Natl Acad Sci 112:E806–E815. doi:10.1073/pnas.1424856112
Doyle SM, Vain T, Robert S (2015b) Small molecules unravel complex interplay between auxin biology and endomembrane trafficking. J Exp Bot 66:4971–4982. doi:10.1093/jxb/erv179
Drakakaki G, Robert S, Szatmari A-M et al (2011) Clusters of bioactive compounds target dynamic endomembrane networks in vivo. Proc Natl Acad Sci 108:17850–17855. doi:10.1073/pnas.1108581108
Drdová EJ, Synek L, Pečenková T et al (2013) The exocyst complex contributes to PIN auxin efflux carrier recycling and polar auxin transport in Arabidopsis. Plant J 73:709–719. doi:10.1111/tpj.12074
Edgerton MD, Tropsha A, Jones AM (1994) Modelling the auxin-binding site of auxin-binding protein 1 of maize. Phytochemistry 35:1111–1123. doi:10.1016/S0031-9422(00)94807-6
Emans N, Zimmermann S, Fischer R (2002) Uptake of a fluorescent marker in plant cells is sensitive to brefeldin A and wortmannin. Plant Cell 14:71–86
Faulkner IJ, Rubery PH (1992) Flavonoids and flavonoid sulphates as probes of auxin-transport regulation in Cucurbita pepo hypocotyl segments and vesicles. Planta 186:618–625. doi:10.1007/BF00198044
Fendrych M, Synek L, Pečenková T et al (2013) Visualization of the exocyst complex dynamics at the plasma membrane of Arabidopsis thaliana. Mol Biol Cell 24:510–520. doi:10.1091/mbc.E12-06-0492
Feraru E, Feraru MI, Asaoka R et al (2012) BEX5/RabA1b regulates trans-Golgi network-to-plasma membrane protein trafficking in Arabidopsis. Plant Cell 24:3074–3086. doi:10.1105/tpc.112.098152
Friml J (2003) Auxin transport—shaping the plant. Curr Opin Plant Biol 6:7–12. doi:10.1016/S1369526602000031
Friml J, Wiśniewska J, Benková E et al (2002) Lateral relocation of auxin efflux regulator PIN3 mediates tropism in Arabidopsis. Nature 415:806–809. doi:10.1038/415806a
Fujita H, Syono K (1997) PIS1, a negative regulator of the action of auxin transport inhibitors in Arabidopsis thaliana. Plant J 12:583–595
Gälweiler L, Guan C, Müller A et al (1998) Regulation of polar auxin transport by AtPIN1 in Arabidopsis vascular tissue. Science 282:2226–2230. doi:10.1126/science.282.5397.2226
Geisler M, Blakeslee JJ, Bouchard R et al (2005) Cellular efflux of auxin catalyzed by the Arabidopsis MDR/PGP transporter AtPGP1. Plant J 44:179–194. doi:10.1111/j.1365-313X.2005.02519.x
Geldner N, Friml J, Stierhof Y-D et al (2001) Auxin transport inhibitors block PIN1 cycling and vesicle trafficking. Nature 413:425–428. doi:10.1038/35096571
Geldner N, Anders N, Wolters H et al (2003) The Arabidopsis GNOM ARF-GEF mediates endosomal recycling, auxin transport, and auxin-dependent plant growth. Cell 112:219–230. doi:10.1016/S0092-8674(03)00003-5
Gillissen B, Bürkle L, André B et al (2000) A new family of high-affinity transporters for adenine, cytosine, and purine derivatives in Arabidopsis. Plant Cell 12:291–300
Godbolé R, Michalke W, Nick P, Hertel R (2000) Cytoskeletal drugs and gravity-induced lateral auxin transport in rice coleoptiles. Plant Biol 2:176–181. doi:10.1055/s-2000-9154
Goldsmith MHM (1977) The polar transport of auxin. Annu Rev Plant Physiol 28:439–478. doi:10.1146/annurev.pp. 28.060177.002255
Grebe M, Xu J, Möbius W et al (2003) Arabidopsis sterol endocytosis involves actin-mediated trafficking via ARA6-positive early endosomes. Curr Biol 13:1378–1387. doi:10.1016/S0960-9822(03)00538-4
Grunewald W, De Smet I, Lewis DR et al (2012) Transcription factor WRKY23 assists auxin distribution patterns during Arabidopsis root development through local control on flavonol biosynthesis. Proc Natl Acad Sci 109:1554–1559. doi:10.1073/pnas.1121134109
Haagen-Smit AJ, Went FW (1935) A physiological analysis of the growth substances. Proceedings Royal Acad. Amsterdam 38:852–857
Habets MEJ, Offringa R (2014) PIN-driven polar auxin transport in plant developmental plasticity: a key target for environmental and endogenous signals. New Phytol 203:362–377. doi:10.1111/nph.12831
Härri E, Loeffler W, Sigg HP et al (1963) Über die isolierung neuer stoffwechselprodukte aus Penicillium brefeldianum DODGE. Helv Chim Acta 46:1235–1243. doi:10.1002/hlca.19630460419
Hellsberg E, Montanari F, Ecker G (2015) The ABC of phytohormone translocation. Planta Med 81:474–487. doi:10.1055/s-0035-1545880
Hertel R (1983) The mechanism of auxin transport as a model for auxin action. Z Pflanzenphysiol 112:53–67. doi:10.1016/S0044-328X(83)80062-2
Hirose N, Makita N, Yamaya T, Sakakibara H (2005) Functional characterization and expression analysis of a gene, OsENT2, encoding an equilibrative nucleoside transporter in rice suggest a function in cytokinin transport. Plant Physiol 138:196–206. doi:10.1104/pp. 105.060137
Horwitz SB (1994) Taxol (paclitaxel): mechanisms of action. Ann Oncol 5(Suppl 6):S3–S6
Imhoff V, Muller P, Guern J, Delbarre A (2000) Inhibitors of the carrier-mediated influx of auxin in suspension-cultured tobacco cells. Planta 210:580–588. doi:10.1007/s004250050047
Jacobs M, Rubery PH (1988) Naturally occurring auxin transport regulators. Science 241:346–349. doi:10.1126/science.241.4863.346
Jelínková A, Müller K, Fílová-Pařezová M, Petrášek J (2015) NtGNL1a ARF-GEF acts in endocytosis in tobacco cells. BMC Plant Biol. doi:10.1186/s12870-015-0621-3
Kang J, Hwang J-U, Lee M et al (2010) PDR-type ABC transporter mediates cellular uptake of the phytohormone abscisic acid. Proc Natl Acad Sci U S A 107:2355–2360. doi:10.1073/pnas.0909222107
Katekar GF (1976) Inhibitors of the geotropic response in plants: a correlation of molecular structures. Phytochemistry 15:1421–1424. doi:10.1016/S0031-9422(00)88906-2
Katekar GF (1979) Auxins: on the nature of the receptor site and molecular requirements for auxin activity. Phytochemistry 18:223–233. doi:10.1016/0031-9422(79)80059-X
Katekar GF, Geissler AE (1980) Auxin transport inhibitors: IV. Evidence of a common mode of action for a proposed class of auxin transport inhibitors: the phytotropins. Plant Physiol 66:1190–1195. doi:10.1104/pp. 66.6.1190
Kim J-Y, Henrichs S, Bailly A et al (2010) Identification of an ABCB/P-glycoprotein-specific inhibitor of auxin transport by chemical genomics. J Biol Chem 285:23309–23317. doi:10.1074/jbc.M110.105981
Kitakura S, Vanneste S, Robert S et al (2011) Clathrin mediates endocytosis and polar distribution of PIN auxin transporters in Arabidopsis. Plant Cell 23:1920–1931. doi:10.1105/tpc.111.083030
Kleine-Vehn J, Dhonukshe P, Swarup R et al (2006) Subcellular trafficking of the Arabidopsis auxin influx carrier AUX1 uses a novel pathway distinct from PIN1. Plant Cell 18:3171–3181. doi:10.1105/tpc.106.042770
Kleine-Vehn J, Dhonukshe P, Sauer M et al (2008a) ARF GEF-dependent transcytosis and polar delivery of PIN auxin carriers in Arabidopsis. Curr Biol 18:526–531. doi:10.1016/j.cub.2008.03.021
Kleine-Vehn J, Langowski L, Wisniewska J et al (2008b) Cellular and molecular requirements for polar PIN targeting and transcytosis in plants. Mol Plant 1:1056–1066. doi:10.1093/mp/ssn062
Kleine-Vehn J, Leitner J, Zwiewka M et al (2008c) Differential degradation of PIN2 auxin efflux carrier by retromer-dependent vacuolar targeting. Proc Natl Acad Sci U S A 105:17812–17817. doi:10.1073/pnas.0808073105
Kleine-Vehn J, Huang F, Naramoto S et al (2009) PIN auxin efflux carrier polarity is regulated by PINOID kinase-mediated recruitment into GNOM-independent trafficking in Arabidopsis. Plant Cell 21:3839–3849. doi:10.1105/tpc.109.071639
Ko D, Kang J, Kiba T et al (2014) Arabidopsis ABCG14 is essential for the root-to-shoot translocation of cytokinin. Proc Natl Acad Sci U S A 111:7150–7155. doi:10.1073/pnas.1321519111
Korbei B, Moulinier-Anzola J, De-Araujo L et al (2013) Arabidopsis TOL proteins act as gatekeepers for vacuolar sorting of PIN2 plasma membrane protein. Curr Biol 23:2500–2505. doi:10.1016/j.cub.2013.10.036
Kretzschmar T, Kohlen W, Sasse J et al (2012) A petunia ABC protein controls strigolactone-dependent symbiotic signalling and branching. Nature 483:341–344. doi:10.1038/nature10873
Kuromori T, Sugimoto E, Shinozaki K (2011) Arabidopsis mutants of AtABCG22, an ABC transporter gene, increase water transpiration and drought susceptibility. Plant J 67:885–894. doi:10.1111/j.1365-313X.2011.04641.x
Kuromori T, Sugimoto E, Shinozaki K (2014) Intertissue signal transfer of abscisic acid from vascular cells to guard cells. Plant Physiol 164:1587–1592. doi:10.1104/pp. 114.235556
Laňková M, Smith RS, Pešek B et al (2010) Auxin influx inhibitors 1-NOA, 2-NOA, and CHPAA interfere with membrane dynamics in tobacco cells. J Exp Bot 61:3589–3598. doi:10.1093/jxb/erq172
Liu C, Xu Z, Chua NH (1993) Auxin polar transport is essential for the establishment of bilateral symmetry during early plant embryogenesis. Plant Cell 5:621–630. doi:10.1105/tpc.5.6.621
Lomax TL, Muday GK, Rubery PH (1995) Auxin transport. In: Davies PJ (ed) Plant hormones: physiology, biochemistry and molecular biology. Springer Netherlands, Dordrecht, p 509–530
Ludwig-Müller J (2014) Auxin and the interaction between plants and microorganisms. In: Zažímalová E, Petrášek J, Benková E (eds) Auxin and Its Role in Plant Development. Springer Vienna, Vienna, pp 413–434
Marhavý P, Bielach A, Abas L et al (2011) Cytokinin modulates endocytic trafficking of PIN1 auxin efflux carrier to control plant organogenesis. Dev Cell 21:796–804. doi:10.1016/j.devcel.2011.08.014
Matsuoka K (1995) Different sensitivity to wortmannin of two vacuolar sorting signals indicates the presence of distinct sorting machineries in tobacco cells. J Cell Biol 130:1307–1318. doi:10.1083/jcb.130.6.1307
Men S, Boutté Y, Ikeda Y et al (2008) Sterol-dependent endocytosis mediates post-cytokinetic acquisition of PIN2 auxin efflux carrier polarity. Nat Cell Biol 10:237–244. doi:10.1038/ncb1686
Miller R (1984) The use and abuse of filipin to localize cholesterol in membranes. Cell Biol Int Rep 8:519–535. doi:10.1016/0309-1651(84)90050-X
Morejohn LC, Bureau TE, Molè-Bajer J et al (1987) Oryzalin, a dinitroaniline herbicide, binds to plant tubulin and inhibits microtubule polymerization in vitro. Planta 172:252–264. doi:10.1007/BF00394595
Morris DA, Rubery PH, Jarman J, Sabater M (1991) Effects of inhibitors of protein synthesis on transmembrane auxin transport in Cucurbita pepo L. Hypocotyl segments. J Exp Bot 42:773–783. doi:10.1093/jxb/42.6.773
Morris DA, Friml J, Zažímalová E (2010) The transport of auxins. In: Davies PJ (ed) Plant Hormones. Springer Netherlands, Dordrecht, pp 451–484
Muday GK, DeLong A (2001) Polar auxin transport: controlling where and how much. Trends Plant Sci 6:535–542. doi:10.1016/S1360-1385(01)02101-X
Murphy AS, Hoogner KR, Peer WA, Taiz L (2002) Identification, purification, and molecular cloning of N-1-naphthylphthalmic acid-binding plasma membrane-associated aminopeptidases from Arabidopsis. Plant Physiol 128:935–950. doi:10.1104/pp. 010519
Nakanishi S, Kakita S, Takahashi I et al (1992) Wortmannin, a microbial product inhibitor of myosin light chain kinase. J Biol Chem 267:2157–2163
Naramoto S, Nodzyński T, Dainobu T et al (2014a) VAN4 encodes a putative TRS120 that is required for normal cell growth and vein development in Arabidopsis. Plant Cell Physiol 55:750–763. doi:10.1093/pcp/pcu012
Naramoto S, Otegui MS, Kutsuna N et al (2014b) Insights into the localization and function of the membrane trafficking regulator GNOM ARF-GEF at the Golgi apparatus in Arabidopsis. Plant Cell 26:3062–3076. doi:10.1105/tpc.114.125880
Nebenführ A, Ritzenthaler C, Robinson DG (2002) Brefeldin A: deciphering an enigmatic inhibitor of secretion. Plant Physiol 130:1102–1108. doi:10.1104/pp. 011569
Ni DA, Wang LJ, Xu ZH, Xia ZA (1999) Foliar modifications induced by inhibition of polar transport of auxin. Cell Res 9:27–35. doi:10.1038/sj.cr.7290003
Noh B, Murphy AS, Spalding EP (2001) Multidrug resistance-like genes of Arabidopsis required for auxin transport and auxin-mediated development. Plant Cell 13:2441–2454. doi:10.1105/tpc.010350
Okada K, Ueda J, Komaki M et al (1991) Requirement of the auxin polar transport system in early stages of Arabidopsis floral bud formation. Plant Cell 3:677–684. doi:10.1105/tpc.3.7.677
Ortiz-Zapater E, Soriano-Ortega E, Marcote MJ et al (2006) Trafficking of the human transferrin receptor in plant cells: effects of tyrphostin A23 and brefeldin A. Plant J 48:757–770. doi:10.1111/j.1365-313X.2006.02909.x
Osbourn AE (2003) Saponins in cereals. Phytochemistry 62:1–4. doi:10.1016/S0031-9422(02)00393-X
Ovečka M, Berson T, Beck M et al (2010) Structural sterols are involved in both the initiation and tip growth of root hairs in Arabidopsis thaliana. Plant Cell 22:2999–3019. doi:10.1105/tpc.109.069880
Pacifici E, Polverari L, Sabatini S (2015) Plant hormone cross-talk: the pivot of root growth. J Exp Bot 66:1113–1121. doi:10.1093/jxb/eru534
Paciorek T, Zažímalová E, Ruthardt N et al (2005) Auxin inhibits endocytosis and promotes its own efflux from cells. Nature 435:1251–1256. doi:10.1038/nature03633
Parry G, Delbarre A, Marchant A et al (2001) Novel auxin transport inhibitors phenocopy the auxin influx carrier mutation aux1. Plant J 25:399–406. doi:10.1046/j.1365-313x.2001.00970.x
Paudyal R, Jamaluddin A, Warren JP et al (2014) Trafficking modulator TENin1 inhibits endocytosis, causes endomembrane protein accumulation at the pre-vacuolar compartment and impairs gravitropic response in Arabidopsis thaliana. Biochem J 460:177–185. doi:10.1042/BJ20131136
Peer WA (2001) Flavonoid accumulation patterns of transparent testa mutants of Arabidopsis. Plant Physiol 126:536–548. doi:10.1104/pp. 126.2.536
Peer WA, Bandyopadhyay A, Blakeslee JJ et al (2004) Variation in expression and protein localization of the PIN family of auxin efflux facilitator proteins in flavonoid mutants with altered auxin transport in Arabidopsis thaliana. Plant Cell 16:1898–1911. doi:10.1105/tpc.021501
Pěnčík A, Simonovik B, Petersson SV et al (2013) Regulation of auxin homeostasis and gradients in Arabidopsis roots through the formation of the indole-3-acetic acid catabolite 2-oxindole-3-acetic acid. Plant Cell 25:3858–3870. doi:10.1105/tpc.113.114421
Petersson SV, Johansson AI, Kowalczyk M et al (2009) An auxin gradient and maximum in the Arabidopsis root apex shown by high-resolution cell-specific analysis of IAA distribution and synthesis. Plant Cell 21:1659–1668. doi:10.1105/tpc.109.066480
Petrášek J, Friml J (2009) Auxin transport routes in plant development. Development 136:2675–2688. doi:10.1242/dev.030353
Petrášek J, Černá A, Schwarzerová K et al (2003) Do phytotropins inhibit auxin efflux by impairing vesicle traffic? Plant Physiol 131:254–263. doi:10.1104/pp. 012740
Petrášek J, Mravec J, Bouchard R et al (2006) PIN proteins perform a rate-limiting function in cellular auxin efflux. Science 312:914–918. doi:10.1126/science.1123542
Rahman A (2001) Chromosaponin I specifically interacts with AUX1 protein in regulating the gravitropic response of Arabidopsis roots. Plant Physiol 125:990–1000. doi:10.1104/pp. 125.2.990
Reichardt I, Stierhof Y-D, Mayer U et al (2007) Plant cytokinesis requires de novo secretory trafficking but not endocytosis. Curr Biol 17:2047–2053. doi:10.1016/j.cub.2007.10.040
Richter S, Geldner N, Schrader J et al (2007) Functional diversification of closely related ARF-GEFs in protein secretion and recycling. Nature 448:488–492. doi:10.1038/nature05967
Robert S, Chary SN, Drakakaki G et al (2008) Endosidin1 defines a compartment involved in endocytosis of the brassinosteroid receptor BRI1 and the auxin transporters PIN2 and AUX1. Proc Natl Acad Sci U S A 105:8464–8469. doi:10.1073/pnas.0711650105
Robert S, Kleine-Vehn J, Barbez E et al (2010) ABP1 mediates auxin inhibition of clathrin-dependent endocytosis in Arabidopsis. Cell 143:111–121. doi:10.1016/j.cell.2010.09.027
Rocher F, Chollet J-F, Legros S et al (2009) Salicylic acid transport in Ricinus communis involves a pH-dependent carrier system in addition to diffusion. Plant Physiol 150:2081–2091. doi:10.1104/pp. 109.140095
Rojas-Pierce M, Titapiwatanakun B, Sohn EJ et al (2007) Arabidopsis P-glycoprotein19 participates in the inhibition of gravitropism by gravacin. Chem Biol 14:1366–1376. doi:10.1016/j.chembiol.2007.10.014
Ross JJ (1998) Effects of auxin transport inhibitors on gibberellins in pea. J Plant Growth Regul 17:141–146. doi:10.1007/PL00007027
Roudier F, Gissot L, Beaudoin F et al (2010) Very-long-chain fatty acids are involved in polar auxin transport and developmental patterning in Arabidopsis. Plant Cell 22:364–375. doi:10.1105/tpc.109.071209
Rubery PH, Sheldrake AR (1974) Carrier-mediated auxin transport. Planta 118:101–121. doi:10.1007/BF00388387
Santelia D, Henrichs S, Vincenzetti V et al (2008) Flavonoids redirect PIN-mediated polar auxin fluxes during root gravitropic responses. J Biol Chem 283:31218–31226. doi:10.1074/jbc.M710122200
Sasse J, Simon S, Gübeli C et al (2015) Asymmetric localizations of the ABC transporter PaPDR1 trace paths of directional strigolactone transport. Curr Biol 25:647–655. doi:10.1016/j.cub.2015.01.015
Sieburth LE, Muday GK, King EJ et al (2006) SCARFACE encodes an ARF-GAP that is required for normal auxin efflux and vein patterning in Arabidopsis. Plant Cell 18:1396–1411. doi:10.1105/tpc.105.039008
Simon S, Kubeš M, Baster P et al (2013) Defining the selectivity of processes along the auxin response chain: a study using auxin analogues. New Phytol 200:1034–1048. doi:10.1111/nph.12437
Spector I, Shochet N, Kashman Y, Groweiss A (1983) Latrunculins: novel marine toxins that disrupt microfilament organization in cultured cells. Science 219:493–495. doi:10.1126/science.6681676
Steinmann T, Geldner N, Grebe M et al (1999) Coordinated polar localization of auxin efflux carrier PIN1 by GNOM ARF GEF. Science 286:316–318. doi:10.1126/science.286.5438.316
Sun J, Hirose N, Wang X et al (2005) Arabidopsis SOI33/AtENT8 gene encodes a putative equilibrative nucleoside transporter that is involved in cytokinin transport in planta. J Integr Plant Biol 47:588–603. doi:10.1111/j.1744-7909.2005.00104.x
Surpin M, Rojas-Pierce M, Carter C et al (2005) The power of chemical genomics to study the link between endomembrane system components and the gravitropic response. Proc Natl Acad Sci U S A 102:4902–4907. doi:10.1073/pnas.0500222102
Sussman MR, Gardner G (1980) Solubilization of the receptor for N-1-naphthylphthalamic acid. Plant Physiol 66:1074–1078. doi:10.1104/pp. 66.6.1074
Tanaka H, Kitakura S, De Rycke R et al (2009) Fluorescence imaging-based screen identifies ARF GEF component of early endosomal trafficking. Curr Biol 19:391–397. doi:10.1016/j.cub.2009.01.057
Tanaka H, Kitakura S, Rakusová H et al (2013) Cell polarity and patterning by PIN trafficking through early endosomal compartments in Arabidopsis thaliana. PLoS Genet 9, e1003540. doi:10.1371/journal.pgen.1003540
Tanaka H, Nodzyński T, Kitakura S et al (2014) BEX1/ARF1A1C is required for BFA-sensitive recycling of PIN auxin transporters and auxin-mediated development in Arabidopsis. Plant Cell Physiol 55:737–749. doi:10.1093/pcp/pct196
Thimann KV (1977) Hormone action in the whole life of plants. The University of Massachusetts Press, Amherst
Thompson HE, Swanson CP, Norman AG (1946) New growth-regulating compounds. I. Summary of growth-inhibitory activities of some organic compounds as determined by three tests. Bot Gaz 107:476–507
Titapiwatanakun B, Blakeslee JJ, Bandyopadhyay A et al (2009) ABCB19/PGP19 stabilises PIN1 in membrane microdomains in Arabidopsis. Plant J 57:27–44. doi:10.1111/j.1365-313X.2008.03668.x
Tsuda E, Yang H, Nishimura T et al (2011) Alkoxy-auxins are selective inhibitors of auxin transport mediated by PIN, ABCB, and AUX1 transporters. J Biol Chem 286:2354–2364. doi:10.1074/jbc.M110.171165
Tsurumi S, Takagi T, Hashimoto T (1992) A γ-pyronyl-triterpenoid saponin from Pisum sativum. Phytochemistry 31:2435–2438. doi:10.1016/0031-9422(92)83294-9
van der Weij HG (1932) Der mechanismus des wuchsstofftransportes. De Bussy, Amsterdam
van Overbeek J, Blondeau R, Horne V (1951) Trans-cinnamic acid as an anti-auxin. Am J Bot 38:589–595
Vogt T (2010) Phenylpropanoid biosynthesis. Mol Plant 3:2–20. doi:10.1093/mp/ssp106
Wang J, Cai Y, Miao Y et al (2009) Wortmannin induces homotypic fusion of plant prevacuolar compartments. J Exp Bot 60:3075–3083. doi:10.1093/jxb/erp136
Wani MC, Taylor HL, Wall ME et al (1971) Plant antitumor agents. VI. Isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc 93:2325–2327. doi:10.1021/ja00738a045
Went FW (1928) Wuchsstoff und wachstum. Rec Trav Bot Néerl 25:1–116
Yaish P, Gazit A, Gilon C, Levitzki A (1988) Blocking of EGF-dependent cell proliferation by EGF receptor kinase inhibitors. Science 242:933–935. doi:10.1126/science.3263702
Yin R, Han K, Heller W et al (2014) Kaempferol 3-O-rhamnoside-7-O-rhamnoside is an endogenous flavonol inhibitor of polar auxin transport in Arabidopsis shoots. New Phytol 201:466–475. doi:10.1111/nph.12558
Acknowledgments
The authors thank the Ministry of Education, Youth and Sports of the Czech Republic (project COST-CZ LD15088) for support of their work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Handling Editor: David Robinson
Rights and permissions
About this article
Cite this article
Klíma, P., Laňková, M. & Zažímalová, E. Inhibitors of plant hormone transport. Protoplasma 253, 1391–1404 (2016). https://doi.org/10.1007/s00709-015-0897-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-015-0897-z