Abstract
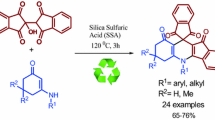
Silica-bonded N-propyltriethylenetetramine was used as a heterogeneous solid base catalyst for the synthesis of spirooxindole pyrimidines by a one-pot condensation reaction of isatin, activated methylene reagent, and barbituric acid or thiobarbituric acid in aqueous medium in good to high yields. Catalyst could be recycled for several times without any additional treatment.
Graphical Abstract




Similar content being viewed by others
References
Cheng D, Ishihara Y, Tan B, Barbas CF (2014) ACS Catal 4:743
Ding K, Lu YP, Coleska N (2006) J Med Chem 49:3432
Galliford CV, Scheidt KA (2007) Angew Chem Int Ed 46:8748
Chai SJ, Lai YF, Xu JC, Zheng H, Zhu Q, Zhang PF (2011) Adv Synth Catal 353:371
Galliford CV, Martenson JA, Stern C, Scheidt KA (2007) Chem Commun 631
Cui CB, Kakeya H, Osada H (1996) Tetrahedron 52:12651
Cui CB, Kakeya H, Osada H (1996) J Antibiot 49:832
Kang TH, Matsumoto K, Murakami Y, Takayama H, Kitajima M, Aimi N, Watanabe H (2002) Eur J Pharmacol 444:39
Jossang A, Jossang P, Hadi HA, Sevenet T, Bodo B (1991) J Org Chem 56:6527
Anderton N, Cockrum PA, Colegate SM, Edgar JA, Flower K, Vit I, Willing RI (1998) Phytochemistry 48:437
Dideberg O, Lamotte-Brasseur J, Dupont L, Campsteyn H, Vermeire M, Angenot L (1977) Acta Crystallogr Sect B 33:1796
Elderfield RC, Gilman RE (1972) Phytochemistry 11:339
Ghedira K, Zeches-Hanrot M, Richard B, Massiot G, Le Men-Oliver L, Sevener T, Goh SH (1988) Phytochemistry 27:3955
Wong WH, Lim PB, Chuah CH (1996) Phytochemistry 41:313
Garnick RL, Le Quesne PW (1978) J Am Chem Soc 100:4213
Sakai S, Aimi N, Yamaguchi K, Ohhira H, Hori K, Haginiwa J (1975) Tetrahedron Lett 16:715
Aimi N, Yamaguchi K, Sakai S, Haginiwa J, Kubo A (1978) Chem Pharm Bull 26:3444
Sakai S, Aimi N, Yamaguchi K, Yamanaka E, Haginiwa J (1982) J Chem Soc Perkin Trans 1:1257
Edmondson S, Danishefsky SJ, Sepp-Lorenzino L, Rosen N (1999) J Am Chem Soc 121:2147
Edmonson SD, Danishefsky SJ (1998) Angew Chem Int Ed 37:1138
Wang HS, Ganesan A (1997) Tetrahedron Lett 38:4327
Wang HS, Ganesan A (2000) J Org Chem 65:4685
Miyake FY, Yakushijin K, Horne DA (2004) Org Lett 6:711
Miyake FY, Yakushijin K, Horne DA (2004) Angew Chem Int Ed 43:5357
Baran PS, Richter JM (2005) J Am Chem Soc 127:15394
Baran PS, Maimone TJ, Richter JM (2007) Nature 446:404
Fuji K, Kawabata T, Ohmori T, Node M (1995) Synlett 367
Fuji K, Node M, Nagasawa H, Naniwa Y, Taga T, Machida K, Snatzke G (1989) J Am Chem Soc 111:7921
Overman LE, Peterson EA (2003) Tetrahedron 59:6905
Matsuura T, Overman LE, Poon DJ (1998) J Am Chem Soc 120:6500
Huang A, Kodanko JJ, Overman LE (2004) J Am Chem Soc 126:14043
Overman LE, Rosen MD (2000) Angew Chem Int Ed 39:4596
Trost BM, Stambuli JP, Silverman SM, Schworer U (2006) J Am Chem Soc 128:13328
Trost BM, Cramer N, Silverman SM (2007) J Am Chem Soc 129:12396
Trost BM, Brennan MK (2009) Synthesis 3003
Meyers C, Carreira EM (2003) Angew Chem 115:718
Marti C, Carreira EM (2005) J Am Chem Soc 127:11505
Wuitschik G, Rogers-Evans M, Buckl A, Bernasconi M, Marki M, Godel T, Fischer H, Wagner B, Parrilla I, Schuler F, Schneider J, Alker A, Schweizer WB, Muller K, Carreira EM (2008) Angew Chem Int Ed 47:4512
Sebahar PR, Williams RM (2000) J Am Chem Soc 122:5666
Onishi T, Sebahar PR, Williams RM (2003) Org Lett 5:3135
Williams RM, Cao J, Tsujishima H, Cox RJ (2003) J Am Chem Soc 125:12172
Chen XH, Wei Q, Luo SW, Xiao H, Gong LZ (2009) J Am Chem Soc 131:13819
Shanthi G, Subbulakshmi G, Perumal PT (2007) Tetrahedron 63:2057
Elinson MN, Ilovaisky AI, Dorofeev AS, Merkulova VM, Stepanov NO, Miloserdov FM, Ogibin YN, Nikishin GI (2007) Tetrahedron 63:10543
Zhu SL, Ji SJ, Zhang Y (2007) Tetrahedron 63:9365
Litvinov YM, Mortikov VY, Shestopalov AM (2008) J Comb Chem 10:741
Gao S, Tsai CH, Tseng C, Yao CF (2008) Tetrahedron 64:9143
Dabiri M, Bahramnejad M, Baghbanzadeh M (2009) Tetrahedron 65:9443
Jadidi K, Ghahremanzadeh R, Bazgir A (2009) J Comb Chem 11:341
Dandia A, Parewa V, Jain AK, Rathore KS (2011) Green Chem 13:2135
Dandia A, Jain AK, Bhati DS (2011) Synth Commun 41:2905
Rad-Moghadam K, Youseftabar-Miri L (2011) Tetrahedron 67:5693
Ziarani GM, Badiei A, Mousavi S, Lashgari N, Shahbazi A (2012) Chin J Catal 33:1832
Safaei HR, Shekouhy M, Shirinfeshan A, Rahmanpur S (2012) Mol Divers 16:669
Zou Y, Hu Y, Liu H, Shi D (2012) ACS Comb Sci 14:38
Karmakar B, Nayak A, Banerji J (2012) Tetrahedron Lett 53:5004
Guo RY, Wang P, Wang GD, Mo LP, Zhang ZH (2013) Tetrahedron 69:2056
Ghahremanzadeh R, Rashid Z, Zarnani AH, Naeimi H (2013) Appl Catal A Gen 467:270
Saluja P, Aggarwal K, Khurana JM (2013) Synth Commun 43:3239
Thakur A, Tripathi M, Rajesh UC, Rawat DS (2013) RSC Adv 3:18142
Satasia SP, Kalaria PN, Avalani JR, Raval DK (2014) Tetrahedron 70:5763
Weitkamp J, Hunger M, Rymsa U (2001) Microporous Mesoporous Mater 48:255
Niknam K, Sadeghi Habibabad M, Deris A, Aeinjamshid N (2013) Monatsh Chem 144:987
Makvandi M, Abiar Dil F, Malekzadeh A, Baghernejad M, Niknam K (2013) Iran J Catal 3:221
Tayebi S, Baghernejad M, Saberi D, Niknam K (2011) Chin J Catal 32:1477
Niknam K, Deris A, Naeimi F, Majleci F (2011) Tetrahedron Lett 52:4642
Nouri Sefat M, Deris A, Niknam K (2011) Chin J Chem 29:2361
Baghernejad M, Niknam K (2012) Int J Chem 4:52
Niknam K, Jamali A, Tajaddod M, Deris A (2012) Chin J Catal 33:1312
Pargaleh Brojeni S, Baghernejad M, Saberi D, Niknam K (2013) Green Chem Lett Rev 6:69
Niknam K, Jamali A (2012) Chin J Catal 33:1840
Niknam K, Borazjani N, Rashidian R, Jamali A (2013) Chin J Catal 34:2245
Niknam K, Rashidian R, Jamali A (2013) Scientia Iran C 20:1863
Acknowledgments
We are thankful to the Persian Gulf University Research Council for the partial support of this work. Also, we are thankful to the School of Chemistry, Manchester University for running NMRs.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Niknam, K., Abolpour, P. Synthesis of spirooxindole pyrimidines catalyzed by silica-bonded N-propyltriethylenetetramine as a recyclable solid base catalyst in aqueous medium. Monatsh Chem 146, 683–690 (2015). https://doi.org/10.1007/s00706-014-1343-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-014-1343-1