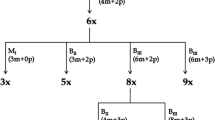
Abstract
Paspalum rufum Nees is a perennial grass that forms a multiploid complex. Natural populations are composed mainly of diploid (2n = 2x = 20) and tetraploid (2n = 4x = 40) cytotypes. The diploid form is sexual and highly self-sterile, while the tetraploid is pseudogamous aposporous apomict and self-fertile. Diploids can also develop aposporous sacs, and some of them complete apomixis. The objective of this work was to analyse apospory expressivity (a gametophytic apomixis component) in diploid hybrids and colchicine-induced autotetraploids of the species. One F1 family was created by crossing two diploid individuals (R6#45 × R5#49) carrying aposporous sacs in 5.8 and 13 % of their ovules, respectively. Moreover, two synthetic autotetraploids were obtained by colchicine treatment of mature seeds from R6#45. The hybrid origin of the F1s was confirmed by segregation analyses of a morphological trait and molecular markers, and the ploidy level of experimental plants was determined by flow cytometry. Apospory expressivity was estimated by embryo sacs observation at anthesis. Out of the 39 hybrids analysed, 38 showed aposporous embryo sacs. Expressivity of the trait ranged from 0 to 36 %, and some individuals differed significantly for both progenitors’ values. Both doubled-diploid plants showed 25 and 32 % of apospory expressivity, which was significantly higher than that observed in R6#45. Results presented in this work revealed a high variability in apospory expressivity of diploid hybrids and suggested that more than one allele are controlling the trait. Moreover, the new induced doubled-diploid plants showed that apospory expressivity is highly ploidy dependent.




Similar content being viewed by others
References
Acuña C, Blount AR, Quesenberry KH, Kenworthy KE, Hanna WW (2011) Tetraploid bahiagrass hybrids: breeding technique, genetic variability and proportion of heterotic hybrids. Euphytica 179:227–235. doi:10.1007/s10681-010-0276-y
Aguilera PM, Sartor ME, Galdeano F, Espinoza F, Quarin CL (2011) Interspecific tetraploid hybrids between two forage grass species: sexual and apomictic. Crop Sci 51:1544–1550. doi:10.2135/cropsci2010.10.0610
Aguilera PM, Galdeano F, Quarin CL, Ortiz JPA, Espinoza F (2015) Inheritance of aposporous apomixis in inter-specific hybrids derived from sexual and apomictic. Crop Sci 55:1947–1956. doi:10.2135/cropsci2014.11.0770
Aliyu OM, Schranz ME, Sharbel TF (2010) Quantitative variation for apomictic reproduction in the genus Boechera (Brassicaceae). Amer J Bot 97:1719–1731. doi:10.3732/ajb.1000188
Asker SE, Jerling L (1992) Apomixis in plants. CRC Press, Boca Raton
Bennett HW, Bashaw EC (1966) Interspecific Hybridization with Paspalum spp. Crop Sci 6:52–54. doi:10.2135/cropsci1966.0011183X000600010016x
Birchler JA (1993) Dosage analysis of maize endosperm development. Annual Rev Genet 27:181–204. doi:10.1146/annurev.ge.27.120193.001145
Burson BL (1991) Genome relationships between tetraploid and hexaploid biotypes of dallisgrass, Paspalum dilatatum. Bot Gaz 152(2):219–223
Carman JG (1997) Asynchronous expression of duplicate genes in angiosperms may cause apomixis, bispory, tetraspory, and polyembryony. Biol J Linn Soc 61:51–94. doi:10.1111/j.1095-8312.1997.tb01778.x
Delgado L, Galdeano F, MaE Sartor, Quarin CL, Espinoza F, Ortiz JP (2014) Analysis of variation for apomictic reproduction in diploid Paspalum rufum. Ann Bot (Oxford) 113:1211–1218. doi:10.1093/aob/mcu056
Espinoza F, Daurelio LD, Pessino SC, Valle EM, Quarin CL (2006) Genetic characterization of Paspalum notatum accessions by AFLP markers. Pl Syst Evol 258:147–159. doi:10.1007/s00606-005-0401-x
Hanna WW, Bashaw EC (1987) Apomixis: its identification and use in plant breeding. Crop Sci 27:1136–1139. doi:10.2135/cropsci1987.0011183X002700060010x
Hojsgaard D, Schegg E, Valls JFM, Martínez EJ, Quarin CL (2008) Sexuality, apomixis, ploidy levels, and genomic relationships among four Paspalum species of the subgenus Anachyris (Poaceae). Flora Morphol Distrib Funct Ecol Plants 203:535–547. doi:10.1016/j.flora.2007.09.005
Martínez EJ, Urbani MH, Quarin CL, Ortiz JP (2001) Inheritance of apospory in bahiagrass, Paspalum notatum. Hereditas 135:19–25. doi:10.1111/j.1601-5223.2001.00019.x
Matzk F, Prodanovic S, Baumlein H, Schubert I (2005) The Inheritance of apomixis in Poa pratensis confirms a five locus model with differences in gene expressivity and penetrance. Pl Cell 17:13–24. doi:10.1105/tpc.104.027359
MeasuringU (2012) MeasuringU. A/B Test Calculator-N-1 Two Proportion test for comparing independent proportions for small and large sample sizes. http://www.measuringu.com/ab-calc.php
Newcombe RG (1998) Two sided confidence intervals for the single proportion: comparison of seven methods. Statist Med 17:857–872. doi:10.1002/(SICI)1097-0258(19980430)17:8<857:AID-SIM777>3.0.CO;2-E
Nogler GA (1984) Gametophytic apomixis. In: Johri BM (ed) Embryology of angiosperms. Springer, Berlin, pp 475–518
Norrmann GA, Quarin CL, Burson BL (1989) Cytogenetics and reproductive behavior of different chromosome races in six Paspalum species. J Heredity 80:24–28
Ortiz JP, Quarin CL, Pessino SC, Acuña C, Martínez EJ, Espinoza F, Hojsgaard DH, Sartor ME, Cáceres ME, Pupilli F (2013) Harnessing apomictic reproduction in grasses: what we have learned from Paspalum. Ann Bot (Oxford) 112:767–787. doi:10.1093/aob/mct152
Podio M, Siena LA, Hojsgaard D, Stein J, Quarin CL, Ortiz JPA (2012) Evaluation of meiotic abnormalities and pollen viability in aposporous and sexual tetraploid Paspalum notatum (Poaceae). Pl Syst Evol 298:1625–1633. doi:10.1007/s00606-012-0664-y
Pupilli F, Barcaccia G (2012) Cloning plants by seeds: inheritance models and candidate genes to increase fundamental knowledge for engineering apomixis in sexual crops. J Biotechnol 154:291–311. doi:10.1016/j.jbiotec.2011.08.028
Pupilli F, Caceres ME, Arcioni S, Quarin CL (1997) Segregation analysis of RFLP markers reveals a tetrasomic inheritance in apomictic Paspalum simplex. Genome 40:822–828. doi:10.1139/g97-806
Pupilli F, Labombarda P, Cáceres ME, Quarn CL, Arcioni S (2001) The chromosome segment related to apomixis in Paspalum simplex is homoeologous to the telomeric region of the long arm of rice chromosome 12. Molec Breed 8:53–61. doi:10.1023/A:1011966922301
Pupilli F, Martínez EJ, Busti A, Calderini O, Quarin CL, Arcioni S (2004) Comparative mapping reveals partial conservation of synteny at the apomixis locus in Paspalum spp. Molec Genet Genomics 270:539–548. doi:10.1007/s00438-003-0949-5
Quarin CL (1986) Seasonal changes in the incidence of apomixis of diploid, triploid, and tetraploid plants of Paspalum cromyorrhizon. Euphytica 35:515–522. doi:10.1007/BF00021860
Quarin CL (1992) The nature of apomixis and its origin in Panicoid grasses. Apomixis Newsl 5:8–15
Quarin CL (1999) Effect of pollen source and pollen ploidy on endosperm formation and seed set in pseudogamous apomictic Paspalum notatum. Sexual Pl Reprod 11:331–335. doi:10.1007/s004970050160
Quarin CL, Norrmann GA (1987) Cytology and reproductive behavior of Paspalum equitans, P. ionanthum, and their hybrids with diploid and tetraploid cytotypes of P. cromyorrhizon. Bot Gaz 148:386–391. doi:10.1086/337667
Quarin CL, Burson BL, Burton GW (1984) Cytology of intra-and interspecific hybrids between two cytotypes of Paspalum notatum and P. cromyorrhizon. Bot Gaz 3:420–426. doi:10.1086/337474
Quarin CL, Norrmann GA, Espinoza F (1998) Evidence for autoploidy in apomictic Paspalum rufum. Hereditas 129:119–124. doi:10.1111/j.1601-5223.1998.00119.x
Quarin CL, Espinoza F, Martinez EJ, Pessino SC, Bovo OA (2001) A rise of ploidy level induces the expression of apomixis in Paspalum notatum. Sexual Pl Reprod 13:243–249. doi:10.1007/s004970100070
Sartor ME, Quarin CL, Espinoza F (2009) Mode of reproduction of colchicine-induced Paspalum plicatulum tetraploids. Crop Sci 49:1270–1276. doi:10.2135/cropsci2008.05.0270
Sartor ME, Quarin CL, Urbani MH, Espinoza F (2011) Ploidy levels and reproductive behaviour in natural populations of five Paspalum species. Pl Syst Evol 293:31–41. doi:10.1007/s00606-011-0416-4
Savidan Y (2000) Apomixis: genetics and breeding. Pl Breed Rev 18:13–86. doi:10.1002/9780470650158.ch2
Schranz ME, Dobes C, Koch MA, Mitchell-Olds T (2005) Sexual reproduction, hybridization, apomixis, and polyploidization in the genus Boechera (Brassicaceae). Amer J Bot 92:1797–1810. doi:10.3732/ajb.92.11.1797
Sharbel TF, Voigt ML, Corral JM, Thiel T, Varshney A, Kumlehn J, Vogel H, Rotter B (2009) Molecular signatures of apomictic and sexual ovules in the Boechera holboellii complex. Pl J 58:870–882. doi:10.1111/j.1365-313X.2009.03826.x
Sharbel TF, Voigt ML, Corral JM, Galla G, Kumlehn J, Klukas C, Schreiber F, Vogel H, Rotter B (2010) Apomictic and sexual ovules of Boechera display heterochronic global gene expression patterns. Pl Cell 22:655–671. doi:10.1105/tpc.109.072223
Siena LA, Sartor ME, Espinoza F, Quarin CL, Ortiz JPA (2008) Genetic and embryological evidences of apomixis at the diploid level in Paspalum rufum support recurrent auto-polyploidization in the species. Sexual Pl Reprod 21:205–215. doi:10.1007/s00497-008-0080-1
Speranza PR (2009) Evolutionary patterns in the Dilatata group (Paspalum, Poaceae). Pl Syst Evol 282:43–56. doi:10.1007/s00606-009-0205-5
Spillane C, Steimer A, Grossniklaus U (2001) Apomixis in agriculture: the quest for clonal seeds. Sexual Pl Reprod 14:179–187. doi:10.1007/s00497-001-0117-1
Stein J, Quarin CL, Martínez EJ, Pessino SC, Ortiz JPA (2004) Tetraploid races of Paspalum notatum show polysomic inheritance and preferential chromosome pairing around the apospory-controlling locus. Theor Appl Genet 109:186–191. doi:10.1007/s00122-004-1614-z
Upton GJ (1992) Fisher’s exact test. J Roy Stat Soc Ser A (Stat Soc) 155:395–402. doi:10.2307/2982890
VassarStats (2016) VassarStats: Website for Statistical computation. Available at: http://vassarstats.net/
Wilson EB (1927) Probable inference, the law of succession, and statistical inference. J Amer Statistical Association 22:209–212. doi:10.1080/01621459.1927.10502953
Young BA, Sherwood RT, Bashaw EC (1979) Cleared-pistil and thick-sectioning techniques for detecting aposporous apomixis in grasses. Canad J Bot 57:1668–1672. doi:10.1139/b79-204
Zilli AL, Brugnoli EA, Marcón F, Billa MB, Rios EF, Martínez EJ, Acuña CA (2015) Heterosis and expressivity of apospory in tetraploid bahiagrass hybrids. Crop Sci 55:1189–1201. doi:10.2135/cropsci2014.10.0685
Zuloaga FO, Morrone O (2005) Revisión de las especies de Paspalum para América del Sur austral (Argentina, Bolivia, sur del Brasil, Chile, Paraguay y Uruguay). Monogr Syst Bot Missouri Bot Gard 102:1–297
Acknowledgments
This work was supported by the Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT), Argentina (PICT 2011–1269 and PICT 2014–1080); Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina [grant no. PIP 11220090100613, PIP 114201101–00283]; Fundación Ciencias Agrarias (PICT-2015). L.D., M.E.S., F.E., M.S., and J.P.A.O. are staff members of CONICET, Argentina.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Information on Electronic Supplementary Material
Online Resource 1. UBC primer screened to detect polymorphism between parental genotypes (R6#45 and R5#49) or segregation in F1 population.
Online Resource 2. Examples of cytometric histograms (in logarithmic scale) of leaves from F1 progenies (R6#45 x R5#49) in the presence of a diploid internal control.
Additional information
Handling editor: Martin Lysak.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Delgado, L., Sartor, M.E., Espinoza, F. et al. Hybridity and autopolyploidy increase the expressivity of apospory in diploid Paspalum rufum . Plant Syst Evol 302, 1471–1481 (2016). https://doi.org/10.1007/s00606-016-1345-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-016-1345-z