Abstract
The authors describe an impedimetric method for the quantitation of the DNA of the human papilloma virus (HPV) type 16. A glassy carbon electrode (GCE) was modified with gold nanosheets and is shown to be superior to a common gold disk electrode. A single-stranded 25mer oligonucleotide (ssDNA) acting as the probe DNA was immobilized via its thiolated 5′ end on both electrodes. After hybridization with target (analyte) DNA, electrochemical impedance spectra were acquired in the presence of hexacyanoferrate as a redox marker. The sensor can distinguish between complementary, non-complementary and single base pair mismatches of HPV ssDNA. At a 1 mM hexacyanoferrate concentration, the biosensors respond to target DNA in the 1 μM to 1 pM concentration range, and the detection limit is 0.15 pM. The results illustrate that the use of gold nanosheets on a GCE distinctly improves the detection and differentiation of HPV compared to using bare gold.

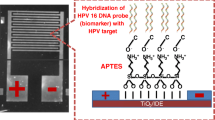
Schematic of exploiting gold nanosheets as a platform for HPV detection. The method works in the 1 μM to 1 pM HPV concentration range and has a 0.15 pM detection limit..





Similar content being viewed by others
References
Berek JS, Novak E (2011) Berek and Novak's gynecology. Wolters Kluwer Health/Lippincott Williams & Wilkins
Hsu IH, Chen W-H, Wu T-K, Sun Y-C (2011) Gold nanoparticle-based inductively coupled plasma mass spectrometry amplification and magnetic separation for the sensitive detection of a virus-specific RNA sequence. J Chromatogr A 1218(14):1795–1801. doi:10.1016/j.chroma.2011.02.005
Sivaraman D, Biswas P, Cella LN, Yates MV, Chen W (2011) Detecting RNA viruses in living mammalian cells by fluorescence microscopy. Trends Biotechnol 29(7):307–313. doi:10.1016/j.tibtech.2011.02.006
Ocaña C, Lukic S, del Valle M (2015) Aptamer-antibody sandwich assay for cytochrome c employing an MWCNT platform and electrochemical impedance. Microchim Acta 182(11–12):2045–2053
Bonanni A, del Valle M (2010) Use of nanomaterials for impedimetric DNA sensors: a review. Anal Chim Acta 678(1):7–17. doi:10.1016/j.aca.2010.08.022
Jia F, Duan N, Wu S, Dai R, Wang Z, Li X (2016) Impedimetric salmonella aptasensor using a glassy carbon electrode modified with an electrodeposited composite consisting of reduced graphene oxide and carbon nanotubes. Microchim Acta 183(1):337–344
Kafka J, Pänke O, Abendroth B, Lisdat F (2008) A label-free DNA sensor based on impedance spectroscopy. Electrochim Acta 53(25):7467–7474. doi:10.1016/j.electacta.2008.01.031
Yao W, Wang L, Wang H, Zhang X, Li L, Zhang N, Pan L, Xing N (2013) An electrochemiluminescent DNA sensor based on nano-gold enhancement and ferrocene quenching. Biosens Bioelectron 40(1):356–361. doi:10.1016/j.bios.2012.08.002
Ebtisam SW, Ravil AS (2006) Biosensors for virus detection. In: Smart biosensor technology. CRC Press, Optical Science and Engineering, pp 567–596. doi:10.1201/9781420019506.ch21
Ito T, Hosokawa K, Maeda M (2007) Detection of single-base mismatch at distal end of DNA duplex by electrochemical impedance spectroscopy. Biosens Bioelectron 22(8):1816–1819. doi:10.1016/j.bios.2006.08.008
Bartolome JP, Echegoyen L, Fragoso A (2015) Reactive carbon Nano-onion modified glassy carbon surfaces as DNA sensors for human papillomavirus oncogene detection with enhanced sensitivity. Anal Chem 87(13):6744–6751. doi:10.1021/acs.analchem.5b00924
Bonanni A, Pividori MI, del Valle M (2010) Impedimetric detection of influenza a (H1N1) DNA sequence using carbon nanotubes platform and gold nanoparticles amplification. Analyst 135(7):1765–1772. doi:10.1039/C000532K
Wang S, Li L, Jin H, Yang T, Bao W, Huang S, Wang J (2013) Electrochemical detection of hepatitis B and papilloma virus DNAs using SWCNT array coated with gold nanoparticles. Biosens Bioelectron 41:205–210. doi:10.1016/j.bios.2012.08.021
Karimizefreh A, Mahyari FA, Vaez Jalali M, Mohammadpour R, Sasanpour P (2014) Human papilloma virus detection using DNA biosensor with electrochemical impedance spectroscopy. Journal of Coupled Systems and Multiscale Dynamics 2(3):164–168
Dreaden EC, Alkilany AM, Huang X, Murphy CJ, El-Sayed MA (2012) The golden age: gold nanoparticles for biomedicine. Chem Soc Rev 41(7):2740–2779. doi:10.1039/c1cs15237h
Lee YJ, Schade NB, Sun L, Fan JA, Bae DR, Mariscal MM, Lee G, Capasso F, Sacanna S, Manoharan VN, Yi GR (2013) Ultrasmooth, highly spherical monocrystalline gold particles for precision plasmonics. ACS Nano 7(12):11064–11070. doi:10.1021/nn404765w
Wu HL, Tsai HR, Hung YT, Lao KU, Liao CW, Chung PJ, Huang JS, Chen IC, Huang MH (2011) A comparative study of gold nanocubes, octahedra, and rhombic dodecahedra as highly sensitive SERS substrates. Inorg Chem 50(17):8106–8111. doi:10.1021/ic200504n
Campos-Ferreira DS, Nascimento GA, Souza EVM, Souto-Maior MA, Arruda MS, Zanforlin DML, Ekert MHF, Bruneska D, Lima-Filho JL (2013) Electrochemical DNA biosensor for human papillomavirus 16 detection in real samples. Anal Chim Acta 804:258–263. doi:10.1016/j.aca.2013.10.038
Hong X, Tan C, Chen J, Xu Z, Zhang H (2015) Synthesis, properties and applications of one- and two-dimensional gold nanostructures. Nano Res 8(1):40–55. doi:10.1007/s12274-014-0636-3
Tohidi M, Mahyari FA, Safavi A (2015) A seed-less method for synthesis of ultra-thin gold nanosheets by using a deep eutectic solvent and gum arabic and their electrocatalytic application. RSC Adv 5(41):32744–32754. doi:10.1039/C4RA17053A
Van Ranst M, Kaplan JB, Burk RD (1992) Phylogenetic classification of human papillomaviruses: correlation with clinical manifestations. J Gen Virol 73(10):2653–2660
Maugeri Z, Dominguez de Maria P (2012) Novel choline-chloride-based deep-eutectic-solvents with renewable hydrogen bond donors: levulinic acid and sugar-based polyols. RSC Adv 2(2):421–425. doi:10.1039/C1RA00630D
Nootchanat S, Thammacharoen C, Lohwongwatana B and Ekgasit S (2013) Formation of large H2O2-reduced gold nanosheets via starch-induced two-dimensional oriented attachment. RSC Adv 3:3707–3716. doi:10.1039/C3RA22830D
Liu B, Xie J, Lee JY, Ting YP, Chen JP (2005) Optimization of high-yield biological synthesis of single-crystalline gold Nanoplates. J Phys Chem B 109(32):15256–15263. doi:10.1021/jp051449n
Civit L, Fragoso A, Hölters S, Dürst M, O'Sullivan CK (2012) Electrochemical genosensor array for the simultaneous detection of multiple high-risk human papillomavirus sequences in clinical samples. Anal Chim Acta 715:93–98. doi:10.1016/j.aca.2011.12.009
Peng H, Soeller C, Cannell MB, Bowmaker GA, Cooney RP, Travas-Sejdic J (2006) Electrochemical detection of DNA hybridization amplified by nanoparticles. Biosens Bioelectron 21(9):1727–1736. doi:10.1016/j.bios.2005.08.011
Jampasa S, Wonsawat W, Rodthongkum N, Siangproh W, Yanatatsaneejit P, Vilaivan T, Chailapakul O (2014) Electrochemical detection of human papillomavirus DNA type 16 using a pyrrolidinyl peptide nucleic acid probe immobilized on screen-printed carbon electrodes. Biosens Bioelectron 54:428–434. doi:10.1016/j.bios.2013.11.023
Civit L, Fragoso A, O'Sullivan CK (2010) Electrochemical biosensor for the multiplexed detection of human papillomavirus genes. Biosens Bioelectron 26(4):1684–1687. doi:10.1016/j.bios.2010.06.072
Zari N, Amine A, Ennaji MM (2009) Label-free DNA biosensor for electrochemical detection of short DNA sequences related to human papilloma virus. Anal Lett 42(3):519–535. doi:10.1080/00032710802421897
Nasirizadeh N, Zare HR, Pournaghi-Azar MH, Hejazi MS (2011) Introduction of hematoxylin as an electroactive label for DNA biosensors and its employment in detection of target DNA sequence and single-base mismatch in human papilloma virus corresponding to oligonucleotide. Biosens Bioelectron 26(5):2638–2644. doi:10.1016/j.bios.2010.11.026
Tran LD, Nguyen DT, Nguyen BH, Do QP, Le Nguyen H (2011) Development of interdigitated arrays coated with functional polyaniline/MWCNT for electrochemical biodetection: application for human papilloma virus. Talanta 85(3):1560–1565. doi:10.1016/j.talanta.2011.06.048
Campos-Ferreira DS, Souza EVM, Nascimento GA, Zanforlin DML, Arruda MS, Beltrão MFS, Melo AL, Bruneska D, Lima-Filho JL (2016) Electrochemical DNA biosensor for the detection of human papillomavirus E6 gene inserted in recombinant plasmid. Arab J Chem 9(3):443–450. doi:10.1016/j.arabjc.2014.05.023
Sabzi RE, Sehatnia B, Pournaghi-Azar MH, Hejazi MS (2008) Electrochemical detection of human papilloma virus (HPV) target DNA using MB on pencil graphite electrode. J Iran Chem Soc 5(3):476–483. doi:10.1007/bf03246005
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 116 kb)
Rights and permissions
About this article
Cite this article
Karimizefreh, A., Mahyari, F.A., VaezJalali, M. et al. Impedimetic biosensor for the DNA of the human papilloma virus based on the use of gold nanosheets. Microchim Acta 184, 1729–1737 (2017). https://doi.org/10.1007/s00604-017-2173-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-017-2173-8